Organometallic reaction chemistry
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What are the two types of external reagents?
Nucleophile: attacks complexes which are cation or bear EWGs.
Electrophiles: attacks complexes with are anions in low O.X. or EDG.
What are the 3 types of reactions?
Addition: attacking species stays attached to complex in product.
Abstraction: attacking species lost in product
Substitution: attacking species undergoes addition, something else is lost.
Define umpolung
Activating a bond/species to the reverse reactivity through metal coordination. E.g. activating C=C to nucleophilic attack instead of electrophilic.
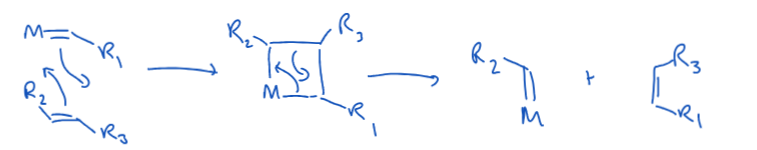
State the 3 DGM rules.
Nu attack preferentially at even polyenes over odd polyenyls
Nu attack to open polyenes over closed.
For open, even polyenes, attack is always at the terminal carbon. For open, odd polyenyls, attack occurs at the terminal carbon only if MLn+ is strongly electron withdrawing.
Explain the rationale behind the DGM rules.
The carbon atom which is attacked by the nucleophile is the one which bears the largest partial positive charge.
Nucleophiles will attack at the atom with the largest contribution to the HOMO (since electron density is donated to the metal).
Even polyenes have two electrons in their HOMO, so all electron density in the bonding MO comes from the polyene. Whereas some electron density must come from the metal in the odd polyenyls. Hence, even polyene donate two electrons from their HOMO to the metal, odd only donate one electron. T.f. more positive charge on even ligands – even before odd.
The symmetry of closed polyenes means that charge is more delocalised than in open polyenes, open before closed.
When can the DGM rules be used?
Relate only to kinetically controlled nucleophilic attack at 18 VE cations containing unsaturated hydrocarbon ligands.
When DGM rules apply, which face does the nucleophile attack?
Exo
What is the exception to the rule for cyclobutadiene
Cyclopentadiene is an even polyene but with unpaired electrons in its HOMO → not counted as a polyene. Hence even polyenes are attacked before cyclobutadiene, but cyclobutadiene is attacked before odd polyenyls.
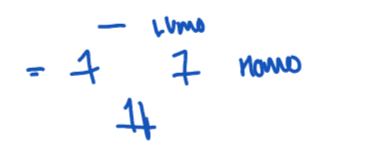
State when there is an exception to the DGM rules
For any reactions that proceed under thermodynamic control, i.e. due to steric hinderances.
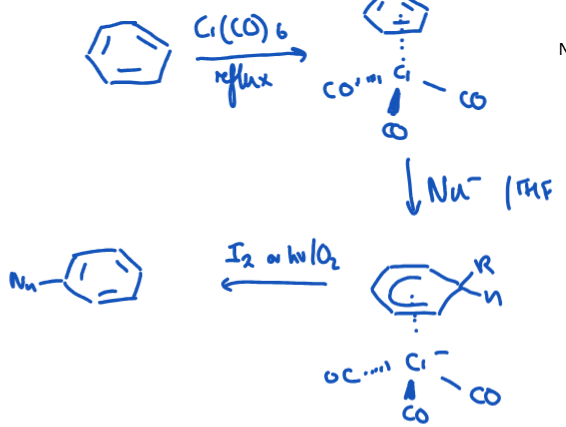
How can Cr(CO)3 be used for an arene to undergo nucleophilic substitution more easily.
3 x CO → strongly withdrawing. Complexed arene becomes activated towards nucleophiles. Gives chlorobenzene similar reactivity to e.g. chloronitrobenzene but much easier to remove the metal (using I2 or hv/O2)than NO2.
What effect do metals have an the reactivity of CO when complexed? Give a reaction example
The metal polarises the carbonyl, making the carbon atom more partially positive and prone to Nu attack.
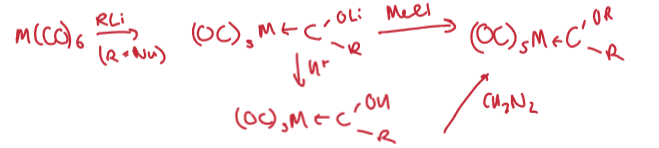
What type of reactions occurs at a Fischer carbene? (2)
Electrophilic carbon so undergoes nucleophilic substitution of a LG attached to the carbon.
The partial positive charge on the alkylidene (carbene with alkyl substituents) also makes the hydrogens on the adjacent carbon more acidic so easily deprotonates.
For polyenes/polyenyls containing CO, what defines whether a nucleophile attacks the CO or the C=C bond.
For charged nucleophile → Kinetic product. Attacks the polyene/polyenyl as predicted in DGM rules.
For neutral nucleophiles → Thermodynamic product, Attacks at the CO.
What nucleophiles can be used for the reaction of a cationic metal carbonyl and a neutral metal carbonyl to undergo loss of CO2 by nucleophilic abstraction. Why?
Cationic: can use water, since it is so prone to Nu attack.
Neutral: requires a more powerful Nu e.g. R3N+O-.
Cationic complexes are electron poor with less pi back bonding to CO ligands → high electrophilicity.

Outline protonation at a metal site. Give H+ sources, and explain how we can determine if protonation has occurred at the metal site.
Presence of a lone pair on a metal → electrophilic attack by H+. H+ sources include HBF4, H2SO4, acetic acid. 1H NMR can be used to determine protonation, as metal hydrides have low, negative shifts.
Explain why chlorosulfonic acid is used over sulfuric acid for the sulfonation reaction of organometallics.
ClSO3H allows for clean sulfonation and prevents oxidation:
What is the effect on electron count of a diene fragment reacting with electrophiles - how is the accounted for?
18VE → 16VE.
Achieves an 18VE count by:
Coordination of the counter anion (requires a coordinating anion).
Coordination of an added ligand (requires a non-coordinating anion e.g. BF4-).
Donation of the electron density in a C-H bond of the CH3 group to the electron deficient metal, forming an agostic bond (requires a non-coordinating anion e.g. BF4-).
What is formed from the alpha proton abstraction reaction of a metal akyl? What is required for this to occur?
Forms Schrock alkylidenes (carbenes).
Requires no beta C-H bonds (otherwise beta-elimination occurs)
Requires a base e.g. internal base (another alkyl group) or an external base (lewis base e.g. Ph3P=CH2)
What is required ffor H+ abstraction of polyene fragments?
Requires a strong base e.g. BuLi. Requires it to be complexed to a strong EW metal complex (e.g. Cr(CO)3) to enhance acidity of H for abstraction.
What is the use of TMEDA in the reaction of BuLi and ferrocene.
Increases basicity → double lithiation
What reagent is used for an electrophilic H- abstraction?
Uses Ph3C+. formed by Ph3C-OH + HX → Ph3C+X-. Removes H- from alkyl → alkene.(Note: can get endo or exo attack)
How can H+ be used in electrophilic abstraction?
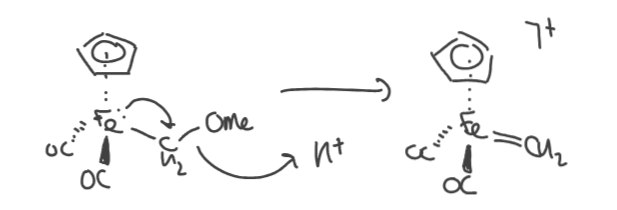
What is the difference between a Schrock carbene and Fischer carbene?
Fischer: late TM in a low O.S. with low lying filled d-orbitals. Has a heteroatom substituent attached to carbon. L type ligands.
Schrock: early TM in high O.S. with high energy empty d orbitals. alkyl groups attached. X2 type ligand.
Outline the 4 synthetic routes to an alkylidene (carbene)
Metal carbonyl + RLi. Followed by electrophilic attack at the O (OLi → OR).
Trapping of a free carbene (CH2N2) by a coordinatively unsaturated metal centre/metal centre with a readily lost ligand (PPh3)
Alpha deprotonation of an alkyl group.
Electrophilic H- abstraction using Ph3C+
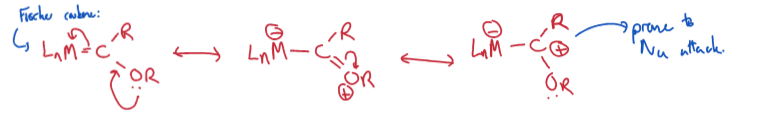
Outline the VB description of Fischer carbenes
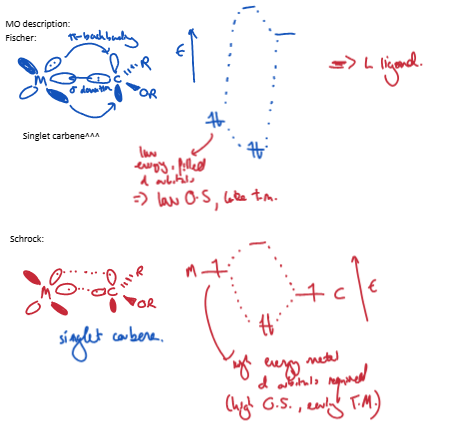
Outline the MO description for Fischer carbenes and Schrock carbenes
How do the reactivities of Fischer and Schrock carbenes differ?
Fischer: act as electrophiles due to partial positive charge. Reactivity like an ester given R = OR.
Schrock: act as nucleophiles due to partial negative charge. Similar behaviour to that of a phosphorous ylid.
Give the general mechanism for alkene metathesis reactions.