Unit 1 AP Bio
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Properties of H2O
Polarity makes it the universal solvent
Hydrogen bonding
Less dense as solid
High specific heat
High heat of vaporization
Polarity
Polarity comes from the covalent bond between O and H’s within a single H2O molecule
A single H2O molecule has “unequal charges”
H = positive
O = negative
Polarity contributes to H bonds forming between
different water molecules
Universal solvent
Polarity makes H2O the US —> it dissolves many substances
Polar substances are
Hydrophilic
Hydrophilic
charged (+ end & - end)
attracted to H2O
dissolved by H2O
examples: sugar, salts
Nonpolar substances are
Hydrophobic
Hydrophobic
uncharged
repelled by H2O
not dissolved by H2O
examples: lipids
Hydrogen bonding
H bonds are attractions between polar molecules
H bonding between H2O molecules is
cohesion
creates surface tension (water is sticky)
H bonding between H2O & other substances is
adhesion
meniscus in cylinder
up a tree
raindrops on glass
H2O is less dense as a
solid (most are more dense when solid)
H bonds form a crystal
Ice floats —> lakes don’t freeze solid so living things can survive below ice
Water’s specific heat is
high
H2O resists changes in temp
takes a lot of heat to raise temp & cooling to lower temp
H2O moderates temps on Earth
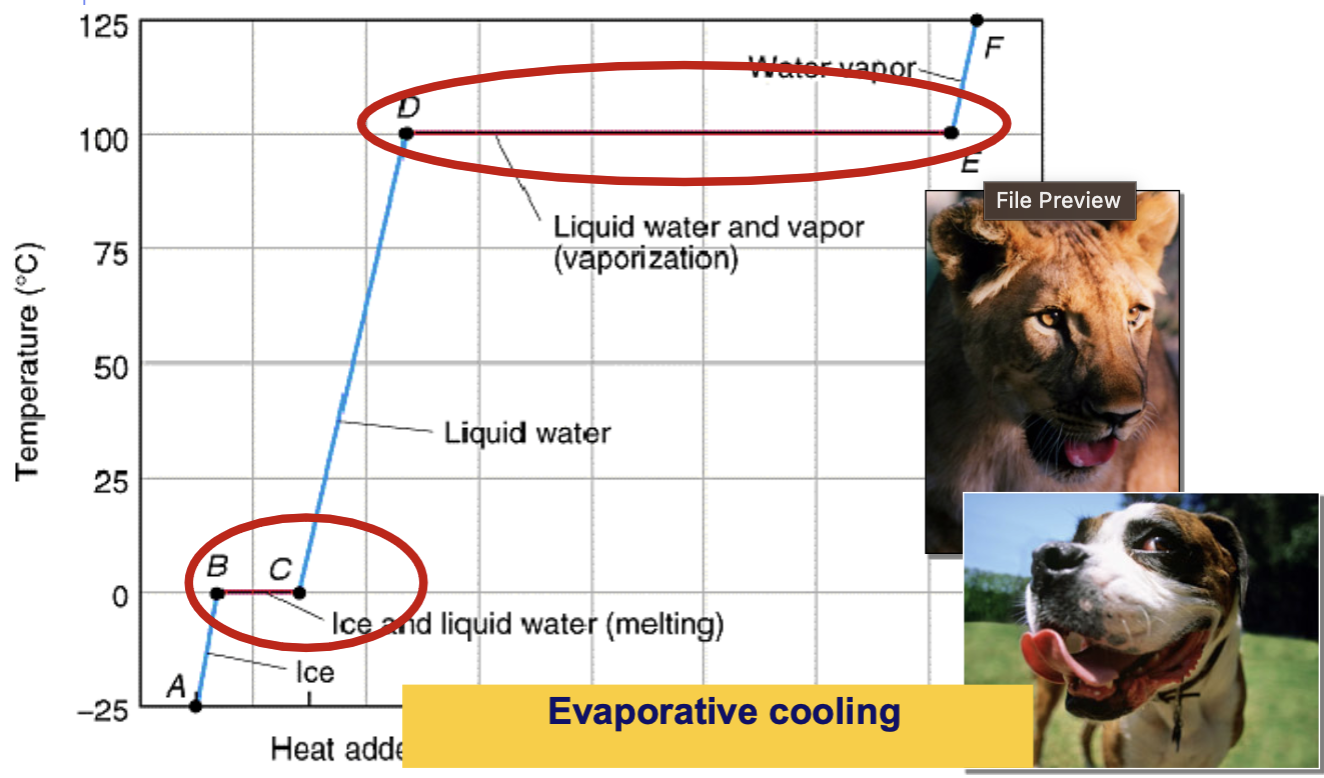
Water requires ___ energy/heat to turn water from solid to gas
high
Atoms & molecules from environment are used by living things to build
biomolecules (biomolecules)
4 biomolecules are essential to all living things
carbohydrates
lipids
proteins
nucleic acids
Elements found in ALL biomolecules
carbon
hydrogen
oxygen
others:
Phosphorus - needed to build nucleic acids & phospholipids
Sulfur - needed to build proteins
Nitrogen - needed to build proteins & nucleic acids
Phosphorus need to build
nucleic acids & phospholipids