neuro devl final good
1/154
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
155 Terms
What is Donald Hebb’s hypothesis regarding synaptic connections?
"Neurons that fire together, wire together."
what does the phrase "Neurons that fire together, wire together." mean
Coordinated activity between a pre- and post-synaptic cell strengthens their connection, whereas uncoordinated activity weakens it
How does neuronal activity influence cell survival during early development?
Activity is crucial for cell and connection survival. If a connection is uncoordinated and weakened, that cell or synapse is more likely to undergo apoptosis (programmed cell death)
What happens if you use curare to block the activity of developing motor neurons?
Blocking neuromuscular junction activity with curare actually halts motor neuron apoptosis. The loss of activity tricks the muscle fibers into believing they have no innervation, so they release more neurotrophic factors, which prevents cell death
What is the difference in muscle fiber innervation between a newborn and an adult?
At birth, a single muscle fiber has polyneuronal innervation (multiple axons innervating it). By adulthood, each fiber is innervated by only one axon (1:1 ratio).
How does muscle fiber innervation change from birth to maturity?
At Birth (Poly-innervation): Each muscle fiber is connected to multiple motor neurons.
The Process (Synapse Elimination): Excess axonal branches retract during the first few weeks of life.
At Maturity (Monomodal): Each fiber is innervated by only one motor neuron.
The Result: The remaining connection grows larger and stronger to ensure reliable muscle contraction.
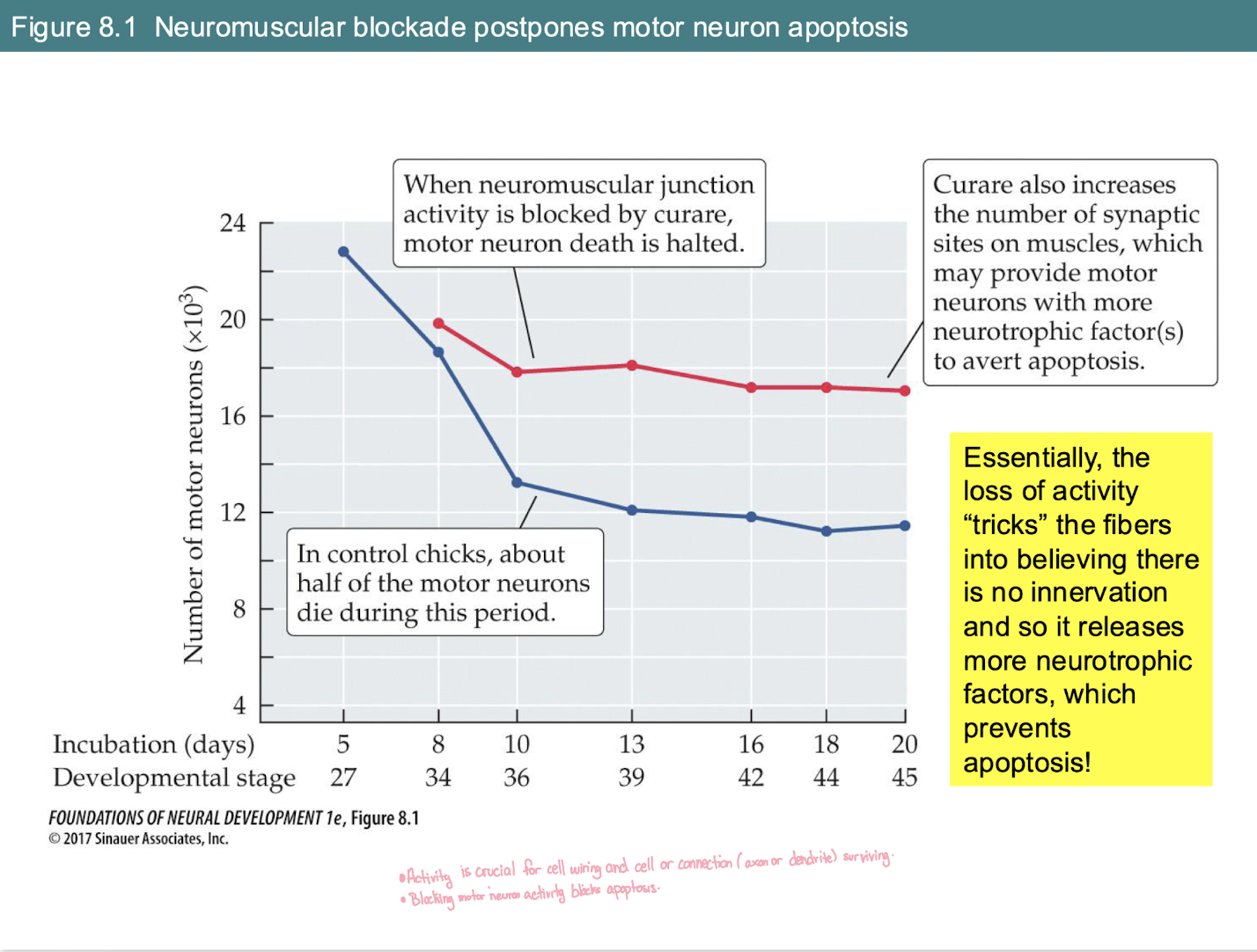
What is the effect of blocking neuromuscular activity (e.g., with Curare) on motor neuron survival during development?
Normal Condition (Blue line): Naturally occurring apoptosis (cell death) kills about half of motor neurons as they compete for targets.
With Blockade (Red line): Motor neuron death is halted/decreased. More neurons survive.
The Mechanism: Blocking activity "tricks" muscle fibers into thinking they aren't innervated.
The Result: Muscles release more neurotrophic factors, which provide the "survival signal" needed to prevent apoptosis.
Functionally, why is it important that an adult has a 1:1 axon-to-muscle fiber ratio?
t allows for incremental control and precision. Instead of all fibers firing at once, the brain can recruit specific numbers of axons to activate a specific number of muscle fibers, granting precise control over movement strength
How does the EPSP (excitatory postsynaptic potential) response in muscle fibers differ between newborns and adults when stimulating multiple axons?
In newborns, stimulating multiple axons incrementally increases the EPSP amplitude because many axons target the same fiber. In adults, stimulating multiple axons recruits more muscle fibers, but the response on a single fiber is all-or-nothing.
What happens to the "losing" axons during synapse elimination?
The losing axons form a retraction bulb and pull away from the junction. This is an activity-based process, not apoptosis.
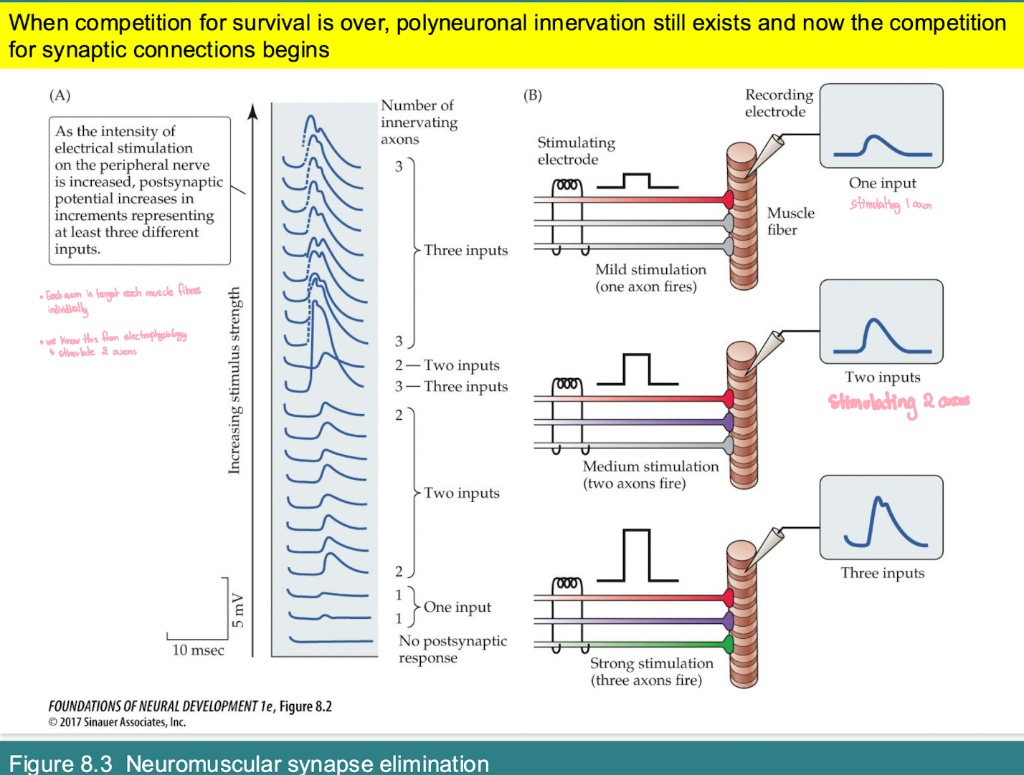
In early development, why does the postsynaptic potential (PSP) increase in distinct "steps" as stimulation strength increases?
The "Steps": Each jump in the wave's amplitude represents the recruitment of an additional axon.
What it proves: It shows that multiple different motor neurons (e.g., 1, 2, or 3) are all connected to the same single muscle fiber.
Mechanism: * Mild Stim: Fires 1 axon (small response).
Stronger Stim: Fires 2 or 3 axons (larger, additive responses).
While polyneuronal innervation is lost over time, what happens to the remaining successful synapse?
The remaining single connection becomes much more extensive. It forms larger contacts and covers significantly more area on the target cell.
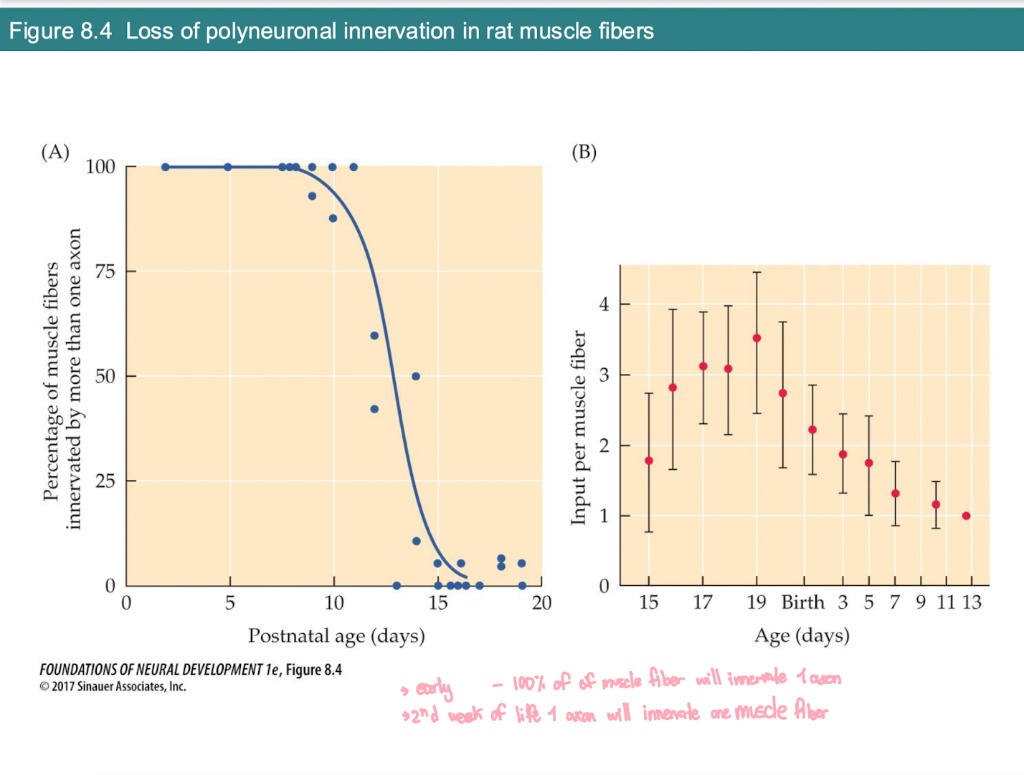
According to Graph A, when does the transition from polyneuronal to single innervation primarily occur in rats?
0–10 Days: 100%of fibers have multiple axons (polyneuronal).
The "Drop" (Days 10–15): A rapid loss of extra connections.
By Day 15+: Nearly 0% of fibers have more than one axon; maturation is complete.
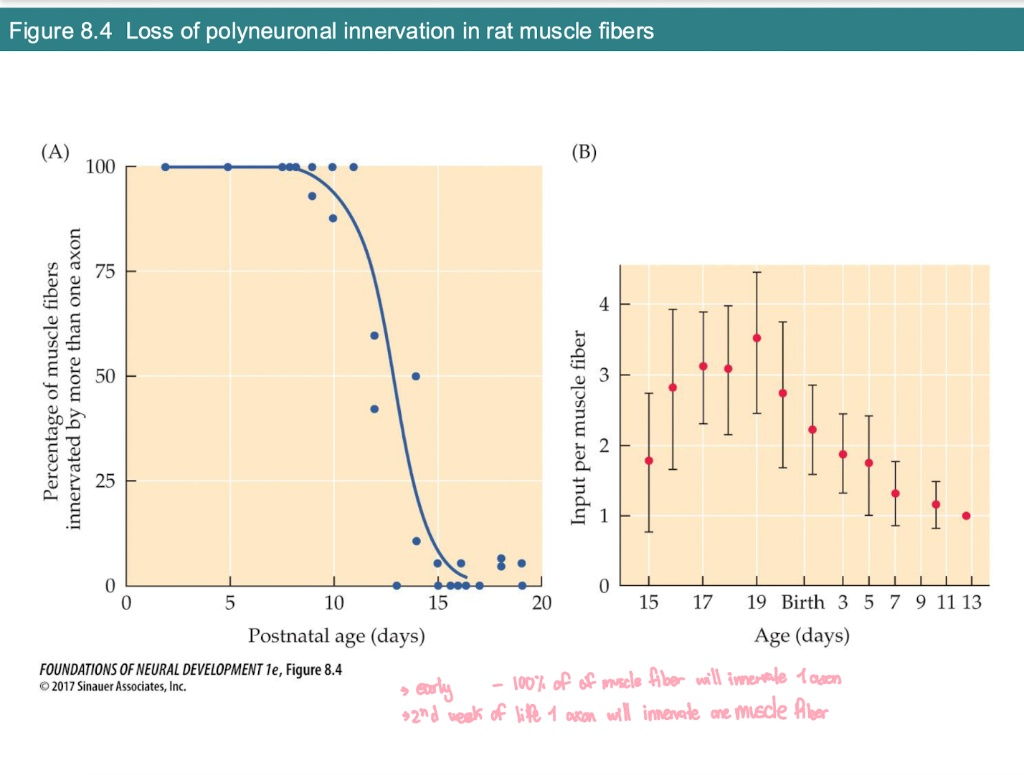
According to Graph B, how does the number of inputs per muscle fiber change before and after birth?
Prenatal (Before Birth): The number of inputs increases as axons find their targets (peaking at ~3.5 inputs).
Postnatal (After Birth): The number of inputs decreases steadily.
Final Goal: It reaches exactly 1 input per fiber by about two weeks after birth.
How does the development of climbing fibers innervating Purkinje cells in the cerebellum mirror muscle fiber development?
Similar to muscles, a newborn's Purkinje cell receives multiple innervations from climbing fibers (originating in the inferior olive). By postnatal day 15, the synapse is eliminated down to a single, extensive innervation per Purkinje cell.
In the developing cortex, what is the relationship between dendrites and innervating axons?
The number of innervating axons matches the complexity of the dendrites. More dendrites equal more axonal inputs
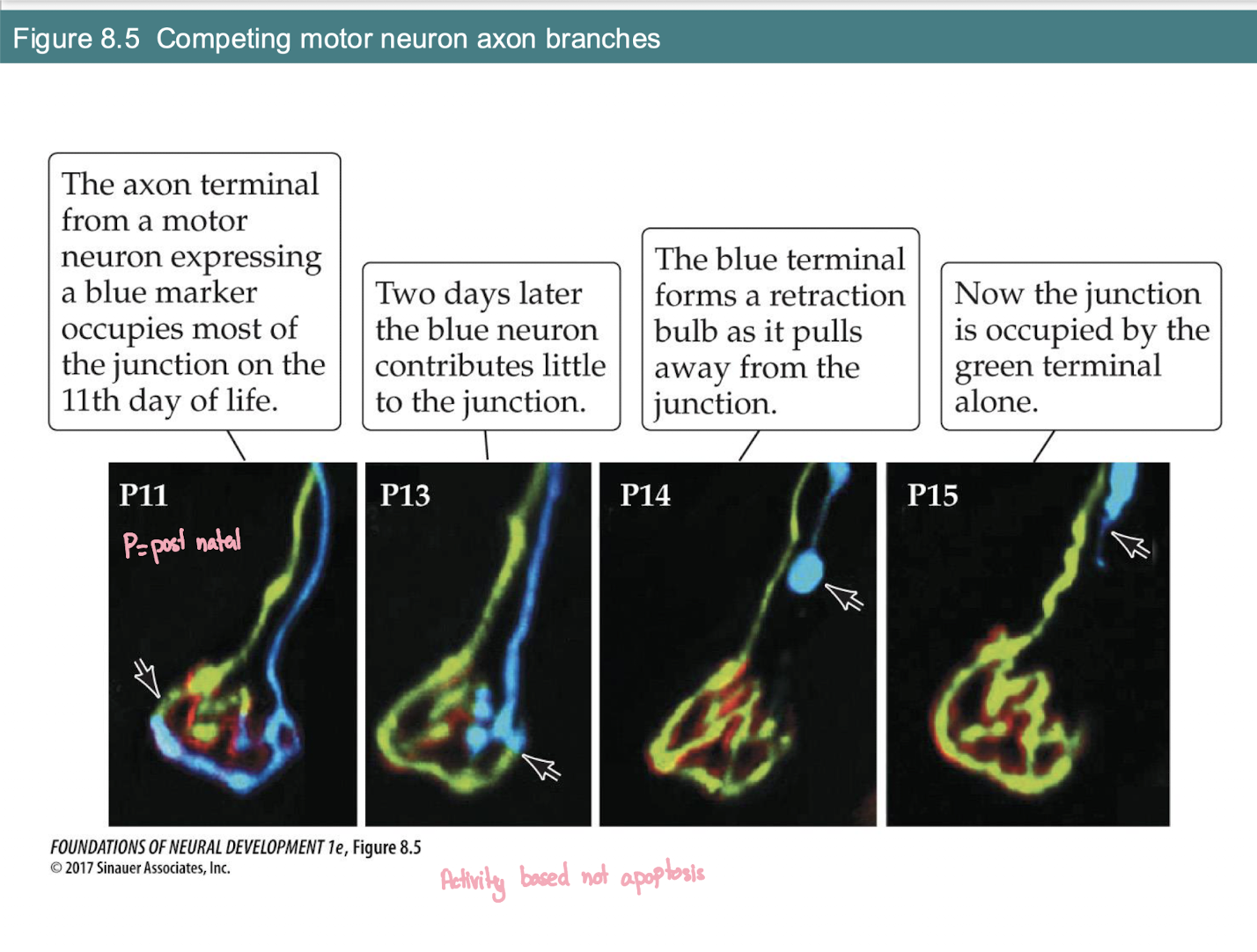
What is the structural sign that an axon is losing the competition at a neuromuscular junction?
The Process: As one axon takes over (Green), the losing axon (Blue) shrinks and pulls away.
Retraction Bulb: The blue axon forms a distinct swollen bulb as it detaches (seen at P14).
Final Result: By P15, the junction is occupied by a single terminal alone.
Key Mechanism: This is Activity-Based competition, not apoptosis (the neuron lives; it just loses this specific branch).
What are the two structural ways a Hebbian synapse can be strengthened between Cell A and Cell B?
1) Cell A increases its synaptic area (wider terminal) for more coverage on Cell B.
2) Cell A forms more synaptic terminals on Cell B. (Note: Both of these mechanisms occur)
How does the innervation of Purkinje cells by Climbing fibers change from birth to adulthood?
At Birth (Newborn): Multiple climbing fibers innervate a single Purkinje cell (Poly-innervation).
Adult: Only one climbing fiber innervates each Purkinje cell.
when does Pruning of Purkinje, specifically, the elimination of climbing fiber inputs. cells happen
Pruning occurs rapidly between Day 5 and Day 15 (Postnatal).
How can you tell an Adult Purkinje cell response from a Newborn response on an electrophysiology graph?
Adult (All-or-Nothing): Increasing stimulus strength does not change the amplitude; there is only one input
Newborn (Graded): Increasing stimulus causes the amplitude to jump in "steps"; this proves multiple inputs are being recruited
What is Long-Term Potentiation (LTP)?
An increase in the amplitude of an EPSP in a postsynaptic neuron that lasts for hours, days, or longer, occurring in response to a strong burst of electrical stimulation (tetanus) at a synapse.
What behavior is LTP considered the neural substrate for?
Associative learning and memory, especially during early development
What are the three major anatomical shifts in Climbing Fiber (CF) innervation from P7 to P20?
P7 (Multi-Target): One CF axon (red) innervates several different Purkinje cell bodies (asterisks).
P15 (Single Target): The axon has retracted from all but one Purkinje cell.
P20 (Dendritic Shift): The axon moves from the cell body to the dendrites of that single cell.
Key Driver: This process is activity-dependent (increased stimulation speeds it up; blocking activity slows it down).
What are the three synaptic pathways in the hippocampal formation that display LTP?
1) Perforant pathway (entorhinal cortex to dentate gyrus)
2) Mossy fiber pathway (dentate gyrus to CA3)
3) Schaffer collaterals (CA3 to CA1)
When LTP is successfully induced, why does the postsynaptic cell become more sensitive?
It requires less input from the presynaptic cell to fire. The EPSP amplitude increases because more neurotransmitter is released AND the postsynaptic membrane has more receptors.
How does the innervation pattern of target cells in autonomic ganglia change from birth to maturity?
At Birth: Each target cell has polyneuronal innervation (connected to multiple different axons).
In Maturity: Polyneuronal innervation is lost; each cell is innervated by a single axon.
The Trade-off: While axons are lost, the remaining axon forms a much more extensive network of synapses on the cell.
what is tetanus
A "strong burst" of activity that is applied to the presynaptic neuron. which causes The amplitude of the EPSP in the postsynaptic cell significantly increases.
where those LTP occur
in the hippocampus; it is the cellular basis for learning and memory.
whats the difference between LTP and action potential
the increase is long-lasting (persisting for hours or days).
What are the three pathways in the hippocampal formation that display LTP (Long-Term Potentiation)?
Perforant Pathway: Entorhinal cortex → Dentate Gyrus.
Mossy Fiber Pathway: Dentate Gyrus → CA3 pyramidal cells.
Schaffer Collaterals: CA3 → CA1 pyramidal cells.
What is the primary input pathway to the hippocampus?
The Perforant Pathway:
Source: Entorhinal Cortex.
Target: Dentate Gyrus.
Key Fact: The Dentate Gyrus generates new neurons even in adults.
What is the Mossy Fiber Pathway?
Route: Dentate Gyrus → CA3 pyramidal cells.
Significance: The second leg of the hippocampal circuit; essential for spatial learning and LTP.
What are the Schaffer Collaterals?
Route: CA3 pyramidal cells → CA1 pyramidal cells.
Significance: The final leg of the "trisynaptic loop" before information is sent out of the hippocampal formation.
What are the two types of ionotropic glutamate receptors critical for LTP?
AMPA receptors and NMDA receptors.
How do AMPA receptors function during normal, weak synaptic transmission?
They respond to glutamate by opening and allowing Sodium (Na+) into the cell, causing slight depolarization
Why is the NMDA receptor blocked at resting potential, and how is the block removed?
It is blocked by a Magnesium (Mg2+) ion resting in the pore.
A strong electrical stimulus (tetanus) depolarizes the postsynaptic membrane, which repels and displaces the Mg2+ block
What two events must occur simultaneously for the NMDA receptor to open?
Depolarization of the postsynaptic membrane (to displace Mg2+).
Activation by glutamate released from the presynaptic neuron.
Once the NMDA receptor is fully open, what vital ion rushes into the postsynaptic cell?
Calcium (Ca2+)
During weak stimulation, why is the NMDA receptor inactive?
The Block: Its pore is physically plugged by a Magnesium ion ($Mg^{2+}$).
Result: Glutamate can only activate AMPA receptors.
Effect: Small EPSP, no long-term change.
Once calcium enters through the NMDA receptor, how does the synapse become stronger?
Second Messengers: Ca2+ triggers internal signals.
Physical Change: More new AMPA receptors are inserted into the membrane.
Final Result: The synapse is now more sensitive to glutamate (LTP is achieved).
What intracellular cascades does Calcium trigger once inside the postsynaptic cell?
Ca2+ activates protein kinases (including PKC, PKA, and CaMKII).
How does the NMDA receptor help induce Long-Term Potentiation (LTP)?
Repeated Stim: Glutamate activates AMPA receptors, depolarizing the cell.
Unlocking: Depolarization drives the Mg2+ ion out of the NMDA channel.
Influx: Calcium enters through the NMDA receptor.
Strengthening: Ca2+ activates kinases (like CaMKII) to insert more AMPA receptors and release retrograde signals to the axon.
What is the role of retrograde messengers (like Nitric Oxide) in LTP?
Source: Postsynaptic dendritic spine.
Target: Presynaptic axon terminal.
Function: Tells the axon to increase neurotransmitter release, ensuring both sides of the synapse get stronger simultaneously.
How does CaMKII activation lead to long-term structural changes in the synapse?
It triggers CREB to bind to gene promoter regions in the nucleus, altering gene transcription. This creates new AMPA receptors that are inserted into the postsynaptic membrane, making it permanently more sensitive.
How does the postsynaptic cell tell the presynaptic cell to release more glutamate in the future?
Through retrograde messengers (like nitric oxide and arachidonic acid) that travel backward to the presynaptic terminal.
What behavioral task is commonly used to test non-spatial memory in rodents?
The Novel Object Recognition Task. It relies on a rodent's natural preference to investigate new, unfamiliar objects over familiar ones
What is the correlational evidence linking LTP to memory in transgenic mice?
If you block NMDA receptors, you block LTP, and the mouse fails the novel object task. If you overexpress NMDA receptors, LTP increases, and the mouse shows superior learning (though they suffer from seizures).
How does blocking neuromuscular activity (e.g., with Curare) affect motor neuron survival?
Normally: About half of motor neurons die via apoptosis due to competition.
With Blockade: Motor neuron death is halted (more survive).
Mechanism: "Quiet" muscles release more neurotrophic factors, providing extra survival signals to the neurons.
On an electrophysiology recording, what does a "step-wise" increase in EPSP amplitude indicate?
Evidence: It proves multiple axons are innervating the same fiber.
Mechanism: As stimulation strength increases, you recruit axons one by one (Step 1 = 1 axon, Step 2 = 2 axons, etc.).
Adult Status: Once mature, the "steps" disappear because only one axon remains.
How does NMDA receptor activation lead to permanent synaptic changes?
Ca2+ Influx: Activates protein kinases (CaMKII, PKA, PKC).
Gene Expression: Kinases activate CREB protein in the nucleus.
Protein Synthesis: New enzymes and structural proteins are made.
Structural Change: Leads to sprouting new dendritic branches to receive more synapses.
In the visual system, where does the brain first integrate input from both eyes to create a single "whole" image?
The Site: The Primary Visual Cortex (V1).
The Flow: Input first enters Layer 4, which then projects to other cortical layers.
The Organization: Left visual field goes to the Right hemisphere; Right visual field goes to the Left hemisphere.
What is the universal goal of Synapse Elimination in the Muscle, Cerebellum, and Autonomic Ganglia?
The Shift: Moving from Polyneuronal (many-to-one) to Monomodal (one-to-one) innervation.
The Purpose: To increase precision and control.
The Trade-off: We lose the number of different axons but gain much larger, more extensive synaptic contacts from the winner.
Is synapse elimination a pre-programmed event or is it influenced by the environment?
Activity-Dependent: It is driven by neural activity.
Increased Activity: Hastens/speeds up the elimination of extra synapses.
Blocked Activity: Forestalls/slows down the process (keeping "messy" connections longer).
What is the final anatomical step for a Climbing Fiber in the adult cerebellum?
The Move: The fiber shifts its contact from the Purkinje cell body to the Purkinje cell dendrites.
Timing: This transition is usually complete by Postnatal Day 20 (P20).
Where is visual input from both eyes first integrated to create a "whole" image?
Visual input travels from the Lateral Geniculate Nucleus (LGN) of the thalamus and integrates in Layer IV of the Primary Visual Cortex (V1)
How are inputs from the two eyes organized in the Lateral Geniculate Nucleus (LGN) of the thalamus?
Strict Separation: Axons from each eye project to distinct, separate layers (they don't mix yet).
Layers 1, 4, 6: Receive input from the Contralateral eye (opposite side).
Layers 2, 3, 5: Receive input from the Ipsilateral eye (same side).
In the context of the visual pathway, what do Ipsilateral and Contralateral mean?
Ipsilateral: Coming from the eye on the same side as the brain hemisphere (e.g., right eye to right LGN).
Contralateral: Coming from the eye on the opposite side of the brain hemisphere (e.g., left eye to right LGN).
The Crossing: Contralateral fibers cross over at the Optic Chiasm.
What happens to the "eye-specific" signals once they move from the LGN to Layer 4 of the Primary Visual Cortex (V1)?
Ocular Dominance Columns: The separate signals from the LGN create "stripes" or columns in Layer 4.
The Integration: Layer 4 is the first place where these two separate streams start to be integrated into a single binocular image.
What are Ocular Dominance Bands?
Distinct, alternating stripes/patches in Layer IV of the visual cortex. Each band receives monocular input exclusively from either the left or the right eye.
How do Ocular Dominance Bands form from birth to adulthood?
In newborns, inputs from both eyes overlap considerably in Layer IV. Through development, visual experience, and spontaneous activity, they segregate into sharp, alternating monocular bands.
What cellular rule drives the segregation of visual inputs into Ocular Dominance Bands?
Synchronous activity. Neighbouring retinal cells in the same eye fire synchronously, driving the postsynaptic cell, thereby strengthening their synapses. Ineffective, out-of-sync inputs from the other eye are lost.
Does the massive increase in human cerebral cortex complexity postnatally come from new cells, axonal pathfinding, or dendritic branching?
Dendritic branching (proliferation). Axons are done migrating; the complexity comes from dendrites acting like tree branches, creating incredibly complex arborizations
Describe the trajectory of synaptic density across a human lifespan.
Synaptic density peaks early in life (visual/auditory cortex prune after age 1, prefrontal cortex peaks a bit later). After age 30, there is a slow loss of dendritic spines across all cortical layers.
How is myelination influenced by neural activity?
Myelination is activity-dependent. Vesicle release from the axon stabilizes the axo-glial junction with the oligodendrocyte. If you block activity (e.g., using tetanus toxin), myelin sheaths retract or fail to form.
How is transsynaptic transport used to label the pathway from the retina to the visual cortex?
Uptake: Labeled amino acids are injected into the Retina and incorporated into proteins.
Anterograde Transport: Proteins travel down the axon to the LGN (Lateral Geniculate Nucleus).
Transneuronal Jump: The label is released by the first neuron and taken up by the second.
Target: The label finally reaches terminals in Layer IV of the Visual Cortex.
How does strong NMDA receptor activation lead to long-term structural changes?
Step 1: Strong stimulation leads to a massive $Ca^{2+}$ influx.
Step 2: $Ca^{2+}$ activates protein kinases (like CaMKII and PKA).
Step 3: These kinases activate CREB protein in the nucleus.
Step 4: Gene transcription creates new proteins that sprout new dendritic branches to receive more synapses.
How can researchers label the pathway from the retina to the visual cortex?
Method: Inject labeled amino acids into the eye.
Transport: Labeled proteins move via anterograde axonal transport to the LGN.
Transneuronal Jump: The label crosses the synapse to reach Layer IV of the visual cortex.
How does the distribution of visual input in Layer IV change from birth to maturity?
Day 15: Input is evenly distributed across Layer IV.
Day 39-92: Input segregates into sharply defined bands (Ocular Dominance Columns).
Mechanism: Likely experience-mediated, moving from overlapping to completely monocular bands.
How is input from the two eyes organized in the LGN?
Separation: Axons from each eye project to distinct, separate layers.
Ipsilateral (Same side): Layers 2, 3, and 5.
Contralateral (Opposite side): Layers 1, 4, and 6.
Where is the first place that inputs from both eyes are integrated to create a "whole" image?
Location: The Primary Visual Cortex (V1).
Process: Input first enters Layer 4 and then projects to other cortical layers for integration.
How does visual input in Layer IV of the cortex change from birth to maturity?
Birth (Day 15): Input from one eye is evenly distributed across the layer.
Maturity (Day 92): Inputs segregate into sharply defined bands (Ocular Dominance Columns).
Result: Neurons in these bands receive monocular input (from only one eye).
What happens to LGN segregation if spontaneous neural activity is blocked?
Normal: Inputs from both eyes segregate into distinct red/green regions by Day 10.
Blocked Activity: Segregation fails; binocular overlap (yellow) is retained.
Single Eye Block: If one eye is blocked, the active eye takes over the LGN completely.
How do we trace the path from the retina to the visual cortex?
Injection: Radioactive amino acids are put into the eye.
Transport: Proteins move via anterograde transport to the LGN.
The Jump: The label crosses the synapse (transneuronal) to reach Layer IV of the cortex.
What is the role of Layer 4 in the Primary Visual Cortex (V1)?
The Entrance: This is where visual input from the LGN first arrives.
The Projector: Layer 4 then projects to other cortical layers.
The Result: This is where inputs from both eyes are integrated to create a "whole" image.
How do synaptic changes relate to learning?
Synaptic changes alter neural circuits, which changes network structure and function → enabling learning and adaptation.
Is neural development purely spontaneous?
No, it is experience-dependent—activity driven by experience shapes neural circuits.
What happens if critical periods are missed?
Synaptic pruning occurs, limiting the ability to form new connections → reduced plasticity.
Why is experience important for visual perception?
The brain must learn to interpret visual input; without experience, proper recognition does not develop.
What happened in cases of visual recovery in adulthood (e.g., after blindness)?
Individuals could not properly recognize objects because:
Critical period had passed
Synaptic pruning had already occurred
Visual cortex had been repurposed
Why can't the brain fully recover visual function after late intervention?
Neural circuits are already reorganized and lack the plasticity needed to relearn visual processing.
What happened when a person wore upside-down glasses?
Over time, they adapted and could function, but slower than normal.
What is the chemoaffinity hypothesis?
Axons find their target cells using chemical signals (ligand-receptor matching).
What role do growth cones play?
They guide axons to their targets by detecting chemical cues.
Why is chemoaffinity alone insufficient?
One signal is too simple
Multiple cues are required
Activity and experience also shape connections
How is frog vision different from human vision?
Mostly contralateral projections
Very little ipsilateral input
No binocular vision
Can frogs regenerate retinal cells?
Yes—even in adulthood.
What was the key finding of the three-eyed frog experiment?
Axons projected to correct targets (supporting chemoaffinity), but formed segregated bands.
What are ocular dominance bands?
Striped regions in the brain where inputs from each eye are segregated.
What causes formation of ocular dominance bands?
Activity-dependent competition between inputs from each eye.
What happens if visual experience is blocked (e.g., dark rearing)?
No competition
No segregation
Bands do not form
What was the effect of blocking NMDA receptors (APV)?
Prevented segregation → shows activity-dependent plasticity is required.
What is strabismus?
Misalignment of the eyes.
How does early strabismus affect vision?
Reduces binocular vision due to lack of coordinated input.
When must strabismus be corrected for normal vision?
Before ~2 years of age (critical period).
What happens if corrected later?
Poor binocular vision permanently.
What happens if one eye is closed during the critical period?
Neurons respond only to the open eye
Closed eye loses cortical representation
What if both eyes are closed during development?
Neurons respond randomly
Visual system fails to mature properly
What do ocular dominance histograms show?
Distribution of neuron responses to each eye.
What is seen in normal development?
Balanced response to both eyes.