Organic Chemistry Ch 18
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
Electrophilic Aromatic Substitution (EAS)
reaction where an aromatic proton is replaced by a strong electrophile or a functional group is introduced to the ring. the ring acts as a nucleophile.
EAS mechanism

bromination of benzene reagents
Br2
AlBr3
chlorination of benzene reagents
Cl2
AlCl3
Nitration of benzene reagents
HNO3
H2SO4
sulfonation of benzene reagents
fuming H2SO4
adding methyl group to benzene reagents
CH3Cl
AlCl3
Sulfonation mechanism
last part has SO3H on it
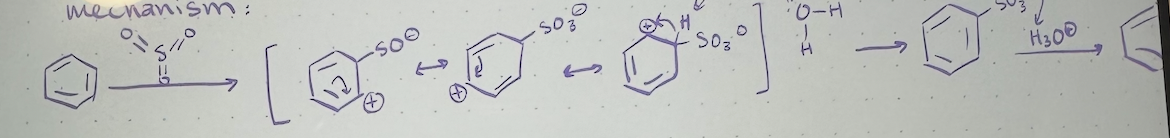
Nitration mechanism

reagents to reduce nitro group to amine (NO2 to NH2)
1) Fe, HCl
2) NaOH
Friedel-Crafts alkylation mechanism
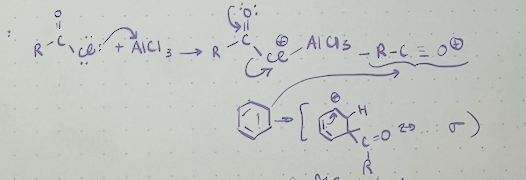
3 limitations of friedel crafts alkylation
1) halide leaving group must be attached to an sp3 C (or no rxn)
2) polyalkylation often occurs
3) some substituted aromatic rings are too deactivated to react
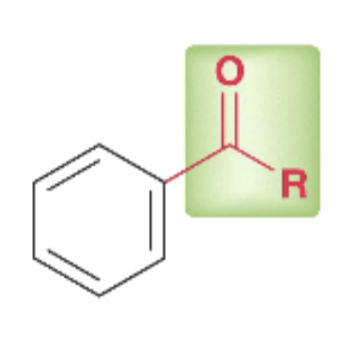
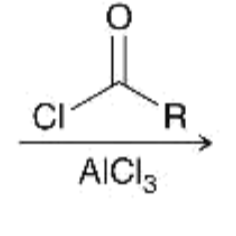
reagents for acylation of benzene ring
most common activating groups
methyl group, methoxy group
usually ortho-para directors
activators
major product of ortho-para directors (usually)
para
why is methyl group an activator
CH3 donates electron to the ring so it acts as a better nucleophile and is more reactive
why is methoxy group an activator
it donates electron density to a ring via resonance (even though it looks like it might remove electron density via induction)
which wins if resonance and induction compete
resonance
most common deactivating group
nitro group, halogens
usually meta directors
deactivating groups
why is nitro group deactivating
inductively electron withdrawing and removes electron density via resonance
why are halogens considered exceptions for deactivating groups
they are ortho-para directors
why are halogens ortho-para directors
they withdraw electrons via induction but donate electron density through resonance
strong activators
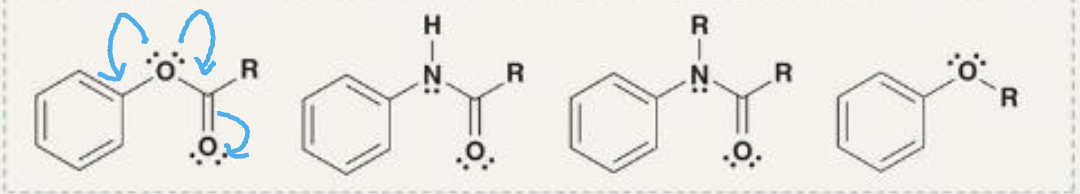
lone pair adjacent to aromatic ring

moderate activators
lone pair delocalized outside ring
weak activator
alkyl (hyperconjugation)

weak deactivator
halogens (exception)

moderate deactivators
carbonyls and analogs
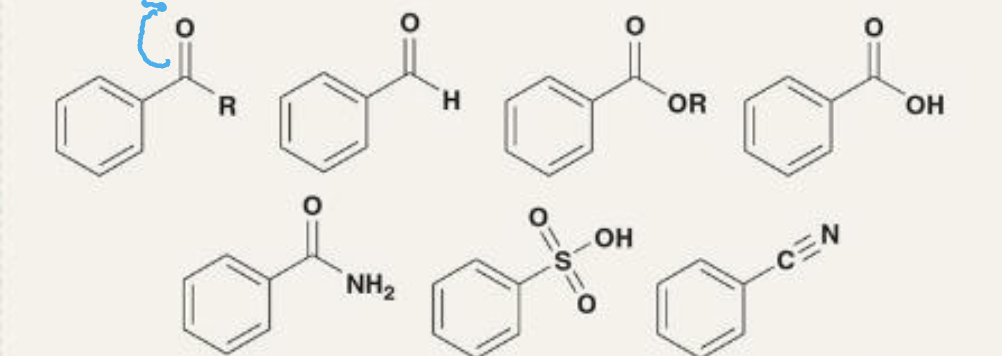
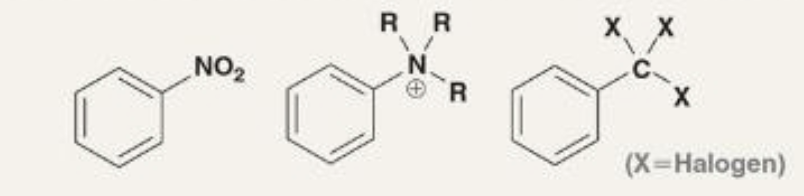
strong deactivators
positively charged or -CX3
where does addition go on a disubstituted ring where both substituents direct to the same carbon
to the carbon that it is directed to by both substituents
where does addition go if there are multiple substituents directing it to different carbons
where the stronger activator is directing it
where does addition go if there are multiple substituents directing it to different carbons on 1,4 disubstituted rings
less sterically hindered site
where does addition go if there are multiple substituents directing it to different carbons on 1,3 disubstituted rings
not at site located between the 2 substituents
how to favor ortho over para from ortho-para directors
use a blocking group to block para position
common blocking group
sulfonation
Nucleophilic aromatic substitution abbreviation
SnAR
what occurs in nucleophilic aromatic substitution
a substituted benzene ring is attacked by a nucleophile
SnAR mechanism
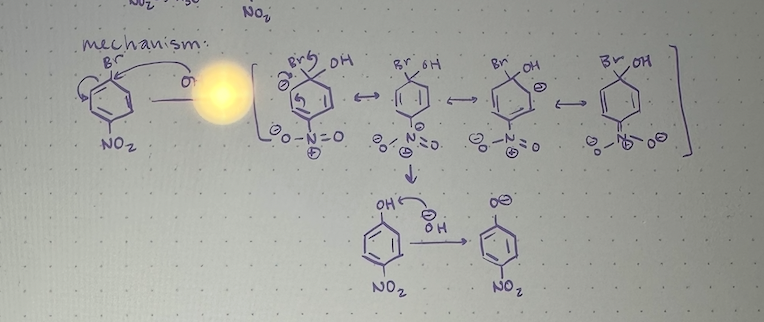
3 requirements for SnAR
1) benzene ring must posses strong electron withdrawing grou
2) ring must possess good leaving group
3) leaving group must be ortho or para to electron leaving group (can’t be meta)
what is good & best leaving groups for SnAR
best is -F, halides in general are good
what is typically ewg in SnAR
-NO2
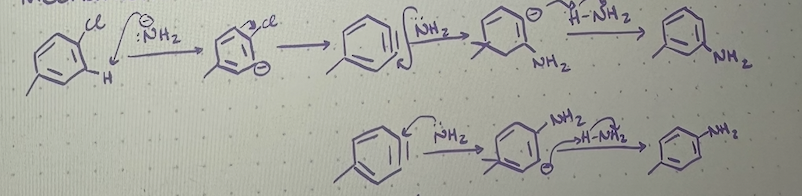
elimination-addition reaction
2 step SnAR reaction, first leaving group is eliminated by strong base which leaves a benzyne intermediate, then a nucleophile is added
benzyne intermediate
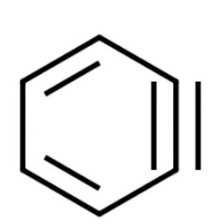
elimination-addition mechanism
Clemmenson reduction reagents
Zn(Hg)
Hcl, heat
Clemmenson reduction what it looks like

Alkyl group
acyl group