chem 3.8 equilibrium constants
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
what state will reversible reactions eventually reach?
dynamic equilirbium
meaning of dynamic equilibrium?
rates of the forward and back reactions are equal and the composition of the mixture remains constant
what does le chatelier’s principle state?
if a system at equilibrium is changed then the position of equilibrium shifts to oppose the change
what are the two equilibrium constants?
Kc
Kp
what does Kc measure?
the amounts of reactants and products in terms of their concentrations
usually used for reactions in solutions
can also be used for gaseous reactions
what does Kp measure?
the amounts of reactants and products in terms of their partial pressures
used for reactions involving gases
what is the expression for Kc?

What conditions is Kc affected by?
affected by changes in temperature → causes increase or decrease depending on the sign of ΔH
unaffected by changes in pressure
unaffected by addition of catalyst
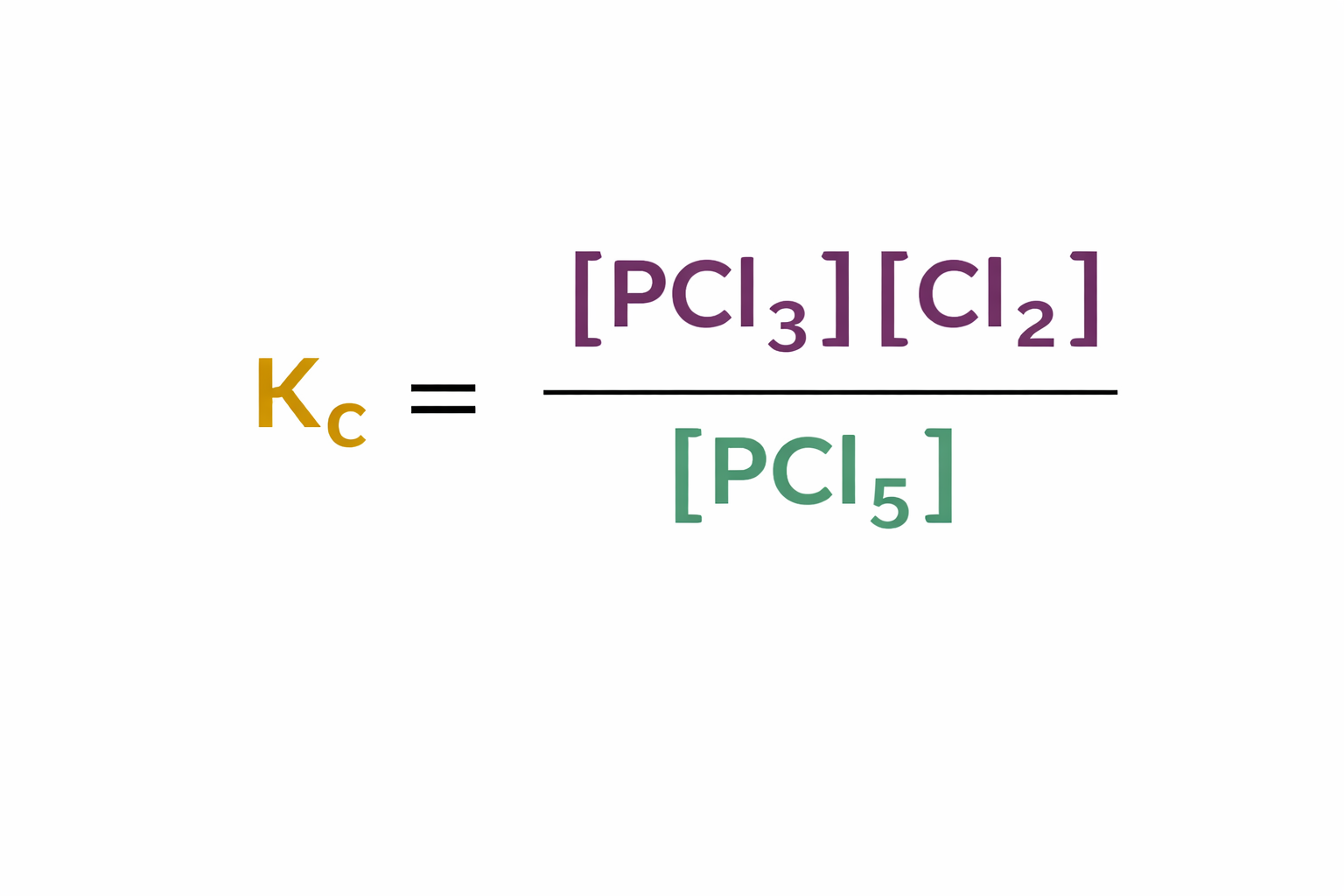
What are the units of Kc for this expression?
moldm-3
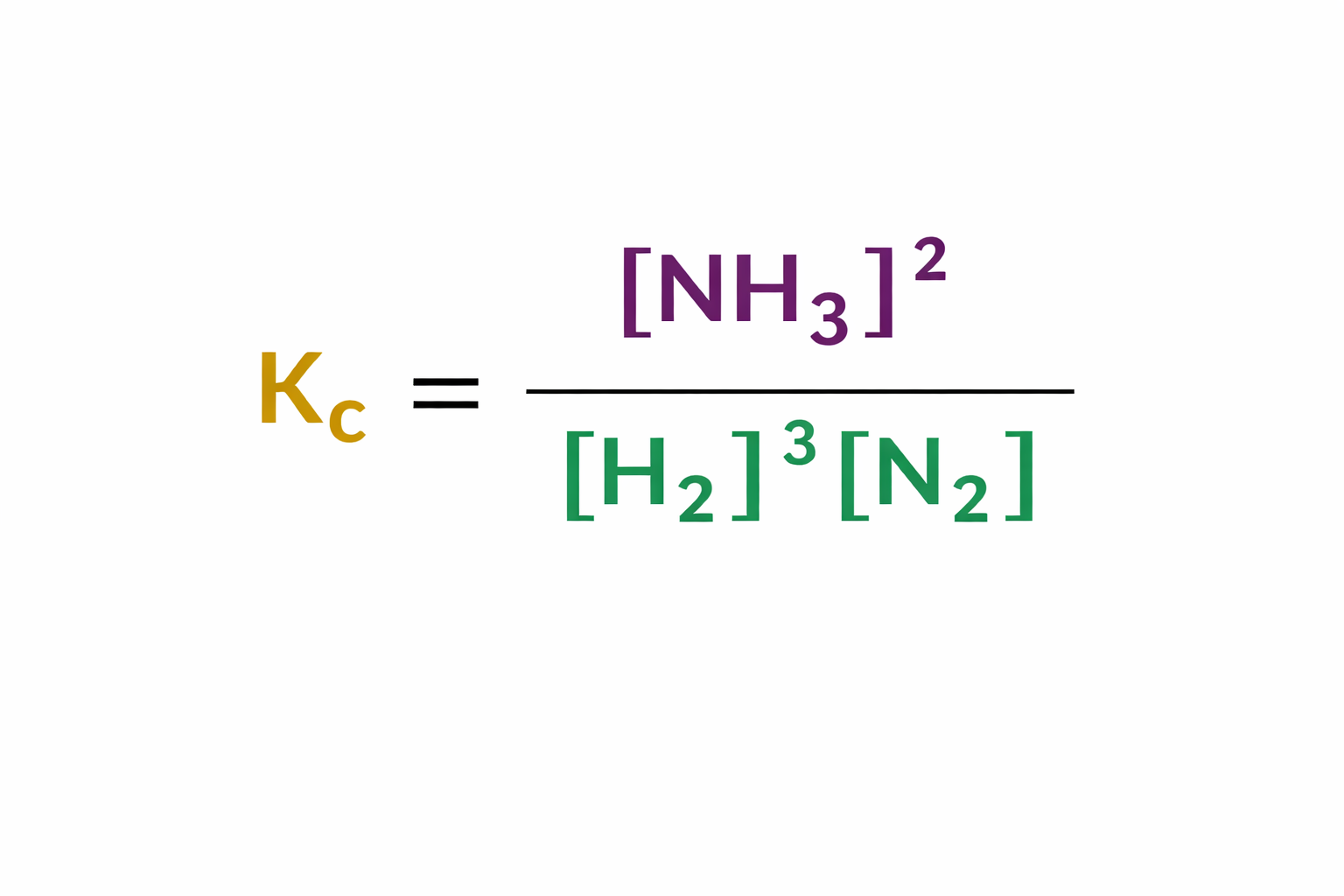
What are the units of Kc for this expression?
mol-2dm6
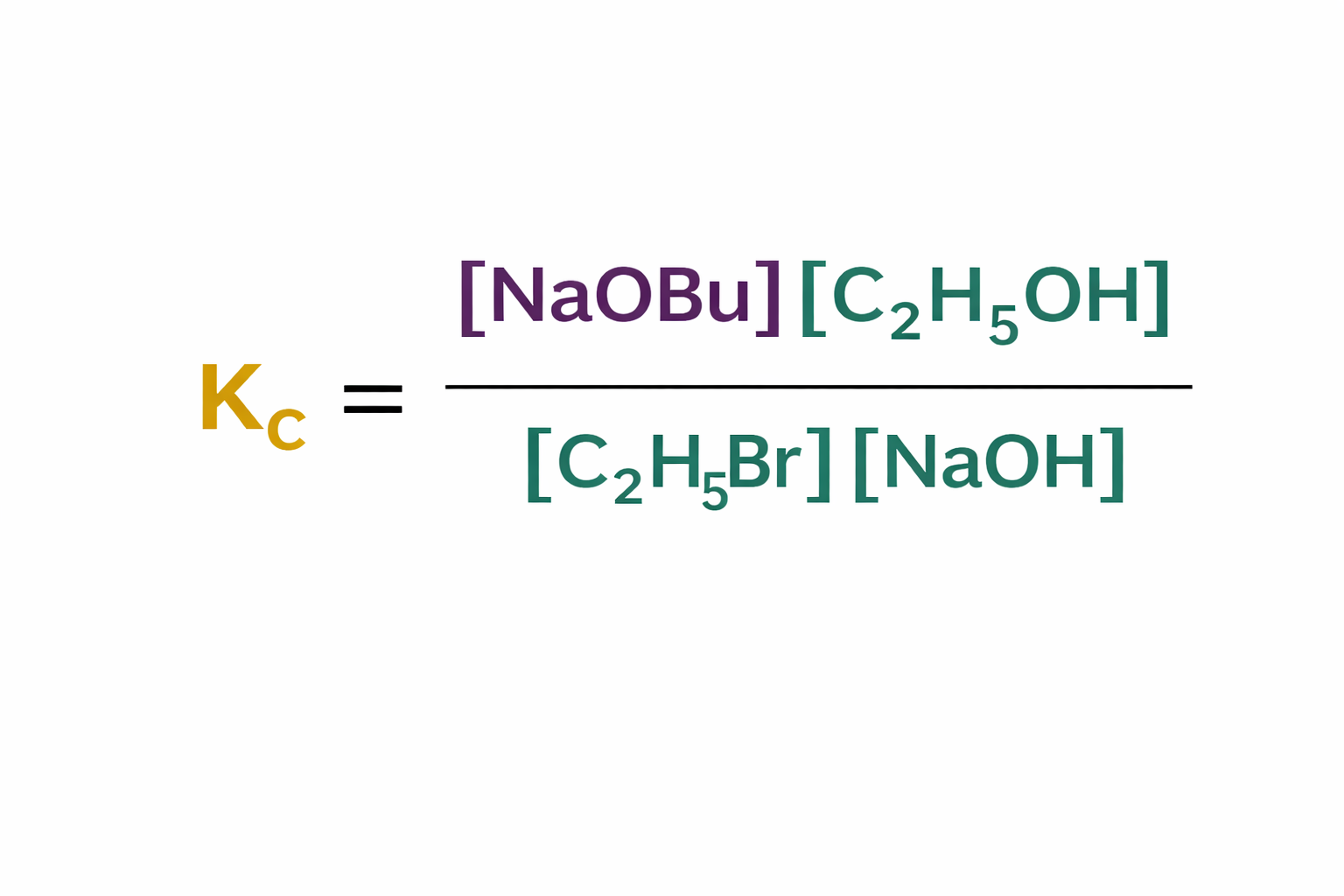
what are the units of Kc for this expression?
no units
what are the 2 types of systems that dynamic equilibria can exist in?
homogenous system
heterogenous system
meaning of homogeneous equilibrium system
has all the substances involved in the equilibrium in the same phase e.g. gaseous reactions or solution reactions
meaning of heterogeneous equilibrium system
has the substances involve in the equilibrium reaction present in more than one phase
what would the Kc be in this homogenous equilibria?
CH3COOH(l) + C2H5OH(l) ⇌ CH3COOC2H5(l) + H2O(l)
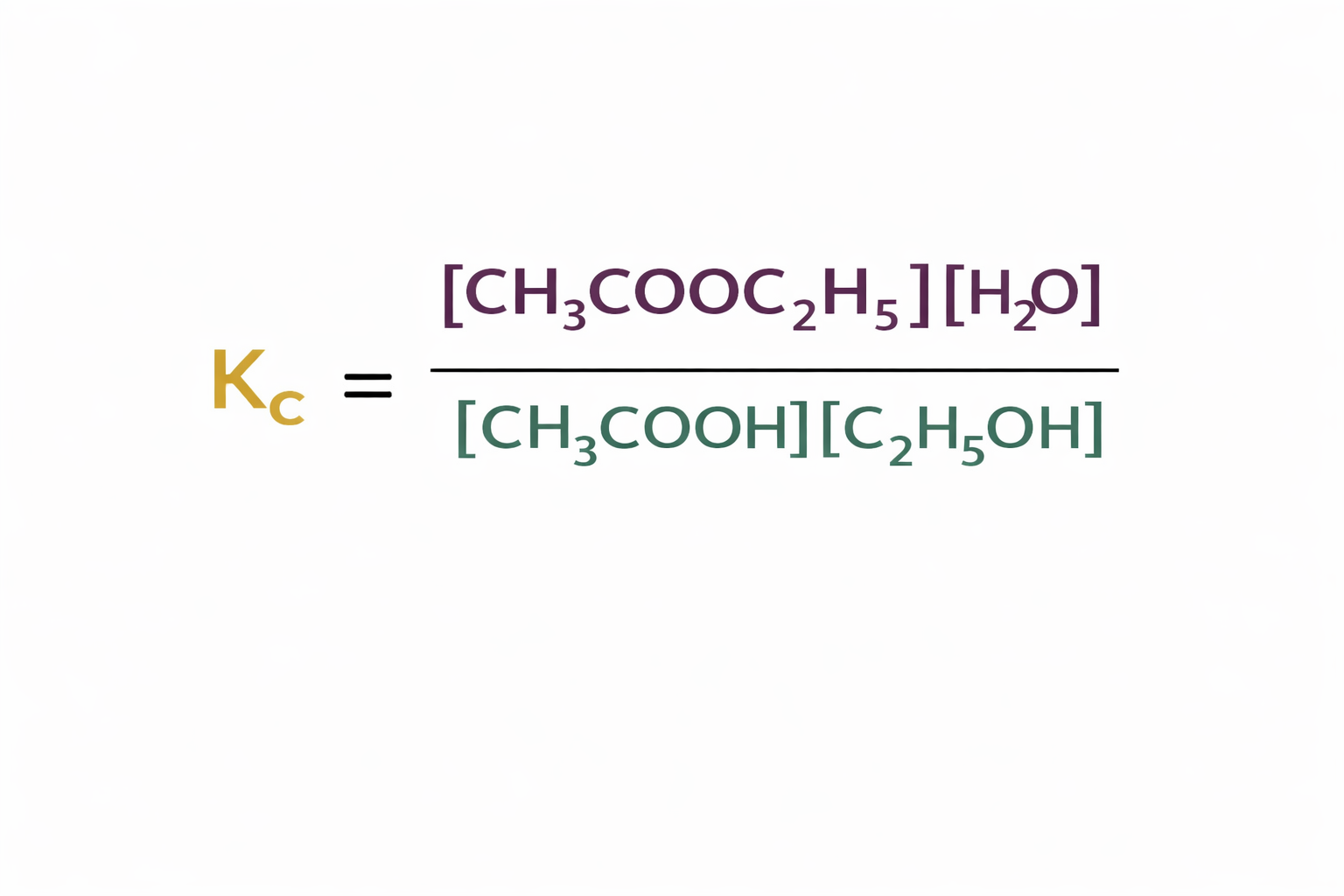
What would the Kc be in this hetereogenous equilibria?
H2O(g) + C(s) ⇌ H2(g) + CO(g)
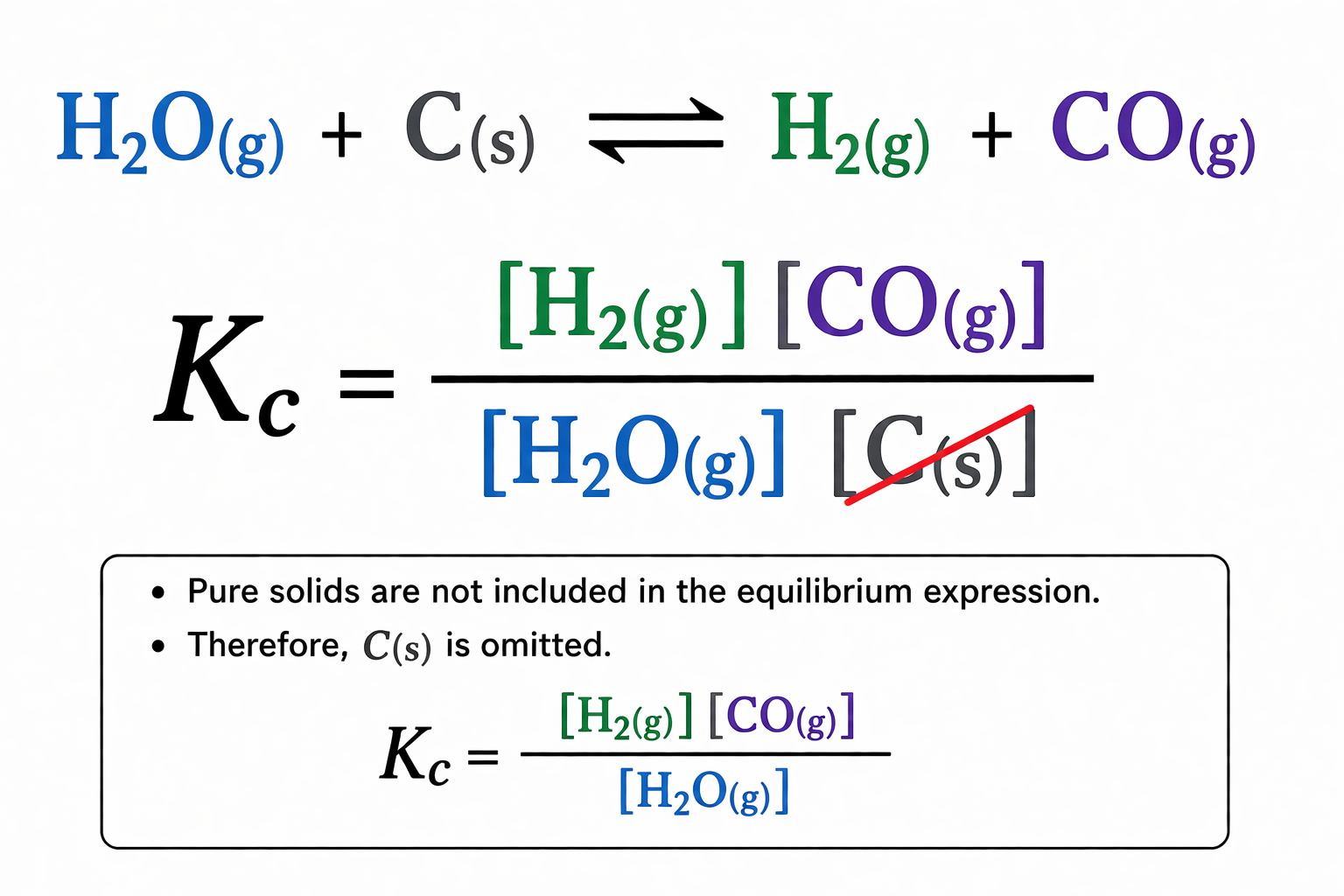
What is the total partial pressure?
the sum of all the partial pressure of the gases present
what is the partial pressure of any gas?
a fraction of the total pressure determined by its mole fraction
what is the expression for Kp?

what are the units of Kp?
kPa
Pa
atm
(same unit must be used for all gases)
If a question gives you the MOLES of all the reactants and products, how would you calculate Kc?
use moles to find concentrations → concentration = moles/volume
sub concentrations into Kc expression
If a question gives you the MOLES of all the reactants and products, how would you calculate Kp?
find total number of moles (add all moles together)
calculate mole fraction of each gas in mixture → moles of each component/total moles
calculate partial pressure of each gas in mixture → mole fraction of each component x total partial pressure
sub into Kp expression
what does the size of Kc or Kp indicate?
how far a reversible reaction has proceeded
does not tell us how fast it goes (nothing to do with rate)
If Kc or Kp = 1, what does this indicate?
equilibrium is half way between reactants and products
If Kc or Kp > 100, what does this indicate?
equilibrium lies well to RHS → favours products
If Kc or Kp < 0.01, what does this indicate?
equilibrium lies well to LHS → favours reactants
what will happen to Kc and Kp when temperature increases in exothermic reactions? (ΔH -ve)
Kc and Kp decrease
equilibrium shifts to LHS
decreases amount of products
increases amount of reactants
what will happen to Kc and Kp when temperature increases in endothermic reactions? (ΔH +ve)
Kc and Kp increase
equilibrium shifts to RHS
increases amount of products
decreases amount of reactants
Describe what happens to Kc when concentration changes?
Kc doesn’t change
initially the value of the expression for Kc changes as the system is no longer at equilibrium
position of equilibrium changes to convert reactants to products or vice versa
new equilibrium reached
Value of Kc restored
describe what happens to Kp when the pressure changes?
Kp does not change
initially the value of the expression for Kp changes as system is no longer at equilibrium
position of equilibrium moves changing the proportions of reactants and products
new equilibrium reached
value of Kp is restored
If a question gives you incomplete information about a mixture, how would you calculate the amount of each substance?
RICE method
what does the RICE method stand for?
R - ratio (big numbers from the equation)
I - initial (initial numbers of moles, if not given, write 0)
C - changes in moles (always - for reactants, always + for products)
E - equilibrium (moles at equilibrium)