POST LAB EXPT 4
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
Enzymes
mostly protein molecules that act as catalysts for biochemical reaction
very specific; each enzyme catalyzes a certain reaction or type of reaction only
Catalysts
increase the rate of reaction without changing the free energy
Oxidoreductase
catalyzes redox reactions
Transferase
catalyzes the transfer of a specific chemical group from one molecule to another
Ligase
catalyzes the joining of 2 molecules
Hydrolase
catalyzes the breaking of bonds within a molecule by using water
Lyase
catalyzes the breaking of certain chemical bonds in molecules through mechanism other than hydrolysis or oxidation
Isomerase
catalyzes the conversion of a molecule into its isomer (rearrangement)
Active site
an enzyme contains an … that binds the substrate, forming an enzyme-substrate complex
Substrate
reactant; substance upon which the enzyme acts
Products
once the reaction has occurred, the catalyst releases the products
Life would not be possible
without enzymes, … since biochemical reactions would not occur as fast as they would
Enzyme activity
refers to the measure of the rate at which enzyme converts substrate to products in a biochemical reaction
Factors affecting enzyme activity
temperature
pH level
substrate concentration
enzyme concentration
presence of inhibitors
Objectives
learn the activity of salivary amylase in the digestion of carbohydrates
determine the effects of enzyme concentration in enzyme activity
determine the effects of pH in enzyme activity
determine the effects of inhibitors
Methodology
collection of saliva sample
subjecting samples to different conditions
dropping samples in a spot plate for iodine test
observation color changes at different time points
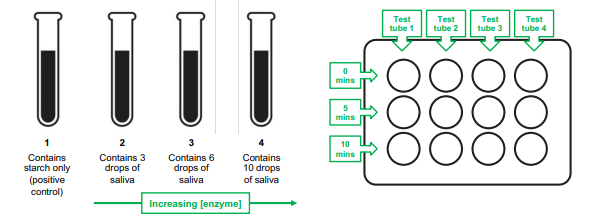
Effect of enzyme concentration (experimental set up)
test tube 1: contains starch only
test tube 2: contains 3 drops of saliva
test tube 3: contains 6 drops of saliva
test tube 4: contains 10 drops of saliva
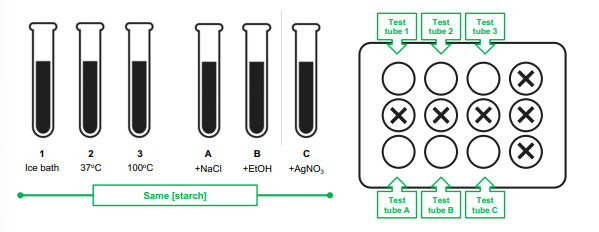
Effect of temperature and inhibitors (experimental set up)
test tube 1: ice bath
test tube 2: 37C
test tube 3: 100C
test tube A: +NaCl
test tube B: +EtOH
test tube C: +AgNO3s
Iodine test
Iodine reacts with starch to form a starch/iodine complex which gives a characteristic blue color to the reaction mixture
Practical iodine test application
iodine testing to analyze fruit ripening process (plant biology) and identify adequate dietary intake or overload of iodine (nutrition)
Dark blue back
amount of starch remaining: all
enzyme activity level: none (0)
Blue
amount of starch remaining: most
enzyme activity level: low (1)
Light brown
amount of starch remaining: some
enzyme activity level: moderate (2)
Gold
amount of starch remaining: none
enzyme activity level: high (3)
Starch
polysaccharide made up of glucose units and consists of 2 main components—amylose and amylopectin
Iodine
not very soluble in water as itself but forms triiodide (I3-) or pentaiodide (i5-) ions that slip into the helical structure of amylose
this causes a visible color change (characteristic blue black) indicative of the presence of starch in the samples
Effect of enzyme concentration
the rate of an enzyme catalyzed reaction increases with an increase in the concentration of an enzyme
more drops of saliva sample
more salivary amylase to break down starch
high enzyme activity
Longer incubation time
allows for more substrate molecule to be converted into product by the enzyme up to a certain point
Prolonged incubation
may also lead to enzyme denaturation (decreases enzymatic activity) due to extended exposure
pH
2, 4, 7, 10
Temperature
~10C (ice bath)
37C (body temperature)
100C (boiling water bath)
a-amylase
has an optimum pH of 7 (neutral) due to this being the pH of saliva which acts in the beginning of starch digestion
has an optimum temperature of 32C-37C because this mimics or is close to the normal human body temperature
deviations (higher or lower values than optimum) from this pH level or temperature may lead to a decrease in enzyme activity possibly either due to less interactions with the substrate or enzyme denaturation
At temperatures lower than the optimum
there may be decreased catalytic activity due to insufficient energy for collisions
At temperature higher than the optimum
enzyme denaturation may happen thus inactivating the enzyme
Small changes in the optimum pH value
can result in denaturation of enzymes since there can be acidic or basic AA residues in the enzyme
Protonation or deprotonation
may inactivate the enzyme
Inhibitors
1% NaCl
95% ethanol
AgNO3
Binding of inhibitors to the enzymes
decrease enzyme activity
1% NaCl
typically used as a reagent in buffer solutions for amylase assays
does not inhibit enzyme activity also due to its low concentration
95% ethanol
alcohols especially concentrated ethanol, inhibit a-amylase activity by disrupting the secondary structure of the enzyme—making it difficult for the enzyme to bind to and break down starch
AgNO3
silver nitrate acts as noncompetitive inhibitor of a-amylase by the interaction of the silver ions to the enzyme, altering its active site and preventing its interaction
also inhibits a-amylase activity by disrupting the secondary structure of the enzyme—making it difficult for the enzyme to bind and break down starch
No biological activity
decreased enzyme activity or enzyme inactivation
Denaturation
unraveling of the 3D structure of a macromolecule caused by the breakdown of noncovalent interactions that can result from:
heat
large changes in pH which alter charges on side chains
detergents, such as sodium dodecyl sulfate (SDS) which disrupt hydrophobic interactions
urea and guanidine hydrochloride, which disrupt hydrogen bonding
B-mercaptoethanol