Nitrogen Metabolism - Applications to Health and Disease
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
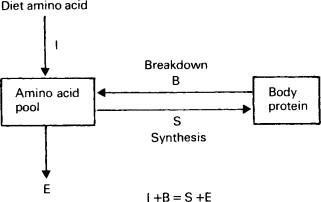
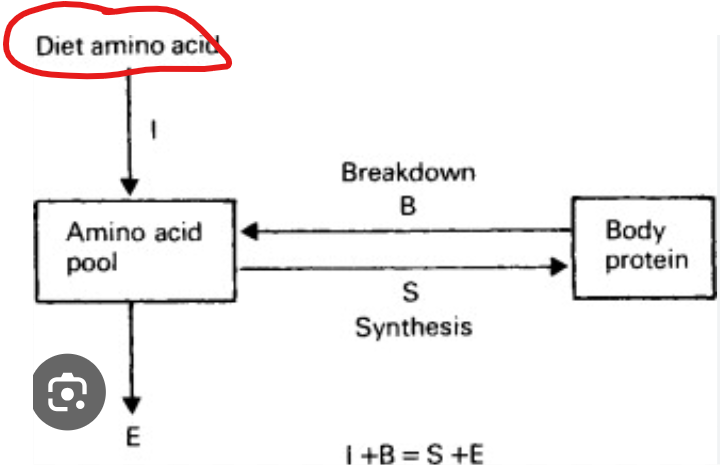
What is nirogen balance
a fundamental concept in metabolism that measures the relationship between nitrogen intake and nitrogen excretion.
In humans, where does nitrogen primarily come from
amino acids in the diet
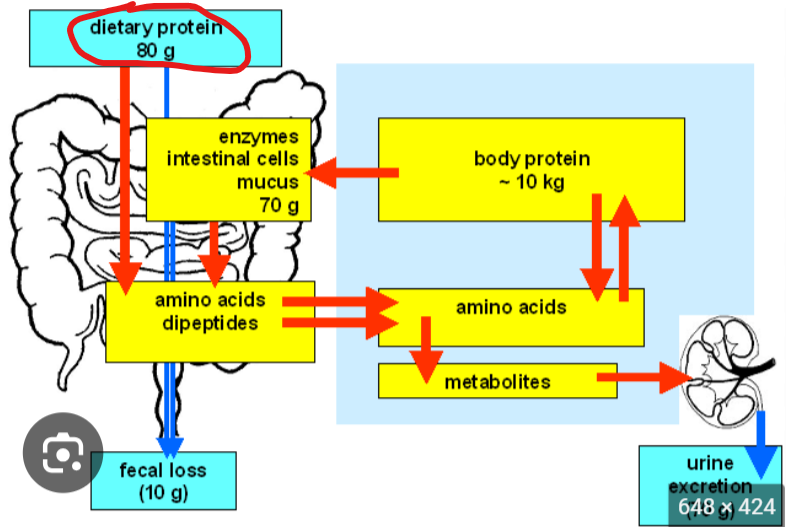
What can amino acids be
essential
non essential
conditionally essential

What happens when nitrogen intake equals nitrogen excretion
the body is said to be in zero nitrogen balance, a state common in healthy adults consuming a balanced diet
What does nitrogen excretion occur mostly as
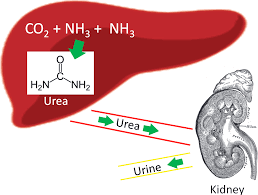
urea derived from protein turnover and excess dietary protein

What state are we in if more nitrogen is excreted than ingested
negative nitrogen balance
when does negative nitrogen balance occur
starvation
malnutrition
or catabolic stress such as trauma or burns.
Why does negative nitrogen balance occur during starvation, malnutrition, or catabolic stress such as trauma or burns
because amino acids are broken down for their carbon skeletons to support energy production or gluconeogenesis, leaving the nitrogen to be excreted

When does positive nitrogen balance occur
when nitrogen intake exceeds excretion
Positive nitrogen balance occurs when nitrogen intake exceeds excretion
When is it essential

periods of growth
pregnancy
tissue repair- where the bnody is actively incorporating amino acids into proteins
What can happen due to severe negative nitrogen balance
malnutrition syndromes
What are the malnutrition syndromes that can occur from severe negative nitrogen balance
Marasmus
Kwashiorkor
Which malnutirion syndrome that occurs from severe negative nitrogen balance is being described:
global wasting of muscle and fat due to general nutrient deficiency
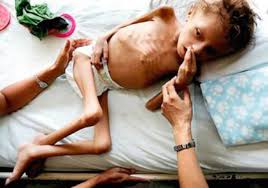
Marasmus
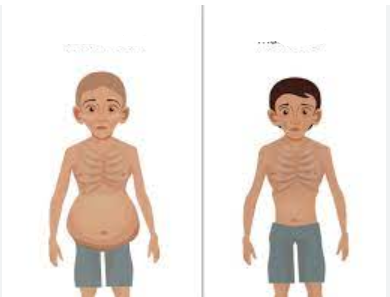
Which malnutirion syndrome that occurs from severe negative nitrogen balance is being described:
protein-specific deficiency leading to edema, hepatomegaly, and a swollen belly
Kwashiorkor
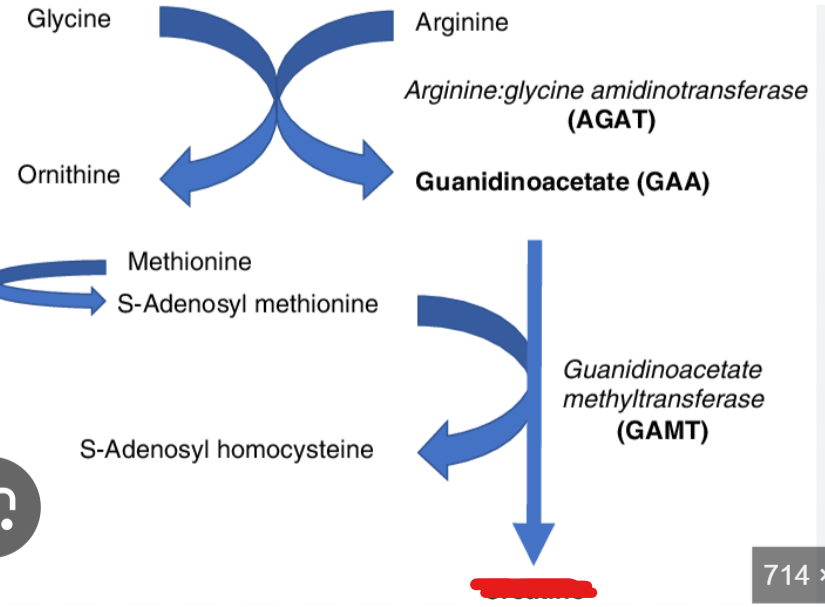
What is a nitrogen containing compunds derived from arginine,glycine, and methionine-
it plays a critical role in energy storage and rapid ATP regeneration particularly in skeletal muscles and the brain
Creatine
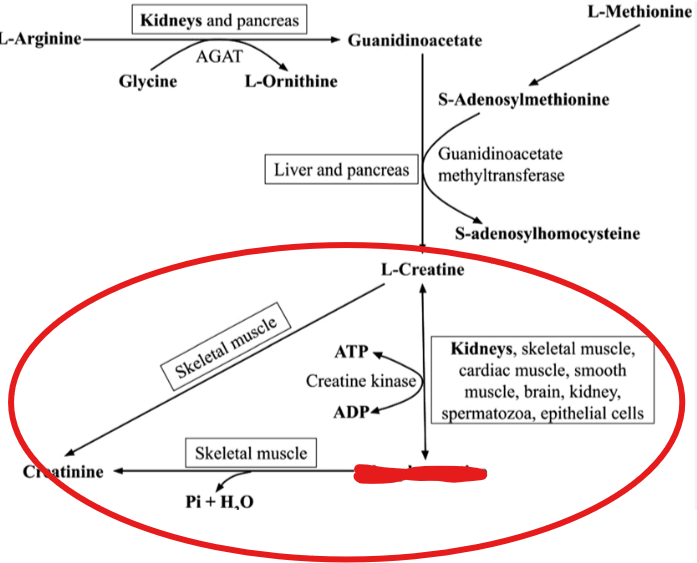
What is creatine converted into- in which it serves as a high energy phosphate reserve that can quickly replenish ATP during periods of high energy demand
phosphocreatine
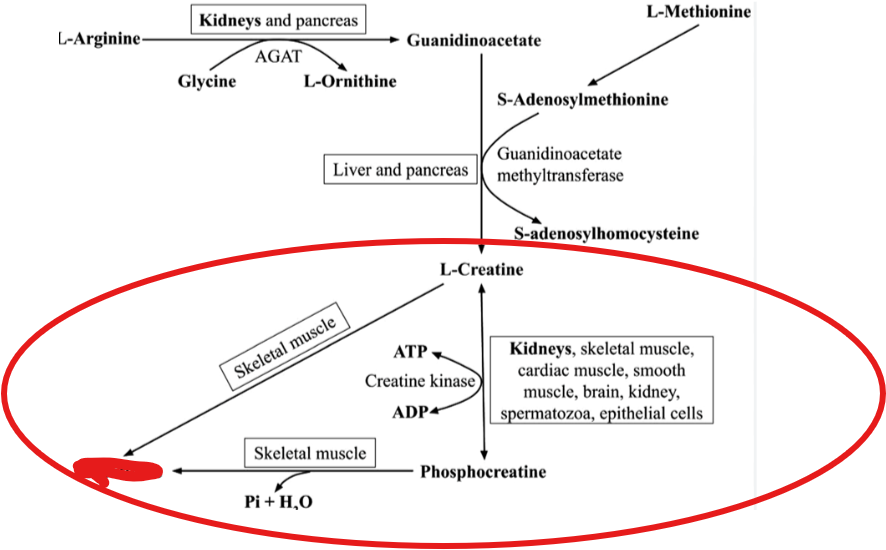
What is the breakdown product of creatine and phosphocreatine, excreted in urine
creatinine
Why does creatinine serve as a clinincal biomarker for renal function
because creatinine levels are relatively stable and propotional to muscle mass- it is filtered exclusively by the kidneys once it is removed from the blood- this means that high levels found within the blood signify renal dysfunction
What does elevated serum creatine indicate?
kidney impairment or conditions of extreme muscle breakdown, such as rhabdomyolysis.
What does dysregulation of the creatin/phosphocreatine system also contribute to due to insufficient energy buffering.
neurological and muscular disorders
What is often called the 21st amino acid, and unique because it conatins selenium an essential micronutrient
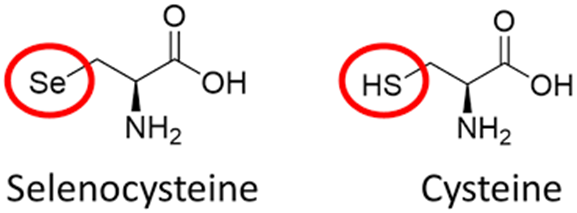
Selenocysteine
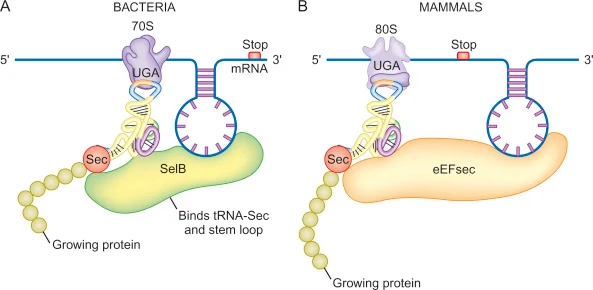
Unlike other amino acids where is selenocysteine is incorporated
its incoporated into proteins at the UGA codon - which normally signals translation termination
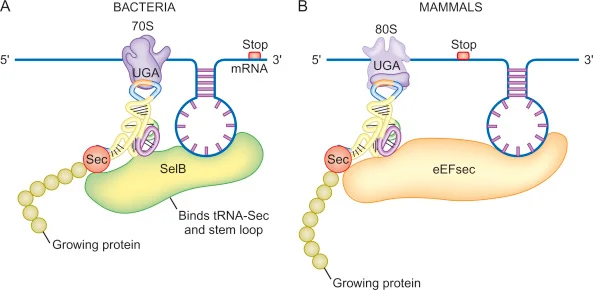
What does the incorporation of selenocysteine require
a selenocysteine insertion sequence (SECIS) in the mRNA, along with sufficient selenium availability
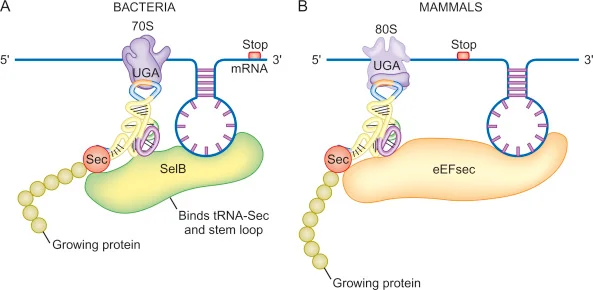
What happens if selenocysteine is absent
the UGA codon fucntions as a stop signal as prevents the synthesis of selenoproteins
If selenium is absent, the UGA codon functions as a stop signal. What is the result of this=?
truncated, nonfunctional protein
It can lead to severe health issues, including thyroid dysfunction (hypothyroidism), muscle pain/weakness, cognitive decline/brain fog, male infertility, and weakened immunity. It is crucial for antioxidant defense, and its absence often causes increased oxidative stress
Selenoproteins are crucial for multiple physiological functions. What are some examples?
antioxidant defense
redox balance
thyroid hormone metabolism
neurological and immune protection
Selenoproteins are crucial for multiple physiological functions, including glutathione peroxidase- what enzyme is used here
glutathione peroxidase
Selenoproteins are crucial for multiple physiological functions, including redox balance, what enzyme is used here
thioredoxin reductase
Selenoproteins are crucial for multiple physiological functions such as thyroid hormone metabolism , what enzyme is used here?
deiodinases that convert T4 to T3
What are nitrogenous, cycliuc molecules that serve as essential intermediates in heme biosynthesis
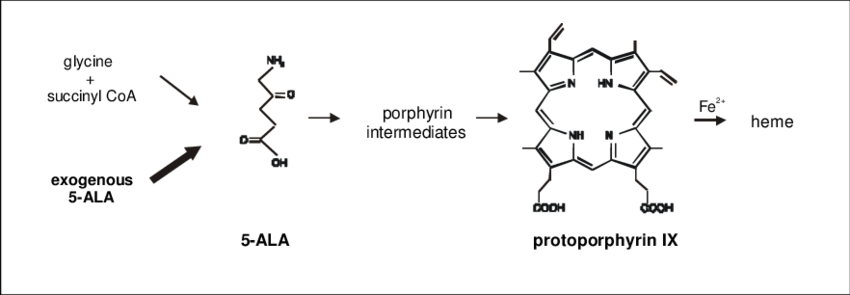
Porphyrins
What is the prosthetic group for hemoglobin , myoglobin, cytochromes, and other redox- active enzymes
Heme
What does porphyrin syntheis begin with
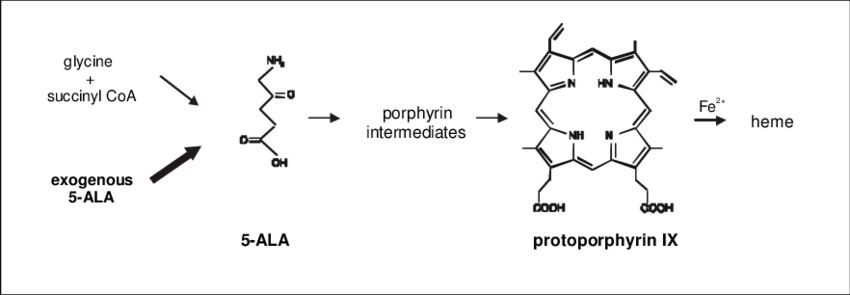
glycine and succinyl-CoA in the mitochondria- highlighting that amino acids are not just building blocks for proteins but also for critical metabolic cofactors
What are the molecules glycine and succinyl-CoA capabale of doing?
they are capable of binding metals and participating in electron transfer reactions, making them indispensable for respiration and other biochemical energy transformations
What can dysregulation in porphyrin metabolism result in
porphyrias
What are porphyrias?
group of rare disorders characterized by the accumulation of porphyrins
What kind of symptoms can Porphyrias present with depending on the specific enzymatic effect ?
cutaneous symptoms
neurological symptoms
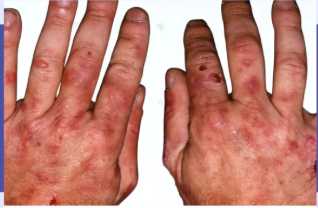
What are some examples of cutaneous symptoms
photosensitivity, blisters
what are some examples of neurological symtoms
abdominal pain , neuropathy
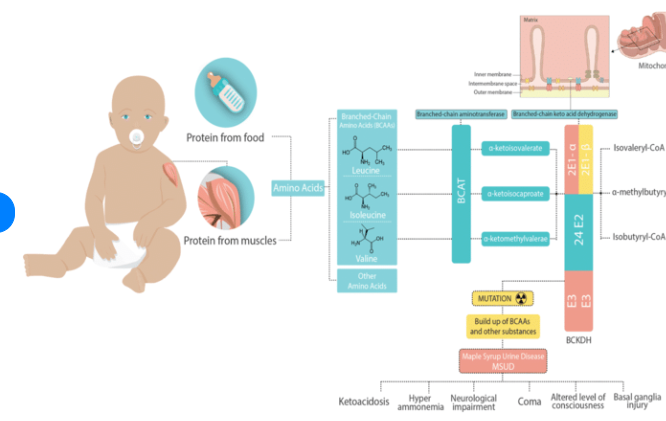
What is a rare, genetic disorder affecting the catabolism of branched- chain amino acids (BCAAs)
Maple Syrup Urine Disease ( MSUD)
What branched chain amino acids are havng thier catabolim affected by MSUD
leucine, isoleucine, and valine
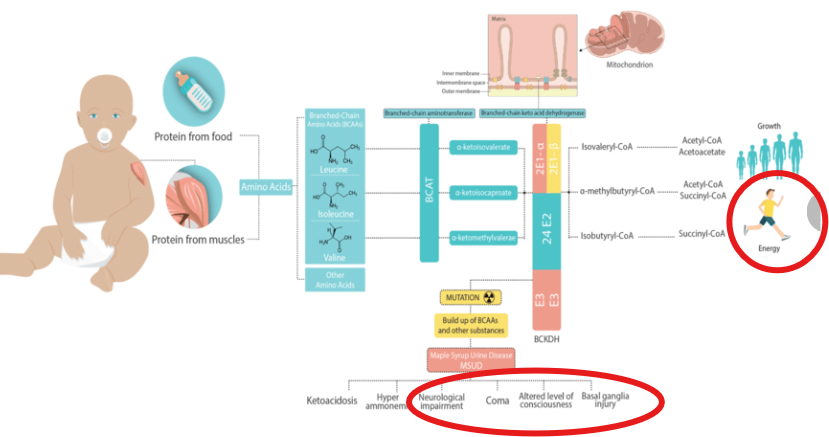
What do branched-chain amino acids (BCAAs) usually do?
they serve as energy substrates ans contribute to neurotransmitter synthesis in the brain
Normally, BCAAs serve as energy substrates and contribute to neurotransmitter synthesis in the brain.
What happens in MSUD?
A deficieny in the branched-chain α-keto acid dehydrogenase complex leads to accumulation of these amino acids and their ketoacid derivatives in plasma, urine, and tissues.
How did the Maple Syrup Urine Disease get its name
the sweet odor of the urine, reminiscent of maple syrup.
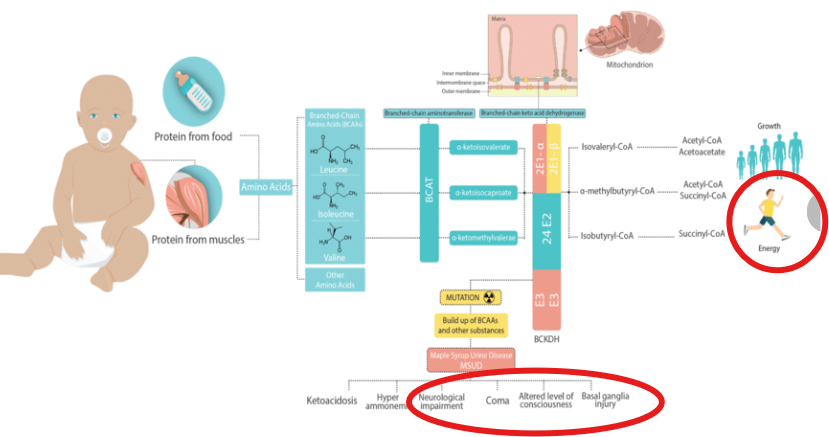
What can untreated MSUD result in
severe neurological damage, seizures, coma, or even death within days of birth
What is management of MSUD?
early dietary restriction of BCAAs and careful metabolic monitoring to prevent toxic accumulation.
What is a genetic disorder charcterized by elevated homocysteine- a metabolite of methionine
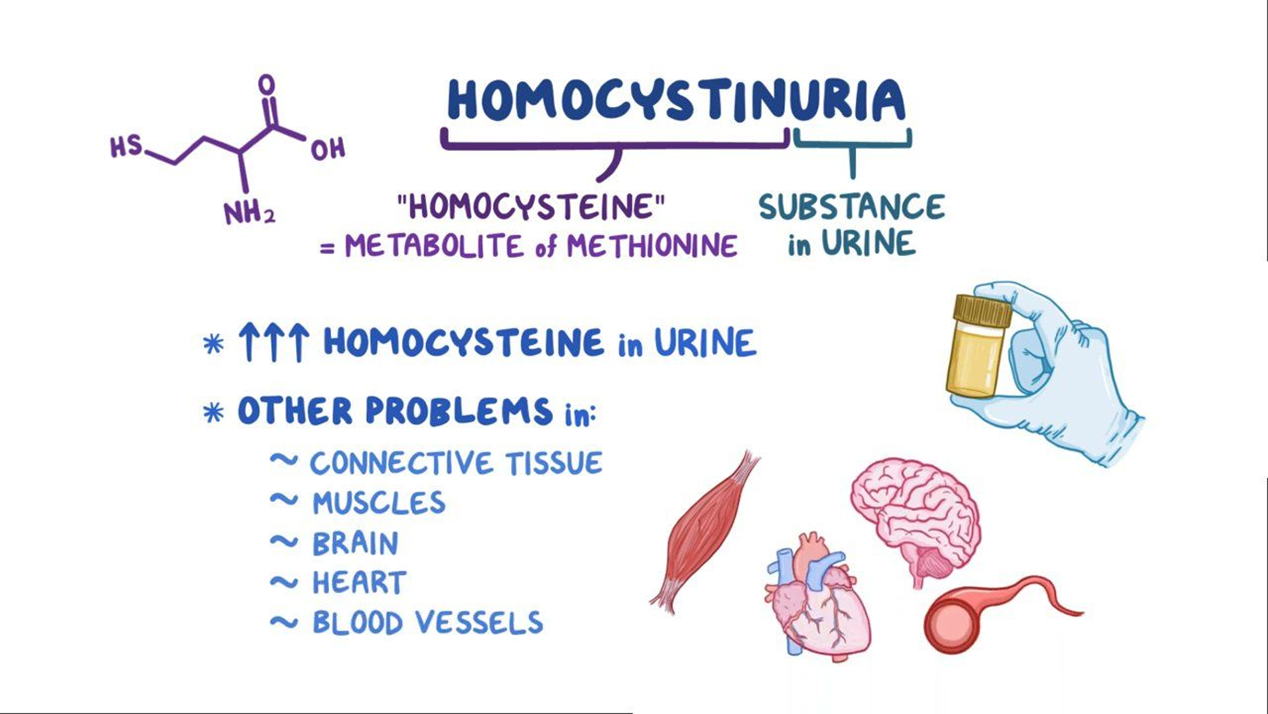
Homocystinuria
What does Homocystinuria often result from
deficiencies in PLP-dependent enzymes, linking it to vitamin B6 metabolism, as B6 is converted into pyridoxal phosphate (PLP), the active cofactor form
What can elevated homocysteine lead to
vascular, ocular, skeletal, and neurological problems
In homocystinurina, what can sometimes ameliorate symptoms by restoring enzyme activity
B6 supplementation
What is heme, after serving its role in hemoglobin and other heme proteins, is degraded to
bilirubin, a lipophilic molecule
What do low physiological levels of bilirubin indicate
they may be cytoprotective, acting as an antioxidant
What does excessive accumulation of bilirubin indicate
toxicity due to its ability to disrupt membranes
What is the relation between fetal development and bilirubin
During fetal development, bilirubin produced from red blood cell breakdown exists as unconjugated bilirubin, which cannot be efficiently processed by the fetal liver due to low activity of conjugating enzymes. As a result, bilirubin is transferred across the placenta into the maternal circulation, where it is conjugated and eliminated. This placental mechanism prevents accumulation of toxic bilirubin in the fetus.
After birth, the placenta is no longer available for bilirubin clearance, and the neonate must rely on its own liver.
However, hepatic conjugation enzymes are still immature, leading to decreased bilirubin clearance. At the same time, increased red blood cell turnover raises bilirubin production. This imbalance results in accumulation of unconjugated bilirubin, causing Neonatal jaundice, which in severe cases can lead to Kernicterus.
what is neonatal jaundice
bilirubin crosses the blood-brain barrier and causes hyperbilirubinemia.
What can convert bilirubin into water soluble forms that are more easily excreted
Phototherapy (blue/green light)
What is a metabolic disorder caused by phenylalanine hydroylase deficiency, preventig the converstion of phenylalanine to tyrosine
Phenylketonuria (PKU)
How can elevated phenylalanine impact an individual
Elevated phenylalanine levels are neurotoxic and can impair brain development, leading to intellectual disability, delayed social and motor skills, tremors, seizures, and hypopigmentation due to lack of tyrosine-derived melanin.
How is PKU detected?
early via newborn heel-prick tests
What is treatment for PKU
a strict low-phenylanine diet- often supplemented wuth tyrosine
What does pyrimidine biosynthesis generate
the nucleotide bases cytosine, thymine, and uracil
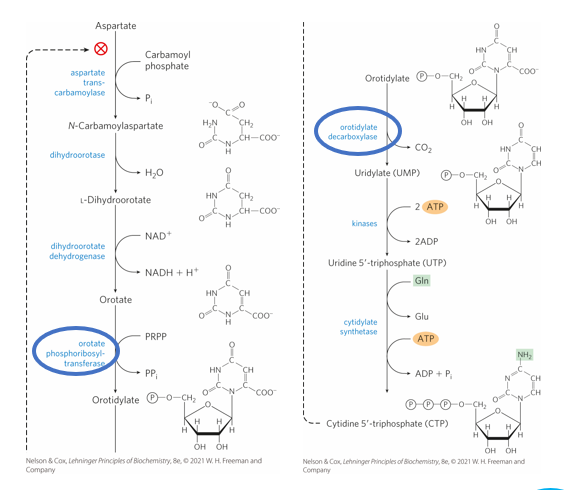
What can casue orotic aciduria and cause an accumulation or orotate
Inherited enzyme deficiencies, some examples are that it could be found in are
orotate phosphoribosyltransferase
orotidine-5'-phosphate decarboxylase
What do defects in pyrimidine synthesis result in
megaloblastic anemia
what is megaloblastic anemia
a condition of impaired DNA synthesis
Unlike B12-responsive megaloblastic anemia, what is pyrimidine- related anemia unresponsive to which is a block directly in nucleotide synthesis rather than folate metabolism
B12
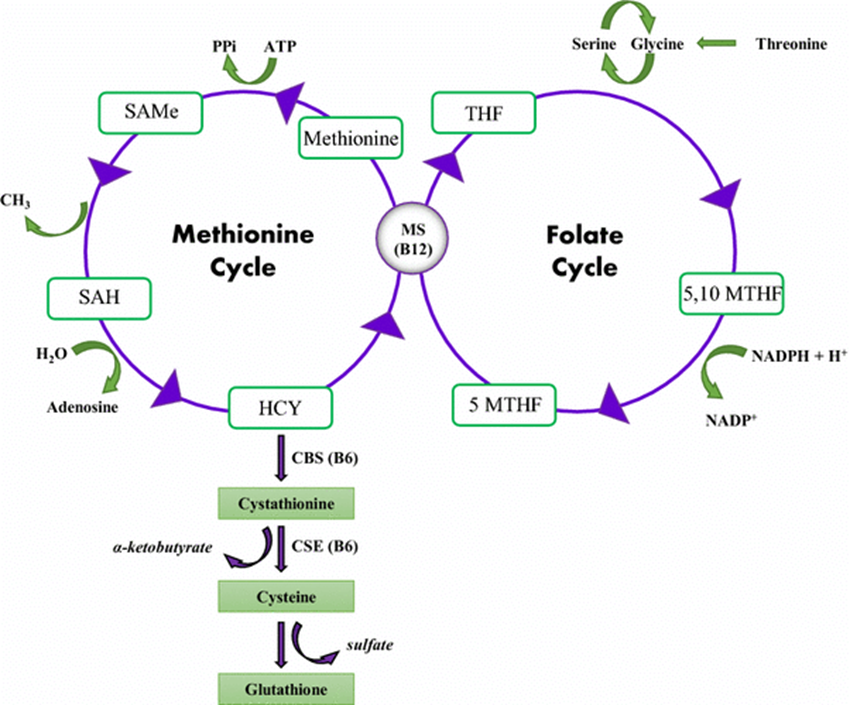
In Folate and Thymidylate Synthase in DNA Synthesis, what does Thymidylate synthase do ?
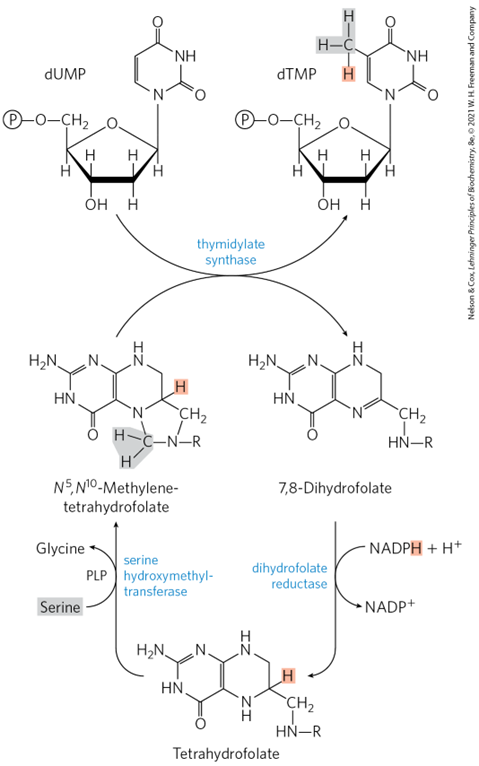
converts dUMP to dTMP, providing the sole de novo source of thymidine for DNA synthesis.
In Folate and Thymidylate Synthase in DNA Synthesis, Thymidylate synthase converts dUMP to dTMP, providing the sole de novo source of thymidine for DNA synthesis.
What does this reaction require as a methyl donor
N^5,N^10-methylene tetrahydrofolate
In Folate and Thymidylate Synthase in DNA Synthesis, Thymidylate synthase converts dUMP to dTMP, providing the sole de novo source of thymidine for DNA synthesis.
This reaction requires N^5,N^10-methylene tetrahydrofolate as a methyl donor
What does folate deficieny limit>
dTMP production,
what does limitations on dTMP production, cause
impaired DNA synthesis, and leading to megaloblastic anemia
Thymidylate synthase converts dUMP to dTMP, providing the sole de novo source of thymidine for DNA synthesis.
This reaction requires N^5,N^10-methylene tetrahydrofolate as a methyl donor.
Folate deficiency limits dTMP production, impairing DNA synthesis, and leading to megaloblastic anemia.
What drug inhibits thymidylate synthase by forming a covalent complex with the enzyme
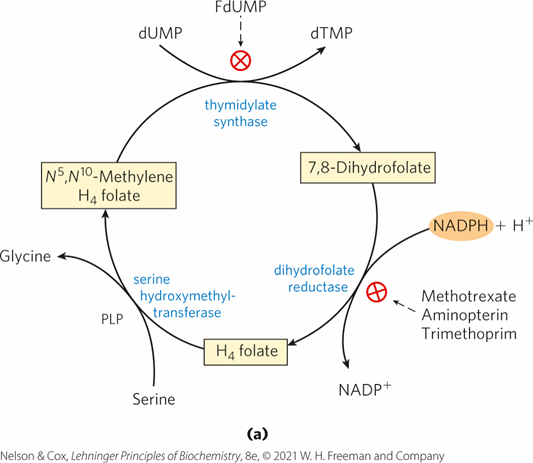
5-fluorouracil (5-FU)
Thymidylate synthase converts dUMP to dTMP, providing the sole de novo source of thymidine for DNA synthesis.
This reaction requires N^5,N^10-methylene tetrahydrofolate as a methyl donor.
Folate deficiency limits dTMP production, impairing DNA synthesis, and leading to megaloblastic anemia.
What drug inhibits dyhydrofolatye reductase, depleting the tetrahydrofolate pool
methotrexate
Thymidylate synthase converts dUMP to dTMP, providing the sole de novo source of thymidine for DNA synthesis.
This reaction requires N^5,N^10-methylene tetrahydrofolate as a methyl donor.
Folate deficiency limits dTMP production, impairing DNA synthesis, and leading to megaloblastic anemia.
Drugs such as 5-fluorouracil (5-FU) inhibit thymidylate synthase by forming a covalent complex with the enzyme, while methotrexate inhibits dihydrofolate reductase, depleting the tetrahydrofolate pool
What do both drugs target
rapidly dividing cells for chemotherapy
Purines can be synthesized via which pathway
de novo
Purines can be recovered via which pathway
salvage pathways
What arises from HGPRT deficiency-impairing purine salvage and causing accumulation of uric acid.
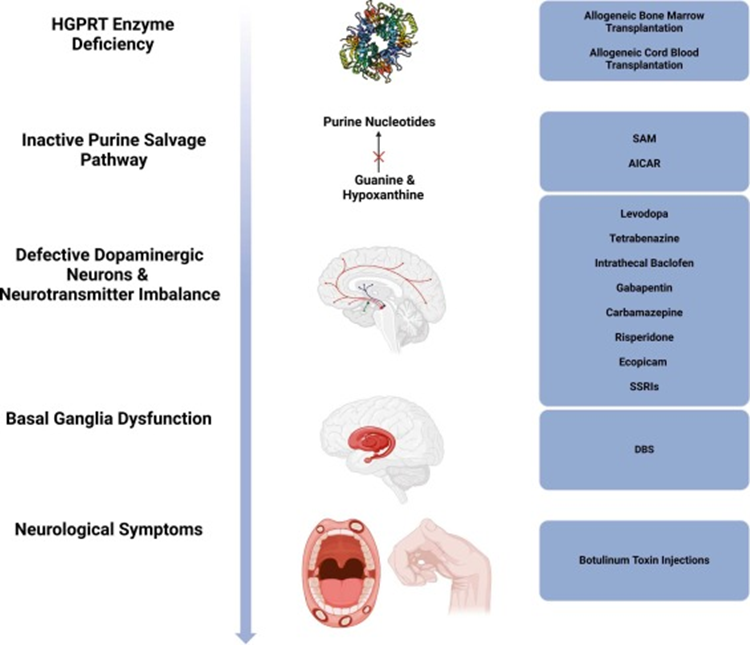
Lesch Nyhan syndrome
What does Nyhan syndrome lead to
hyperuricemia, gout, self-injurious behaviors, and motor dysfunction.
What is gout caused by
chronic hyperuricemia- either from overproduction of uric acid (purine catabolism) or underexcretion by the kidneys
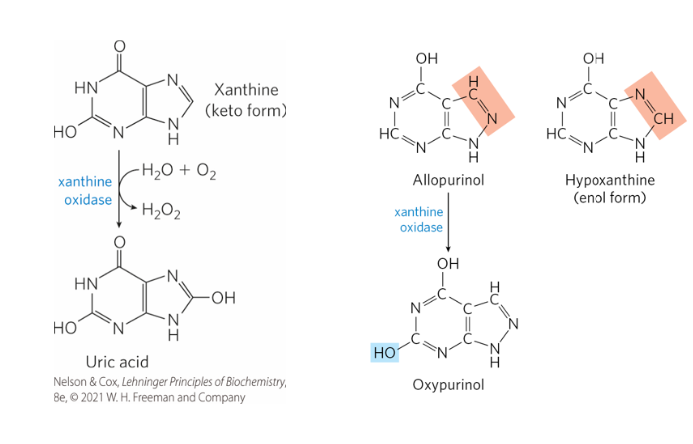
What happens in gout
Crystallized uric acid deposits in joints cause inflammation and pain, commonly in the big toe.
What is the treatment for Gout
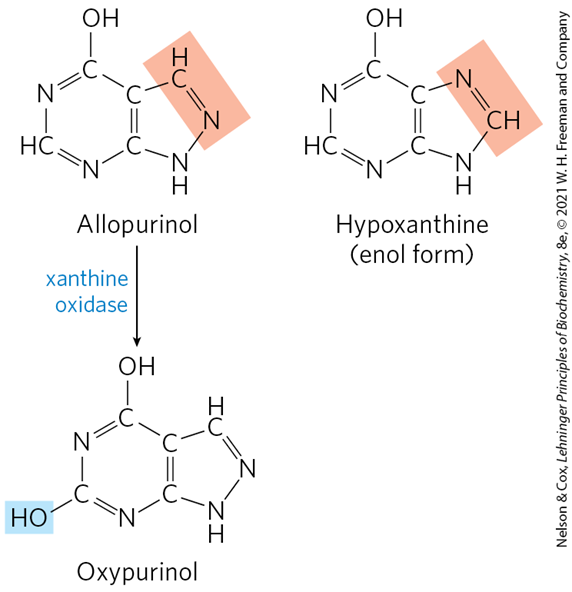
allopurinol, an inhibitor of xanthine oxidase, which reduces uric acid production.