C4 - Predicting and Identifying reactions and products
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
Appearance when an alkali metal is freshly cut
- silvery and shiny
Appearance when an alkali metal is cut and left in air
- tarnishes
- turns grey/black quickly
Why are group one elements stored in oil
- because they are very reactive and would react with the water and oxygen in air
Physical Properties of alkali metals
- low mp and bp
- low density (Li, NA, K can float on water)
- soft (increases as you descend the group)
What happens when Lithium is added to water
- fizzes
-melts
- floats
- moves across surface of the water
What happens when Sodium is added to water
- fizzes
-melts
- floats
- moves across surface of the water
- catches fire
What happens when Potassium is added to water
- fizzes
-melts
- floats
- moves across surface of the water
- catches fire
- hydrogen gas produced
PH of metal hydroxide
- alkaline
Metal + oxygen —->>>
Metal oxide
Metal + water ——>
Metal hydroxide + hydrogen
Metal + chlorine ——>>
Metal chloride
Why is reacting metal + chlorine dangerous
- it is highly reactive and can form volatile and explosive compounds
Rubidium and Caesium reaction in water
- react vigorously and violently
- explode
Why does reactivity increase as you go down group one
- group 1 elements react by losing one electron
- as you go down the group there is more shielding
- because the outer shell electron gets further away from the nucleus there is less nuclear attraction
- easier to lose an electron
Why does density increase down group one
- even though the atoms get larger the mass of the atom increases as there are more protons and neutrons
- density is mass over volume so it increases
What determines the group of an element
- if the elements in the group have similar chemical properties
- they will react in a similar way because they have the same number of outer shell electrons
How do group 7 elements exist
- diatomic molecules
- one covalent bond between them
Why does reactivity decrease down group 7
- group 7 elements react by gaining one electron
- as you go down the group there is more shielding
- because the outer shell electron gets further away from the nucleus there is less nuclear attraction
- more difficult to gain an electron
Physical properties of group 7
- mp and bp increase as you go down
Colour and State of F2 at rt
Pale yellow gas
Colour and State of Cl2 at rt + properties
Pale green gas
- low bp
-quite reactive
- poisonous
Colour and State of Br2 at rt + properties
- reddish brown liquid
- gives off an orange vapour
- poisonous
Colour and State of F2 at rt + properties
- dark grey crystalline solid
- gives off purple vapour when heated
What are metal halides
salts
- formed from alkali metals + halogens
Which halogens can Cl2 displace
- Br2
-I2
(out of the 3)
Which halogens can Br2 displace
- I2 (out of the 3)
Which halogens can I2 displace
none (out of the 3)
Example of displacement reaction
Chlorine + potassium bromide —————> Bromine + potassium chloride
Cl2 + 2KBr —————> Br2 + 2KCl
Practical to show reactivity trends of displacement reactions
- 1) measure out a small volume of a halide salt solution in a test tube
- 2) add a few drops of a halogen solution to the test tube and shake gently
- 3) observe to see if there is a colour change to know the reaction has happened
- If there is no colour change then the halogen did not displace the halide ions from the salt
- 4) repeat using different combinations of halide salt and halogen
Colour change when Br2 is formed in displacement reaction from halogens and halide ions in salts
- orange solution
Colour change when I2 is formed in displacement reaction from halogens and halide ions in salts
- brown solution
Physical properties of noble gases
- colourless
- gases at rt
- monatomic (made up of single atoms so just He instead of He2)
- inert
- non flammable
- bp and mp increase down the group
- density increases down the group
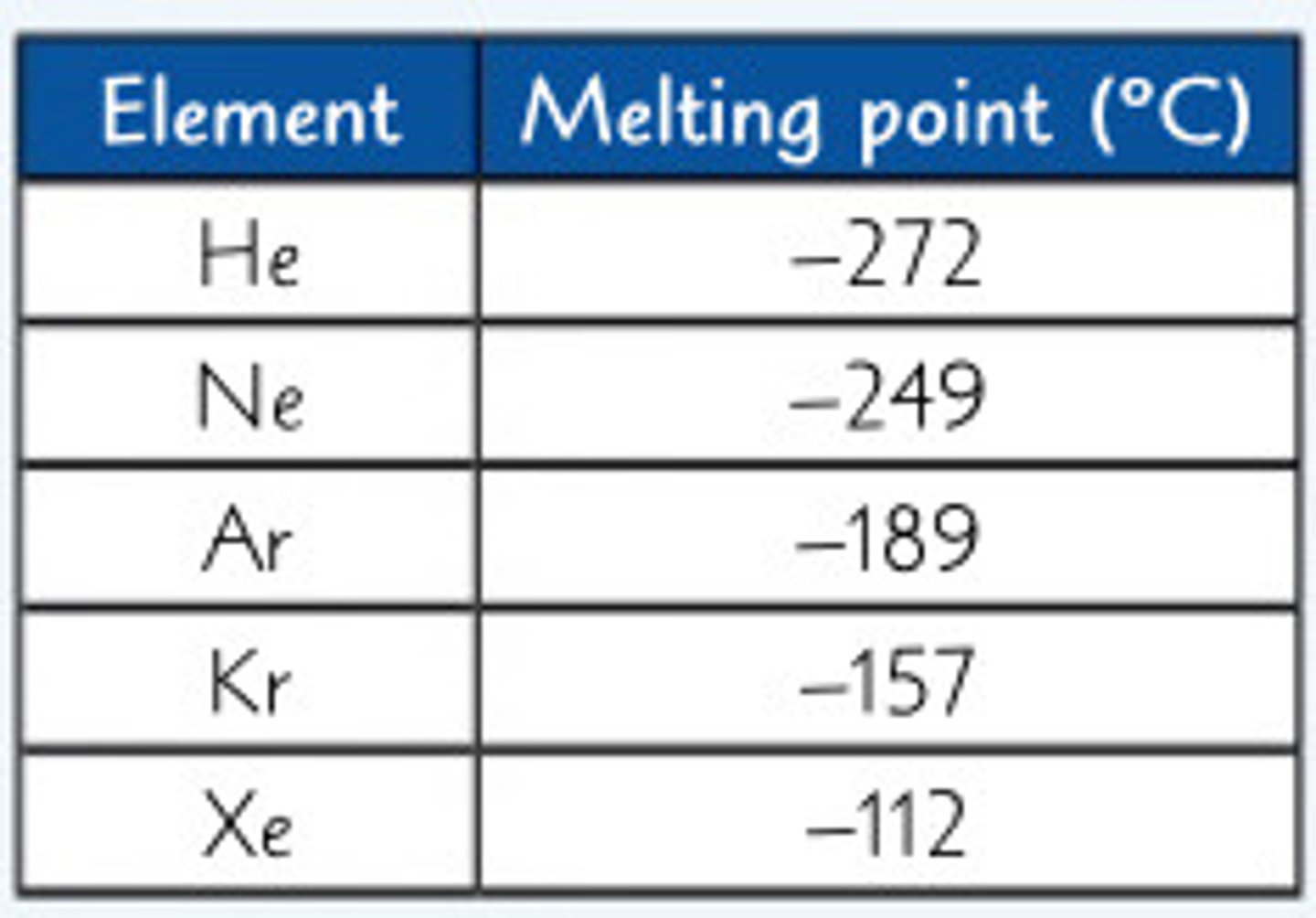
How to predict the mp/bp of an element based on information given in a table
—-> EG predict the mp of Radon
- identify the general trend in data
(Mp increases as you go down)
- calculate the gaps between each element
(He to Ne is 23, Ne to Ar is 60, Ar to Hr is 32, Kr to Xe is 40)
- take an average of all the gaps
(40)
- add 40 to mp of Xe (add bc of the trend)
(-112 + 40 = 72)
Use the densities of helium (0.2 kg/m') and argon (1.8 kg/m?) to predict the density of neon.
- Neon comes between helium and argon in the group, so you can predict that its density will be roughly halfway between their densities: (0.2 + 1.8) ÷ 2 = 1
- Neon should have a density of about 1.0 kg/m'.
Properties of transition metals
- hard
- strong
- shiny
- good conductors of heat and electricity
- high mp (apart from mercury bc it is a liquid at rt)
- high densities (generally higher than group one)
Chemical properties of transition metals
- transitions metals and their compounds make good catalysts
(E.g iron in the haber process for making ammonia and Vanadium pentoxide in the contact process for making sulfuric acid )
- often have more than one ion (e.g Fe2+ and Fe3+)
- form colourful compounds
- relatively unreactive
why will iron react with steam and not water
- gas molecules in steam have more energy so they will collide faster with the iron particles
Reactivity series order
Please - Potassium
Send - Sodium
Charlies - Calcium
Monkeys - Magnesium
And - Aluminium
(Carbon)
In - Iron
To - Tin
Lead - Lead
(Hydrogen)
Cages - Copper
Securely - Silver
Guarded - Gold
Please - Platinum
Precautions for tests for gases
- carry out in a fume cupboard if gases are toxic
- wear goggles
- wear gloves
Test for CO2 gas
- bubble CO2 through limewater
- limewater will turn milky
Test for H2 gas
- a lighted splint makes a squeaky pop sound
Test for O2 gas
- a glowing splint will relight
Test for Cl2 gas
- take damp blue litmus paper
- it will turn red (bc a solution of chlorine is acidic) then bleach white
test for halide ions
— add some dilute nitric acid
- add a few drops of silver nitrate solution
Results for halide ion test
- chloride = white ppt
Ag+(aq) = Cl-(aq) —————> AgCl(s)
- bromide = cream ppt
Ag+(aq) = Br-(aq) —————> AgBr(s)
- Iodide = yellow ppt
Ag+(aq) = I-(aq) —————> AgBr(s)
Why is nitric acid added in halide ion test
- to get rid of any carbonate ions that would change the results
Why do you have to use nitric acid and not hydrochloric acid in halide ion test
- you would be added chloride ions which can change the results
Test for carbonate ions
- add some barium chloride solution
- white ppt of barium carbonate will form
- THEN, add some dilute HCl
- if carbonate ions are present then the mixture will bubble and fizz because carbon dioxide gas will form
- collect the gas and do CO2 test
- after all the barium carbonate has reacted there will be a colourless solution of Barium ions left
Test for sulfate ions
- add some barium chloride solution
- white ppt of barium carbonate will form
- THEN, add some dilute HCl
- barium sulfate will not react with the HCl so the white ppt wont dissolve so you know sulfate ions are present
How to carry out a flame test
- clean the loop of a nichrome wire by dipping it into HVL and rinsing in deionised water
- dip the wire loop into a sample of the metal compound
- put the loop at the edge of a roaring blue Bunsen flame
(that is the clear part so you can see the colour best)
- record the colour of the flame
Why is flame test not good if the compound contains for than one type of metal ion
- you will get a mixture of colours so it will be difficult to tell which metals are present
Flame colour of lithium
Crimson red
Flame colour of sodium
yellow
Flame colour of potassium
lilac
Flame colour of calcium
Brick red
Flame colour of copper
blue-green
Test for cations using sodium hydroxide
- add a few drops of sodium hydroxide solution to the mystery solution
- check what metal ion is present based on the colour of hydroxide ppt
Colour of hydroxide ppt for Calcium and ionic equation
White
Ca2+ (aq) + 2OH- (aq) —————> Ca(OH)2 (s)
Colour of hydroxide ppt for copper and ionic equation
Blue
Cu2+ (aq) + 2OH- (aq) —————> Cu(OH)2 (s)
Colour of hydroxide ppt for Iron 2+ and ionic equation
Green
Fe2+ (aq) + 2OH- (aq) —————> Fe(OH)2 (s)
Colour of hydroxide ppt for Iron 3+ and ionic equation
Brown
Fe3+ (aq) + 3OH- (aq) —————> Fe(OH)3 (s)
Colour of hydroxide ppt for Zinc and ionic equation
White at first then you add sodium hydroxide in excess and it will form a colourless solution
Zn2+ (aq) + 2OH- (aq) —————> Zn(OH)2 (s)
THEN
Zn(OH)2 (s) + 2OH- (aq) —————> Zn(OH)4 2- (aq)
—-> that product is an ion bc the zinc hydroxide dissolves so its ions are left in the solution
What are instrumental methods
- using machines to analyse and identify elements and compounds
Advantages of instrumental analysis
- very sensitive (they can detect even the tiniest amounts of a substance)
- very fast and tests can be automated)
- very accurate (dont contain human error like manual analysis does)
How does infrared spectroscopy work
- it produces a graph showing frequencies of infrared radiation that a molecule absorbs or transmits
- you can use the absorbance pattern on the graph to identify the molecule
How does ultraviolet spectroscopy work
- it produces a graph showing frequencies of ultraviolet radiation that a molecule absorbs or transmits
- you can use the absorbance pattern on the graph to identify the molecule
How does gas chromatography work
- used to separate the chemicals in a mixture
- produces a graph (chromatogram) with one peak for each chemical
- the retention time can be used to identify the chemical
How does mass spectroscopy work
- a technique used to find the relative molecular mass of a mystery comping or relative atomic mass of a mystery element
- an electron is removed from each molecule of the mystery sample to form a molecular ion (M+)
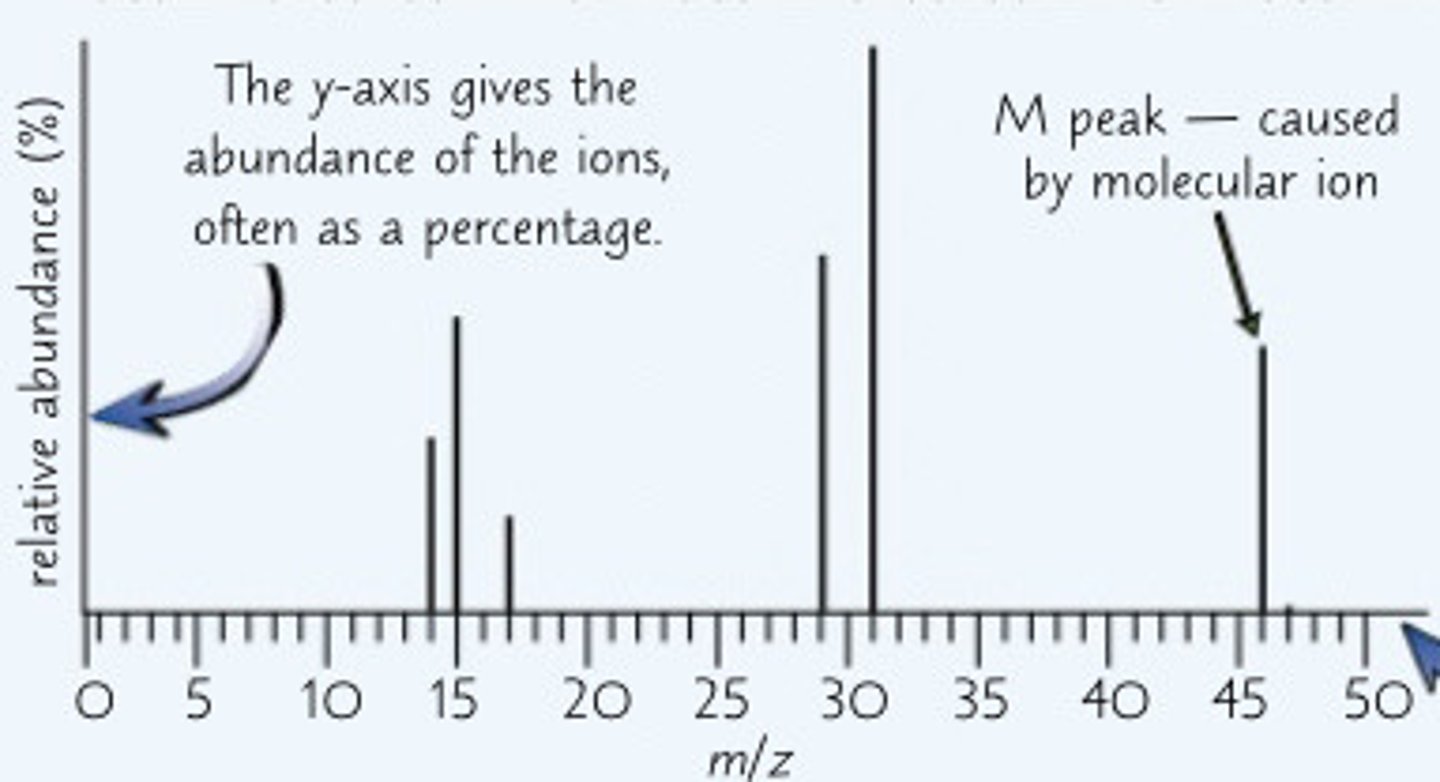
What does the last peak on a mass spectroscopy graph tell you
- the value on the X axis of the last peak is the Mr of the compound
- the last peak is called the molecular ion peak (the M peak)
Example: determine the identity of the alcohol from the spectrum
- The m/z value of the M peak is 46, so the Mr of the alcohol must be 46
- If you calculate the molecular masses of the first few alcohols, you'll find that the one with an Mr of 46
- Mr of ethanol = (2 × 12.0) + (5 × 1.0) + 16.0 + 1.0 = 46.0
- The alcohol that produced this mass spectrum must be ethanol