Alkenes
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Describe the structure of alkenes
Unsaturated hydrocarbons
Contains a C=C bond comprising a Pi-bond and a sigma bond
Restricted rotation of the Pi bond
What is a Pi bond?
Sideways overlap of adjacent p-orbitals above and below the bonding C atoms
Explain the trigonal planar shape of an alkene C=C bond
3 bonding regions & 0 lone pairs
Electron pairs repel as far as possible
Creating trigonal planar shape with bond angle 120
Define the term “stereoisomers”
Same structural formula, different arrangement of atoms in space
Define the term “E/Z isomerism”
An example of stereoisomerism. Molecules must have
A C=C bond
Different groups must be attached to each carbon of the double bond
Define the term “cis-trans isomerism”
A special case of E/Z isomerism, where 2 of the groups attached to each carbon atom of the C=C group are the same
Cahn-Ingold-Prelog rules
If higher priority groups are on the same side = Z isomer
If higher priority groups are diagonally places across the double bond = E isomer
Explain why alkenes are more reactive than alkanes
Alkenes have Pi-bond present
Pi-bond has a low bond enthalpy, so it’s broken more readily
Less energy is needed to overcome the bond
Hydrogenation of alkenes
Alkene + Hydrogen gas —> alkane
Nickel catalyst at 150C
Halogenation of alkenes
Alkene + halogen —> Haloalkane
Test for C=C double bond
Bromine water decolourises in presence of C=C double bond
From orange to colourless
Alkene reaction with Hydrogen Halides
Alkene + Gaseous Hydrogen Halides —> Haloalkanes
If alkene is gas - 2 gases are mixed
If alkene is liquid - hydrogen halide is bubbled through
Hydration of alkenes
Alkene + steam —> alcohol
Phosphoric acid catalyst >100C
Define the term “electrophile”
An electron pair acceptor
Type of mechanism between alkenes and hydrogen halide to form haloalkane
Electrophilic addition
Mechanism for but-2-ene & HBr to form 2-bromobutane
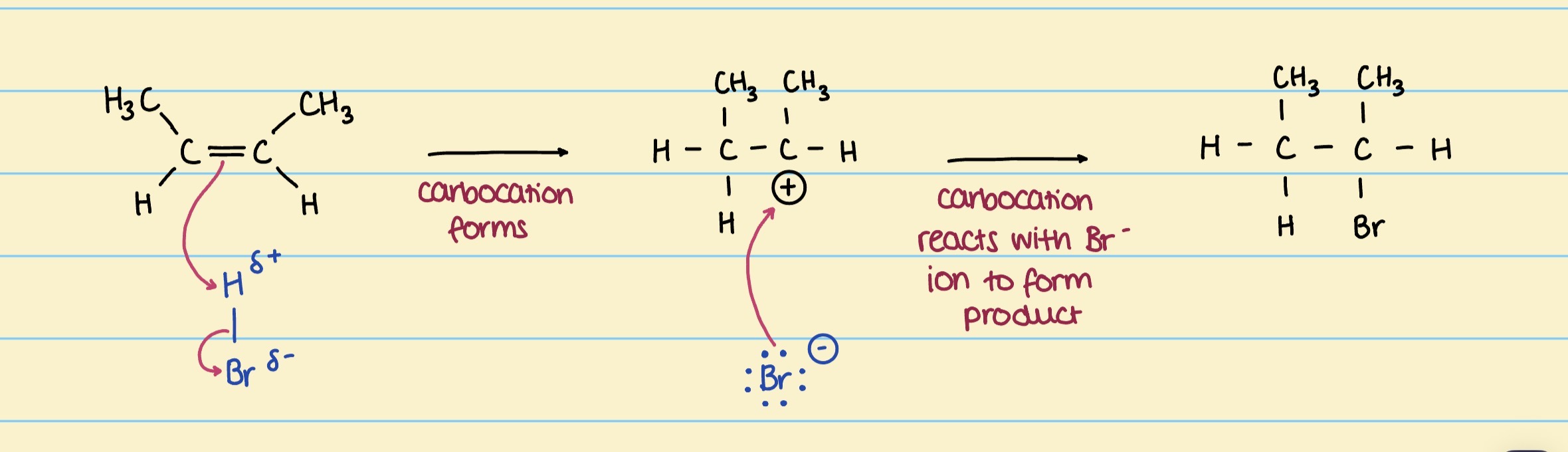
Using Markowinkoff’s rule to predict the major & minor product of electrophilic addition
Tertiary carbocations are more stable than secondary carbocations, which are more stable than primary carbocations.
The more stable product = major product
The less stable product = minor product
Describe the benefits of processing waste polymers by combustion for energy production
Can be incinerated to produce heat, generating steam to drive a turbine producing electricity - renewable energy source
Describe the benefits of using waste polymers as organic feedstock for production of plastics & other organic chemicals
Able to handle unsorted & unwashed polymers
Products of feedstock recycling can be used as raw materials for production of new polymers
Describe the benefits of using waste polymers to remove toxic waste products during the disposal by combustion of PVC
Combustion of PVC produces HCl, a corrosive gas
Solvents are used to dissolve the polymer.
High grade PVC is recovered by precipitation from the solvent
Solvent is used again
Benefits of biodegradable polymers on sustainability
Polymers made of starch or cellulose, which are broken down by microorganisms
They degrade and leave no visible or toxic residue
Benefits of photodegradable polymers on sustainability
Oil-based polymers
Contain bonds which weaken when absorbing light to start degradation