1.1 carbohydrates and 1.2 lipids
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
define monosaccharides and name the three hexose monosaccharides
organic monomer that cannot be hydrolyzed to a simpler sugar
hexose: glucose, fructose, galactose
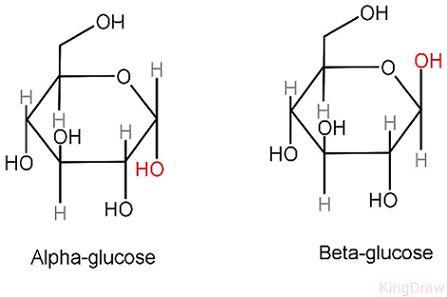
structure of alpha and beta glucose
alpha: down up down down
beta: down up down up
relate the structure of glucose to its functions
small and water soluble: easily transported
compare the structure of a disaccharide with glycogen
similarities:
both contain C,H,O
both contain glycosidic bonds
both contain glucose
differences:
glycogen contains 1,4 and 1,6 glycosidic bonds whereas disaccharides only contain 1,4
glycogen only contains glucose where as disaccharides contain glucose and other monosaccharides
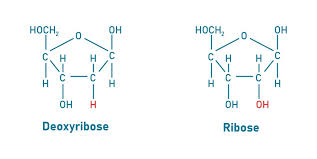
structure of ribose and deoxyribose
ribose: pentose, monosaccharide
deoxyribose: hexose, monosaccharide
condensation and hydrolysis
condensation: chemical bond forms between 2 molecules and a molecule of water is produced
hydrolysis: water molecule is used to break a chemical bond between 2 molecules
what type of bond forms when monosaccharides from?
glycosidic bond
2 monomers: disaccharide
2 or more monomers: polysaccharides
3 disaccharides and how they form
sucrose: fructose and glucose
maltose: glucose and glucose
lactose: galactose and glucose
polysaccharide and 3 examples
made from repeating monosaccharide unit:
starch
glycogen
cellulose
structure and function of starch
storage polymer in plant cells ( they are large and insoluble ) → prevents affecting osmosis
compare amylose and amylopectin
amylose:
1,4 glycosidic bond
straight chain
unbranched
compact
amylopectin:
1,4 and 1,6 glycosidic bond
branched (rapid hydrolysis can happen to release glucose)
similarities:
both contain alpha glucose
both contain glycosidic bond
structure and function of glycogen
main storage polymer in animal cells
1,4 and 1,6 glycosic bond
branched
insoluble
compact
structure and function of cellulose
polymer that gives rigidity to plant cell walls
1,4 glycosidic bonds
straight chain
unbranched
high tensile strength
many hydrogen bonds hold molecules together to prevent cell lysis
polar nature of glucose allows water to diffuse through
triglycerides formation
condesnation reaction between 1 molecule of glycerol and 3 fatty acids to form ester bonds
compare saturated and unsaturated fatty acids
saturated:
contains only single bonds
straight chain molecules
high melting point (solid)
found in animal fats
unsaturated:
contain C=C double bonds
‘kinked’ spread wider apart
lower melting point (liquid) → weaker intermolecular forces
found in plant oils
structure and function of phospholipids
glycerol backbone attached to hydrophobic fatty acid tail and hydrophilic polar phosphate head
phospholipid bilayer in water that is a component of cell membranes
waterproofing
polysaccharides formation
glycogen and starch formed by condensation of alpha glucose
cellulose formed by condensation of beta glucose
test for reducing and non reducing sugars
add benedicts solution to test for reducing sugars and heat
compare next to chart for the colour change to see concentration
add HCL into the non reducing sugar solution and heat
this neutralises the HCl (making the non reducing sugar solution neutral or alkali
then add the same volume and concentration of benedict’s test and compare next to chart to see colour change and concentration
devise and experiment to confirm the relative sweetness of sugars
same concentration of sugar used
tested by same person
sweetness compared to sucrose
water or buscuit in between tasting
compare glucose and fructose
similarities:
both are hexose
both contain covalent bonds (basically the glycosidic bond)
differences:
glucose is hexagon fructose is pentagon
glucose has one CH2Oh and fructose has two
advantages of having fructose in seminal fluid
source of energy for sperm
because movement requires ATP
in order to reduce competition
why lipids are used as energy storage and insulation
non polar
insoluble
don’t conduct heat because loosely packed large molecules
method to see if length of fatty acid chaings and degree of saturation affect melting point
heat solid samples and measure temp at which they melt
use fatty acids of the same chain length but with different C=C double bonds
use fatty acid of different chain length but same number of single bonds
repeat and calculate mean
properties of lipoproteins that enable cholesterol and fatty acids to be transported in blood
cholesterol are hydrophobic
phospholipids tails are hydrophobic so they can be carried inside the lipoprotein
lipoprotein are hydrophilic so they can transport in the bloodstream
why glycogen releases energy more slowly than glucose
glycogen is a polymer
so glycosidic bonds need to be broken
why are triglycerides a good energy store
carbohydrates and proteins are hydrophillic
triglycerides are hydrophobic
therefore the water will want to associate with proteins and carbohydrates but would be repelled by triglycerides