Elements VS Compounds VS Mixtures
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
pure substance
composition is same throughout, formed with one type of matter. EX- Element or Compound
homogeneous mixture
a mixture that does not have distinguishable parts; looks the same throughout

heterogeneous mixture
a mixture that has visibly different parts; does NOT look the same throughout

Element
A substance consisting of one type of atom. All the atoms in an element have the same number of protons.
Element Examples
EX- O2, Fe, Mg, N2
Compound
Two or more different elements chemically combined ex: KCl
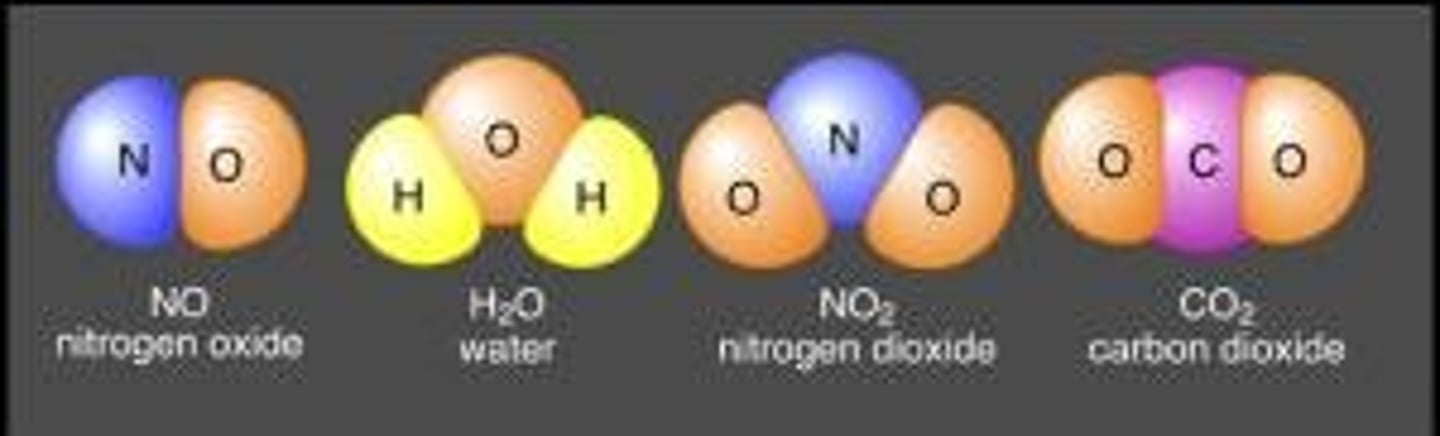
Compound Examples
NaCl (Salt), C6H12O6, H20(Water), CO2(Carbon Dioxide)
Solution
homogeneous mixture where a solute is dissolved in a solution.
heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

heterogeneous mixture

homogeneous mixture

Element

homogeneous mixture

homogeneous

Compound
H2O

homogeneous

Picture of a Compound Example
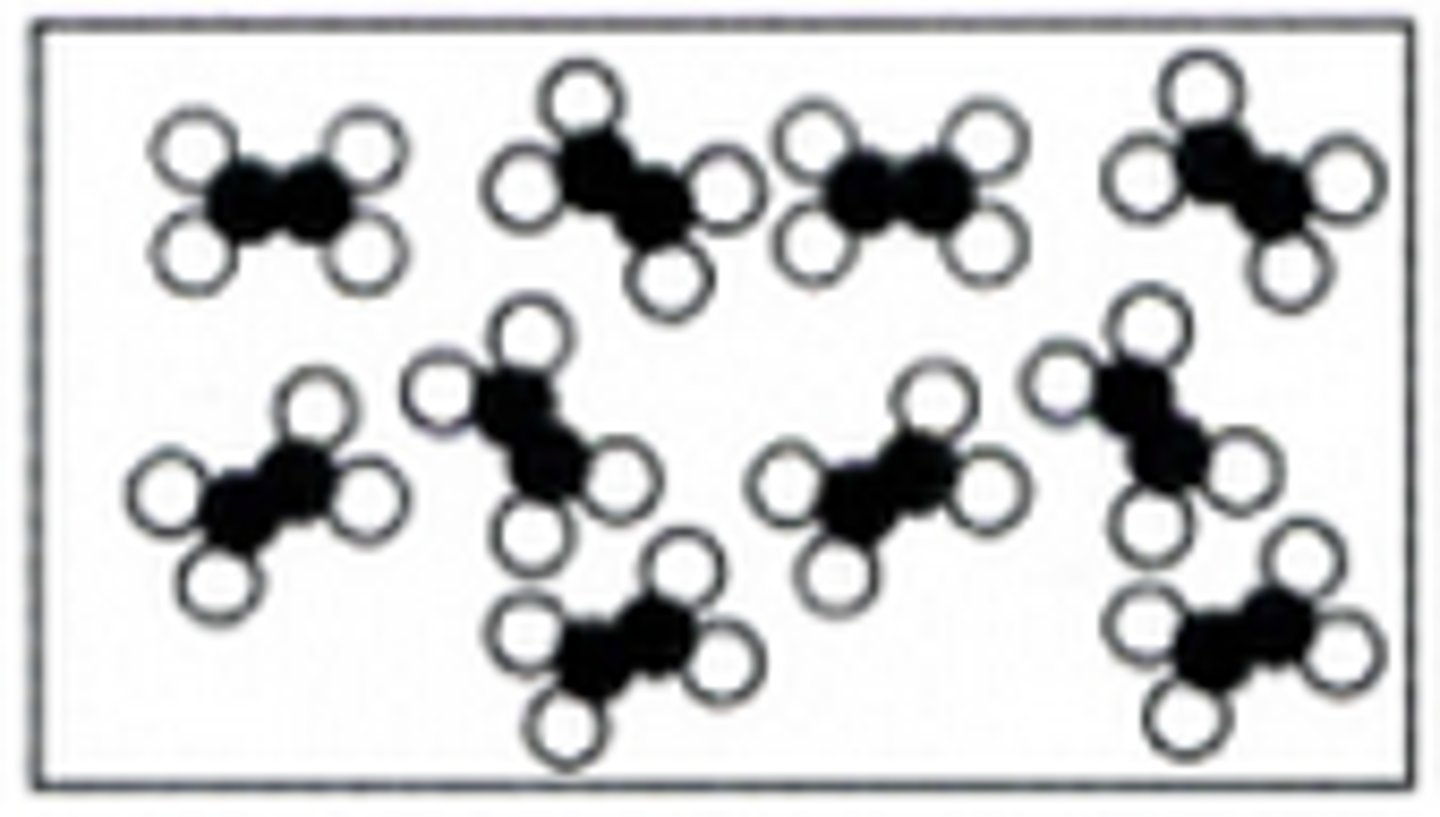
Picture of a Mixture Example
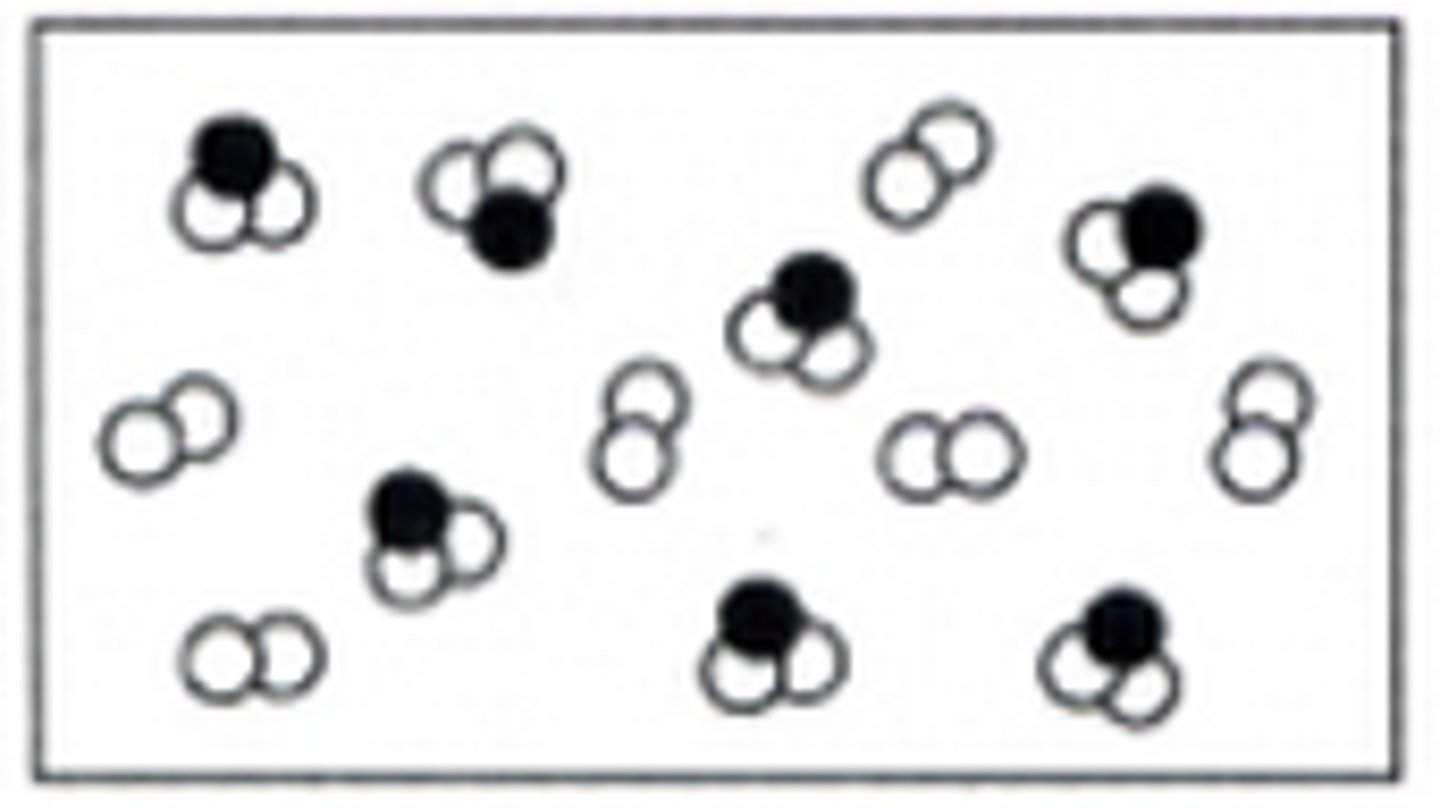
Picture of a Element Example

Element

Compound

Element

Molecule
1 single (unit) group of atoms bonded together. . Example is CO2 or H3
Compound
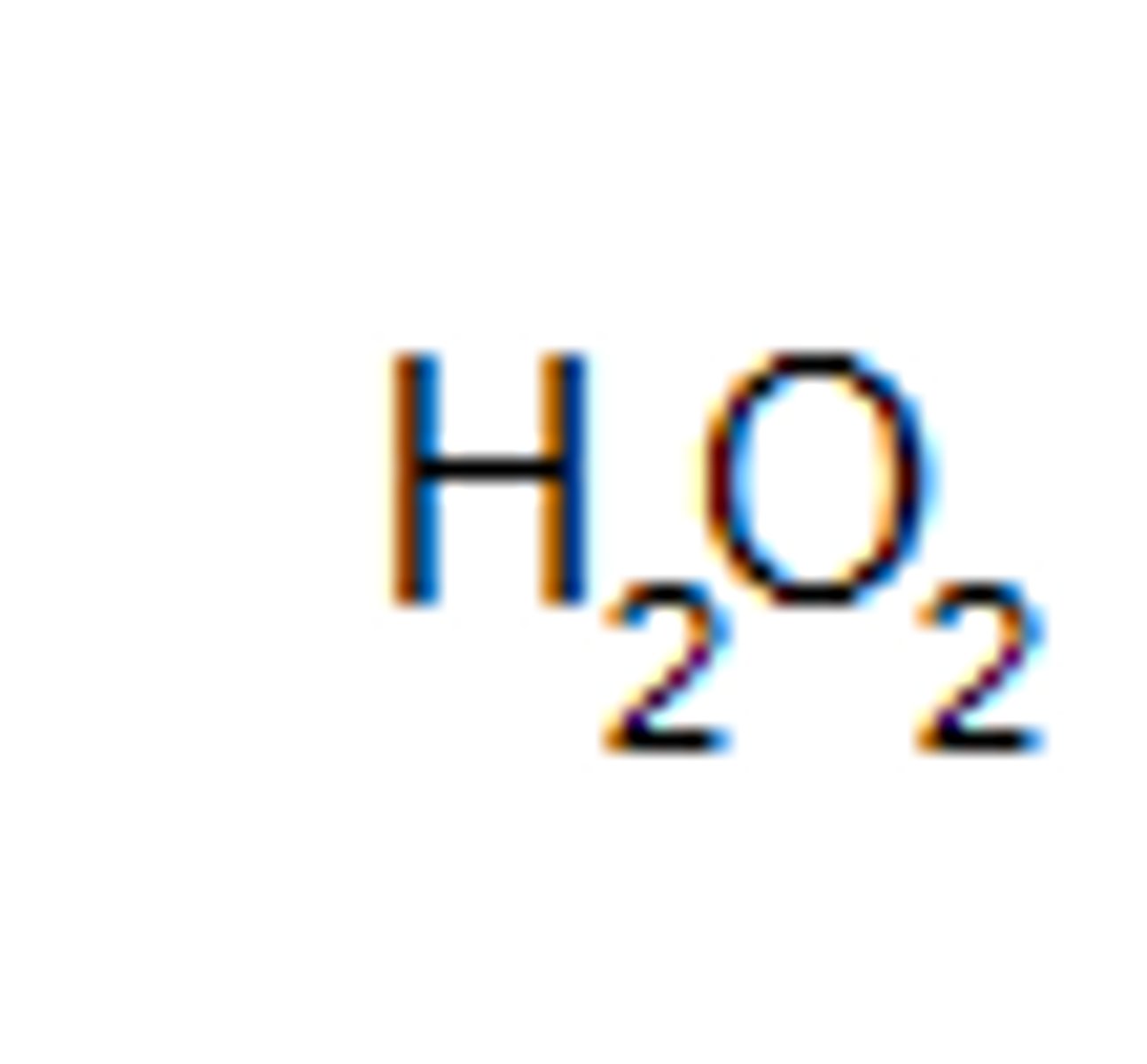
Compound
