Unit 2 Biology
1/94
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
Metabolism
A set of linked reactions that degrade fuel molecules and construct biomolecules
What do cells use ATP for?
1) Synthesis of macromolecules
2) Signal transduction and active transport of molecules of ion
3) Mechanical work or muscle movement
Phototrophs + Examples
Obtain energy from sunlight. Use photosynthesis. Examples include plants and cyanobacteria.
Chemotrophs + Examples
Obtain energy from oxidation of carbon fuels. Fuel sources come from sugars (glucose and glycogen,) lipids (fatty acids stored at triglycerides,) and proteins (during starvation). Examples include plants, animals, and bacteria.
Catabolic Pathway
Breakdown of carbon “fuels” to synthesize ATP. Exergonic pathways with energy as a product. EX: Glycolysis
Anabolic Pathway
Uses ATP to synthesize larger biomolecules. Endergonic pathways that require energy. Energy is captured in chemical bonds. EX: Photosynthesis, protein synthesis
Catabolism and Anabolism
The energy harvested as ATP during the break down of molecules in catabolic pathways can be used to synthesize molecules in anabolic pathways
ATP Structure
Base (adenine), ribose (5-carbon sugar), 3 phosphates (2 phosphoanhydride bonds), and one gamma phosphate is one of the end.
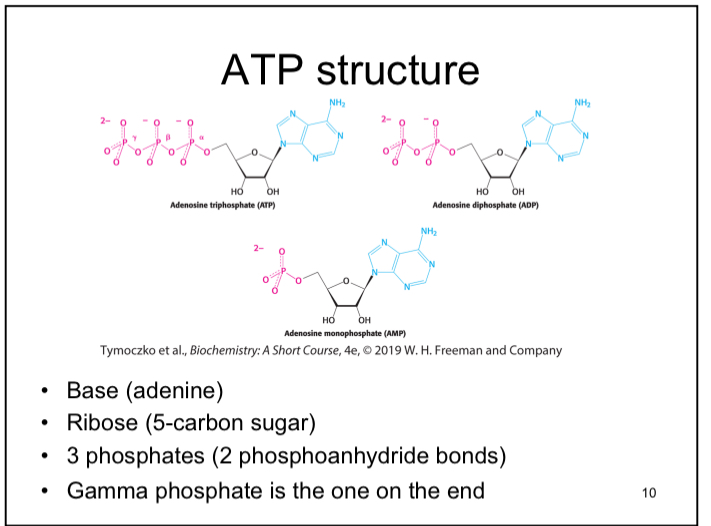
Phosphoanhydride bonds
High energy bonds in ATP that store chemical energy (potential energy).
Energetic Coupling of Reactions
The exergonic nature of ATP hydrolysis coupled to an endergonic reaction can result in an overall exergonic reaction. EX: Transfer of the gamma phosphate to a substrate
Step 1 of Glycolysis
Exergonic reaction of ATP hydrolysis can be used to create a high energy molecule used in glycolysis
Energy Charge of the Cell Used to Regulate Pathways
Catabolic pathways activated when the energy charge is low in the cell. Anabolic pathways activated when energy charge is high in the cell.
Energy Charge
Reflects the energy status of the cell
High Energy Molecules
Include PEP, (1,3- BPG), and creatinine phosphate
Creatine Phosphate
Creatine phosphate is used by muscle cells. It is even more exergonic than ATP and can be used to drive generate ATP is muscle cells. Creatine Phosphate + ADP —> ATP +Creatine
Glucose Oxidation Reaction
C6H12O6 + 6O2 —> 6H2O + 6CO2 + energy. Similar to a combustion reaction. Used to generate ATP
Oxidation and Reduction
Oxidation and reduction go together. Oxidation is the loss of electrons, reduction is the gain of electrons.
Oxidation
Carbon has more bonds to oxygen. Often associated with losing a hydride (H+ + 2e-) or with gaining a bond to oxygen
Reduction
Carbon has more bonds to hydrogen. Often associated with gaining a hydride (H+ + 2e-) or with gaining a bond to hydrogen
Glycolysis Overview
10 enzymatic steps converting 1 molecule of glucose to 2 pyruvate. First 5 steps are energy investing, second 5 steps are energy harvesting
Where does glycolysis occur?
Cytoplasm
What does glycolysis produce?
2 ATP, 2 pyruvate, 2 NADH, 2 water
First 5 Steps of Glycolysis
5 steps, net 2 ATP. Creates 2 molecules of GAPs. Step 3 commits to glycolysis via Phosphofructokinase (PFK).
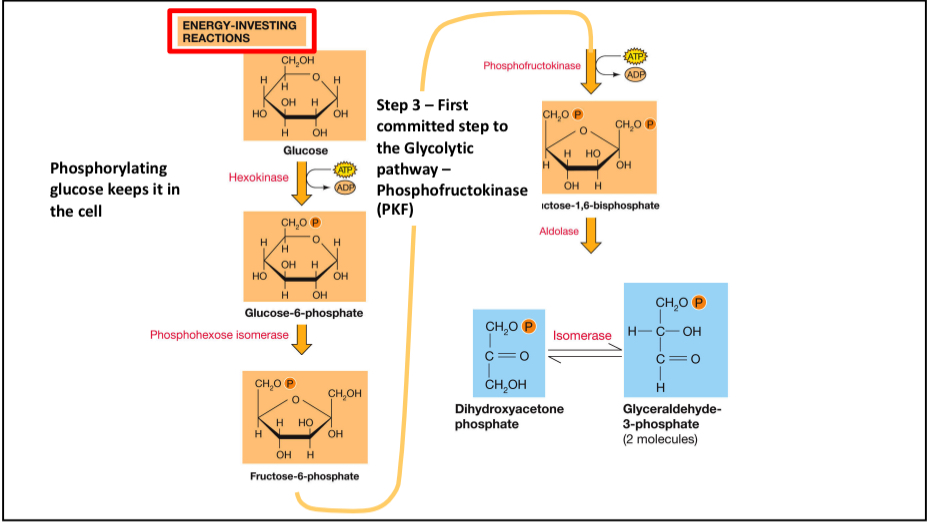
Step 1 Glycolysis
Hexokinase catalyzes the reaction between ATP and glucose to make glucose-6-phosphate
Hexokinase
Hexokinase closes once both substrates are bound
Closes out water molecules from the aqueous cytoplasm
Brings substrates in close proximity to react
Mg²+ in the active site stabilizes ATP
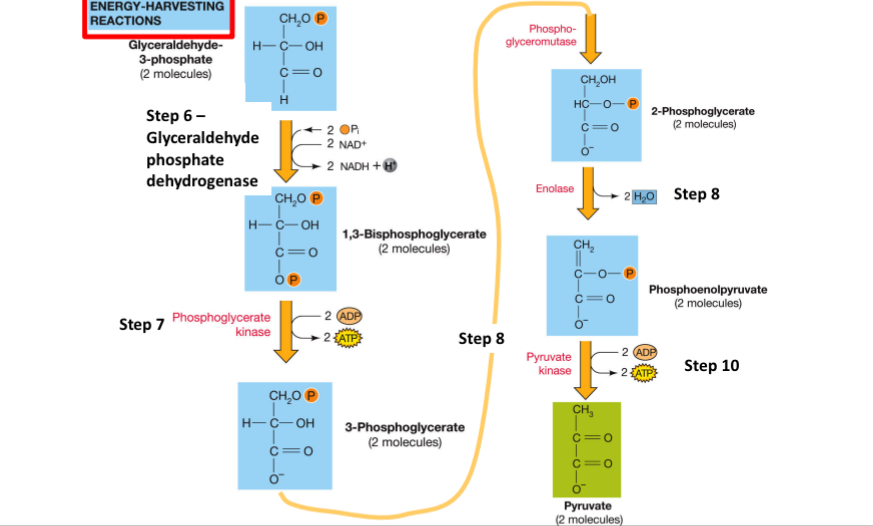
Steps 5-10 (Energy Harvesting Reactions)
ATP is produced at 2 different steps (7 and 10). NADH is produced (step 6). Pyruvate (2) are produced
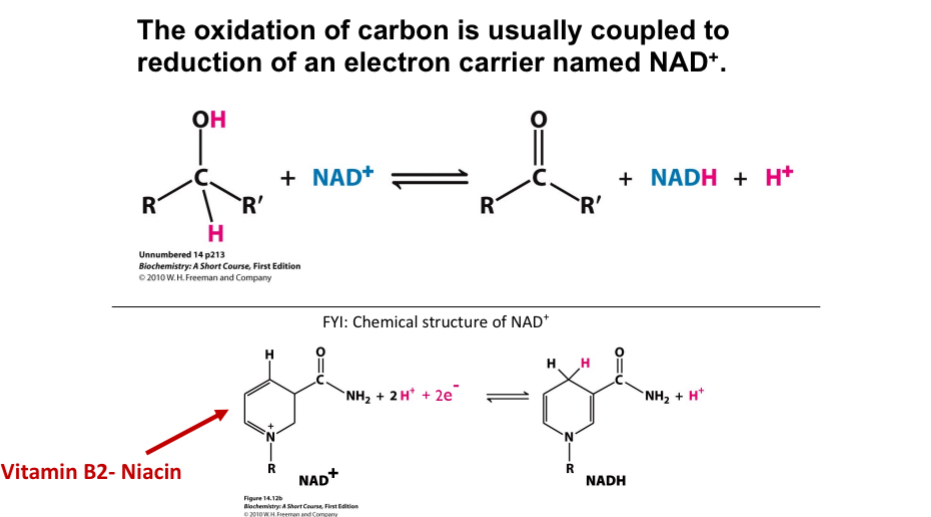
Carbon oxidation
Coupled with reduction of an electron carrier named NAD+
Regulation of Irreversible Steps
Phosphorylation, feedback inhibition and/or allosteric inhibitors, transcription regulation, enzyme degredation
Reversible Steps
Typically not regulated and determined by substrate v product concentrations
Phosphofructokinase
Irreversible reaction
Has two substrates: ATP and fructose 6 phosphate
ATP is both a substrate and allosteric regulator
Under low ATP concentrations, ATP only binds to the active site
Under high ATP concentrations, ATP binds to the allosteric site and prevents phosphorylation
Km in PFK
Low in active site, high in allosteric site
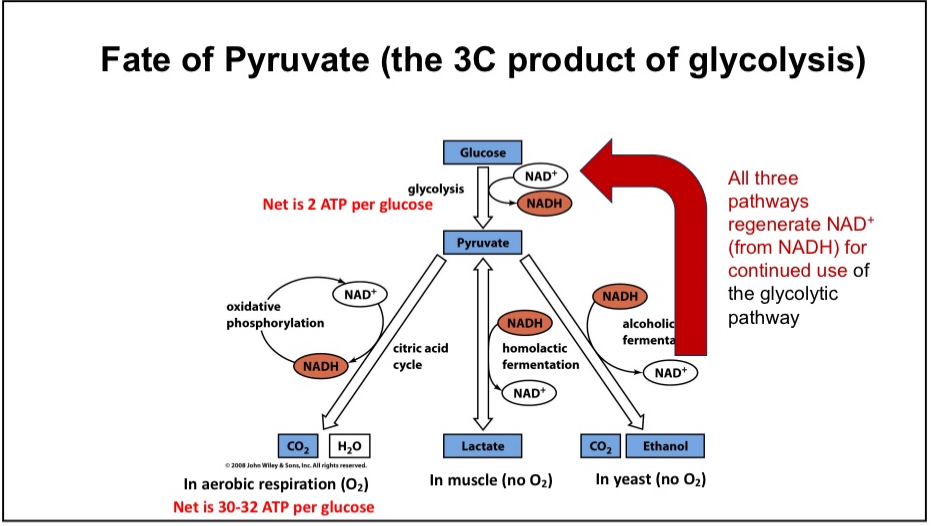
Fermentation Pathway
Active when no oxygen is present
Cellular purpose is to regenerate NAD+ so cells can continue to glycolysis
Mammalian fermentation pathways produce lactic acid
Fermentation pathways in yeast and bacteria generate useful products such as acetic acid, ethanol, acetone, yogurt and cheese`
Fate of Pyruvate
Aerobic Respiration or anaerobic respiration
Oxidation of Pyruvate
Occurs in the mitochondrial matrix
Creates 2 Acetyl CoA, 2 CO2, and 2 NADH
Catalyzed by pyruvate dehydrogenase
Acetyl-CoA
Acetyl-CoA is a central metabolite that is in common with fatty acid degradation, amino acid degradation, and sugar metabolism
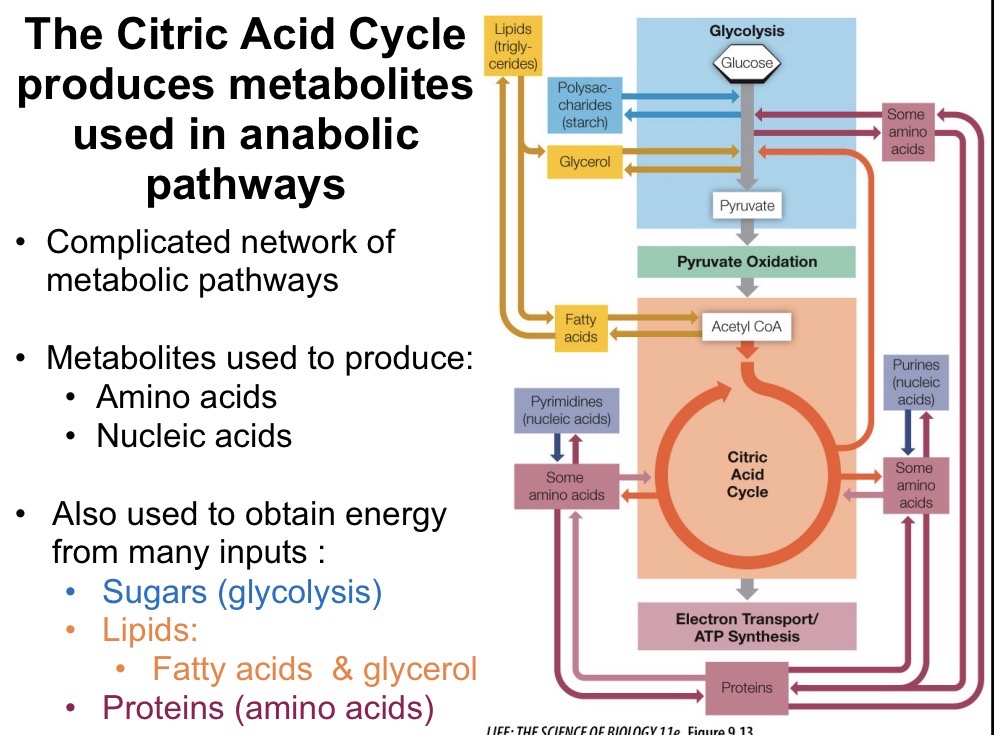
Mitochondria Structure
Outer membrane contains pores permeable to small molecules
Inner membrane is highly folded to form cristae, where ETC takes place
Matrix is the site of pyruvate oxidation and citric acid cycle
Intermembrane space low pH, high H+
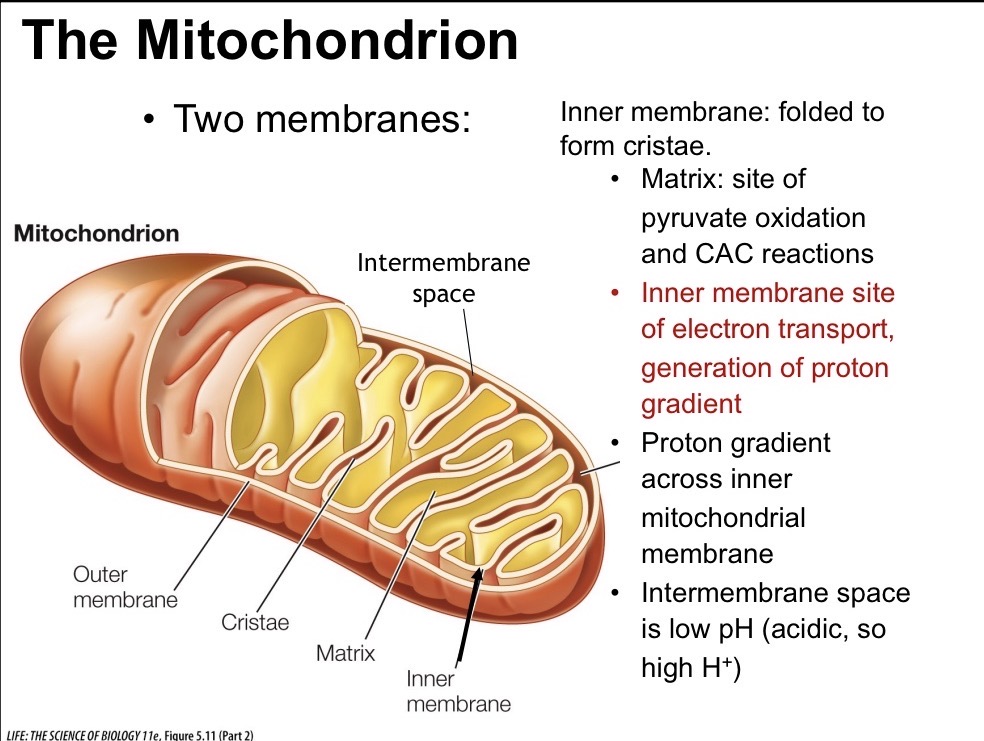
Last Step of CAC
Malate is oxidized to oxaloacetate
What are the products of the CAC?
4 CO2, 6 NADH, 2 FADH, 2 ATP
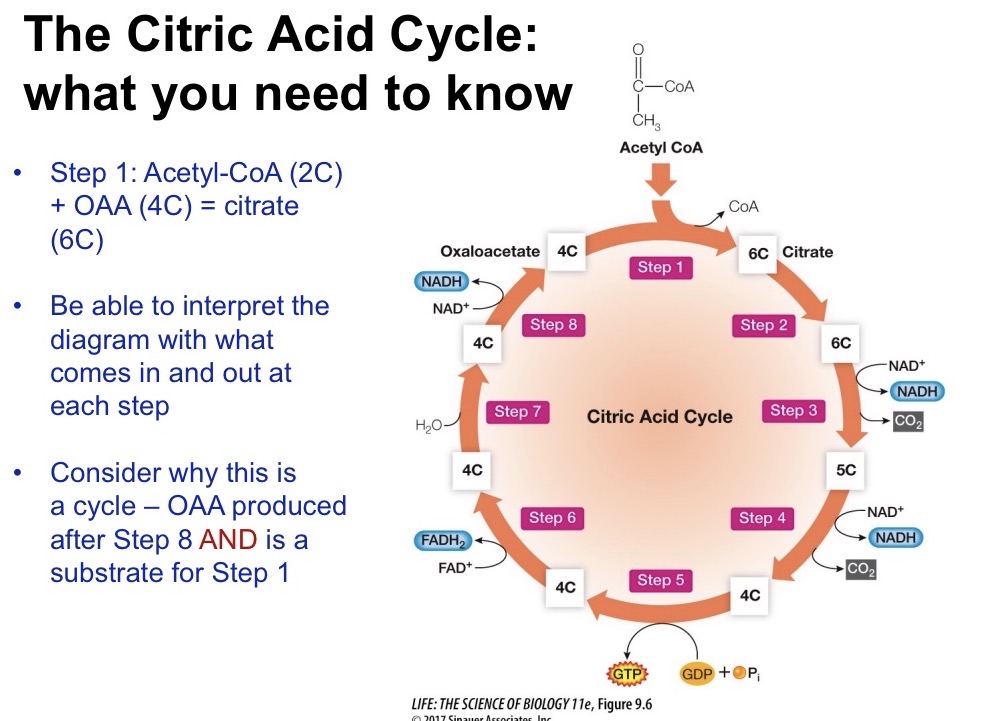
Diagram Glycolysis, Pyruvate Oxidation, CAC. What does each step generate and where does each step happen?
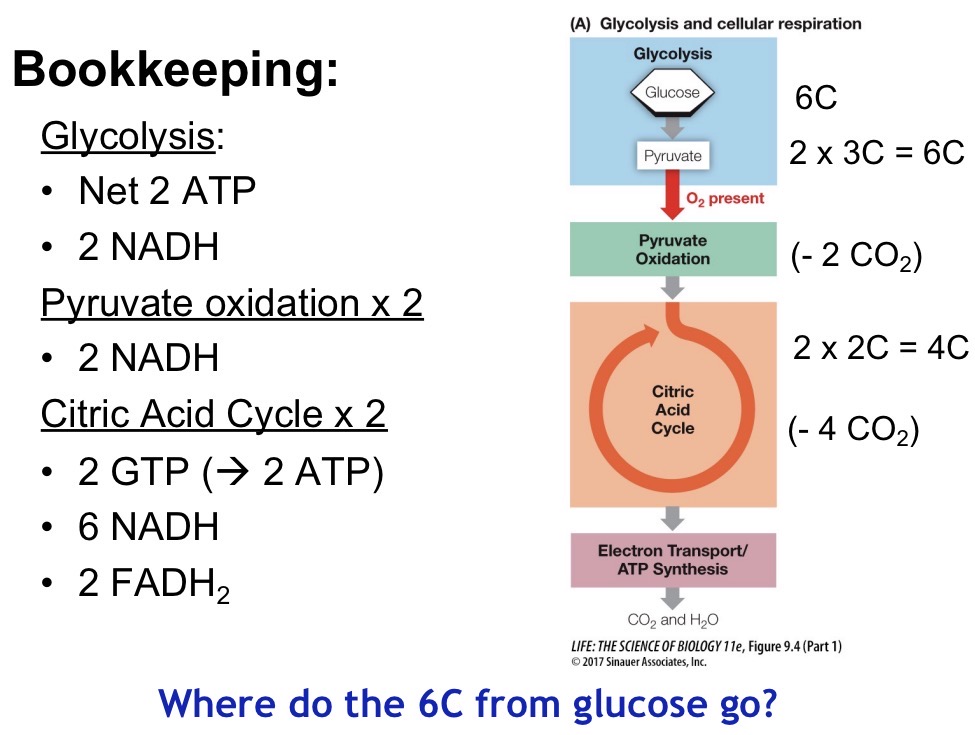
ETC
Flow of electrons from high to low free energy
Complex I: NADH donates electrons and pumps H+ into intermembrane space
Complex II: FADH donates electrons but none are pumped into the intermembrane space
Complex III: H+ are pumped into the intermembrane space
Complex IV: H+ is pumped into the intermembrane space, O2 is reduced to H2O
Complex V (ATP Synthase): Protons flow down gradient and synthesize ATP
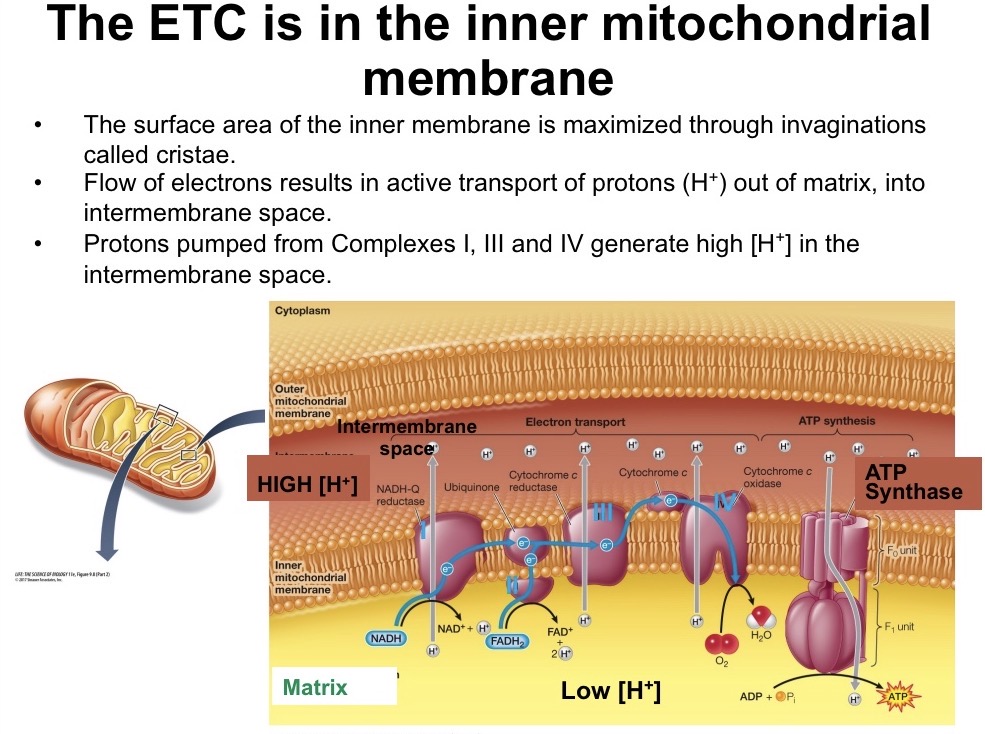
NADH vs FADH2
Both carry 2 electrons. However, NADH generates 2.5 ATP/e- pair while FADH2 generates 1.5 ATP/e- pair because NADH donates to the ETC at a higher energy level
Chemiosmotic Hypothesis
Proton gradient (from the intermembrane space to the matrix) provides the energy needed to make ATP
Adipose Tissue
Stores and releases fatty acids
Liver
Maintains constant level of blood glucose
Brain
Requires glucose and oxygen (ketone bondies during starvation)
Muscle
Stores glycogen, metabolizes FA
Insulin
Stimulates organs that can store glucose to take it out of the blood and store it as either glycogen or lipids
Glucagon and epinephrine
Reverse the effects of insulin and act to mobilize glucose and increase glucose in the blood
ATP Synthase Structure
Made of 8 subunit types
Subunits group to form two main component F0 and F1
F0
Proton Channel
1) Rotation of the c ring rotates the gamma subunit
2) The irregular shape of the gamma subunit forces conformational changes of the beta subunits: Open (O), Loose (L), and Tight (T0)
3) Chemical energy of proton gradient is converted to mechanical energy of rotating c ring, which is converted to chemical energy in ATP
F1
An enzyme wheel in the mitochondrial matrix that catalyzes ATP synthesis from ADP and P
Aspartic Acid
Proton enters subunit a from the intermembrane space and moves to a subunit c helix. Subunit c contains aspartic acid which holds the H+ from the proton gradient, becomes neutralized and can rotate
Beta Subunits
Result in ATP Synthesis
Open: ADP, Pi, and ATP free to diffuse in and out of B subunit
Loose: ADP+Pi trapped
Tight: ADP+Pi converted to ATP in yellow B subunit
ATP Synthase Stoichiometry
One 360 degree rotation of gamma results in 3 units
Photosynthesis Reactions
6CO2+12H2O+light energy —>C6H12O6+6H2O+6O2, involve light independent and light dependent reactions
Where does photosynthesis take place?
Mesophyll cells with chloroplasts
Where do the light dependent reaction occur?
Thylakoid membrane
Absorption Spectra for Chlorophyll
A and b
Photosystem
Absorbs a photon, excites an electron (oxidized) which gets accepted by an electron acceptor
Thylakoid lumen
Analogous to intermembrane space, high H+ concentration
Cytochrome
Passes electrons
H2O and O2 in photosynthesis
H2O is oxidized to O2 which diffuses across membrane
NADPH
Contains a phosphate group so that it won’t be used in glycolysis
Z Scheme
Photosystem II (P680) —> Cytochrome complex —> Photosystem I (P700) —> FD-NADP+ Reductase —> NADPH
Calvin Cycle
Location: Stroma
Inputs: NADPH, ATP, CO2
Outputs: GAP, O2, ADP, NADP+
3 Stages of Calvin Cycle
1) Carboxylation (add carbon to RuBP, degrades into 2 3 carbon molecules)
2) Reduction
3) Regeneration of RuBP
Carboxylation
CO2 is combined with RuBP via rubisco, unstable 6 carbon intermediate that splits into 2 × 3 carbon molecules
ETC Disruption
Atrazine blocks e- transfer at PS II
Paraquat blocks e- transport at PS1
Stoichiometry of Calvin Cycle
6 Carbon Enter, there are 6 RuBP molecules so 30C toatl. Go to 36C total (12 3PG) —> 12 G3P. Two G3P leave the cell as sugars (one glucose -6C). Rubisco generation
C3 Plants
CO2 is fixed directly by the enzyme rubisco in mesophyll cells. When O2 levels are high in hot conditions, photorespiration occurs.
C4 Plants
CO2 first enters in mesophyll cells. The enzyme PEP carboxylase fixes CO2 into a 4-carbon molecule. The 4 carbon molecule moves to the bundle sheath cells.
Central Dogma
DNA —> RNA —> Protein
Griffiths Experiment
S strain killed the mouse
Heat killed S strain did not kill the mouse
Heat killed mixed with R strain killed the mouse, must have been a transformation
Griffiths Experiment Techniques
1) Separated components of the heat killed S strain to see if transformation was present
2) Used chemicals/enzymes to destroy fractions that had the transforming property to see if that property could be specifically destroyed
Nucleotide Structure
Contains a sugar, nitrogenous base, and phosphate
5 carbon sugars (Draw)
Differ on the 2’ carbon, ribose contains a hydroxyl group, DNA does not
Purine and Pyramidens
Purine: Contains 2 rings. Includes Adenine and Guanine
Pyrimidines: Contain 1 ring. Include Thymine, Cytosine, and Uracil
Thymine and Uracil
Thymine contains a methyl group
Structure of Nucleotides
The base is always attached to the C1’ of the sugar via a B-glycosidic linkage
Sugar-Phosphate Backbone Structure
Polarity goes from 3’ —> 5’. Anti-parallel because of hydrophobic interactions’. Helix Nucleotide backbone connected via 3’-5; phosphodiester bonds
B-Form Helix
Contains 10.5 bp per turn
Right handed helix
Has major and minor grooves where DNA binds to
Linus Pauling
Proposed that DNA had a helix of 3 strands, bases on the outside and phosphate groups on the inside
DNA synthesis in eukaryotes vs prokaryotes
In prokaryotes: Happens continuously
In eukaryotes: There are checkpoints
Methods of DNA Replication
1) Semi Conservative
2) Conservative
3) Dispersive
Explain the Experiment and Results for Each
DNA Replication
1) DNA polymerase catalyzes the polymerization of deoxyribonucleotide triphosphates (dNTPs)
2) Requires a single-stranded DNA template
3) Newly synthesized strand must be antiparallel to the template strant. 5’ —> 3’ direction
4_ Requires a primer to a 3’ -OH group
Helicase
Separates DNA strands, allowing DNA polymerases access to single-stranded templates. Requires ATP hydrolyses
Single-stranded binding protein (SSBP)
Binds ssDNA after helicase, holds strands apart
Primase
Synthesizes a short (~10 nucleotides) RNA primer
Provides a 3’ -OH group for DNA polymerases to begin synthesis
Okazaki Fragments
Occur due to the 5’ to 3’ polymerizatiom
DNA Polymerase I
Digests the RNA primer (5’ —> 3’) and fills in the gap
DNA Ligase
Nick sealed by DNA ligase
Initiation of DNA Transcription
Starts at oriC. DnaA hexamer binds to binding sites. Lots of AT base pairs
Draw Replication (two sided with helicase)
Plasmid
Small circular extrachromosomal DNA molecules that replicate independently of the bacterial host chromosome and have fixed origions of replications (ori)