Shapes of molecules
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
What is the Valence shell?
the outermost shell of an atom where the electron sharing takes place
What is the electron pair repulsion theory?
pairs of electrons repel each other (as both have -ive charge) so they adopt certain shapes and bond angles
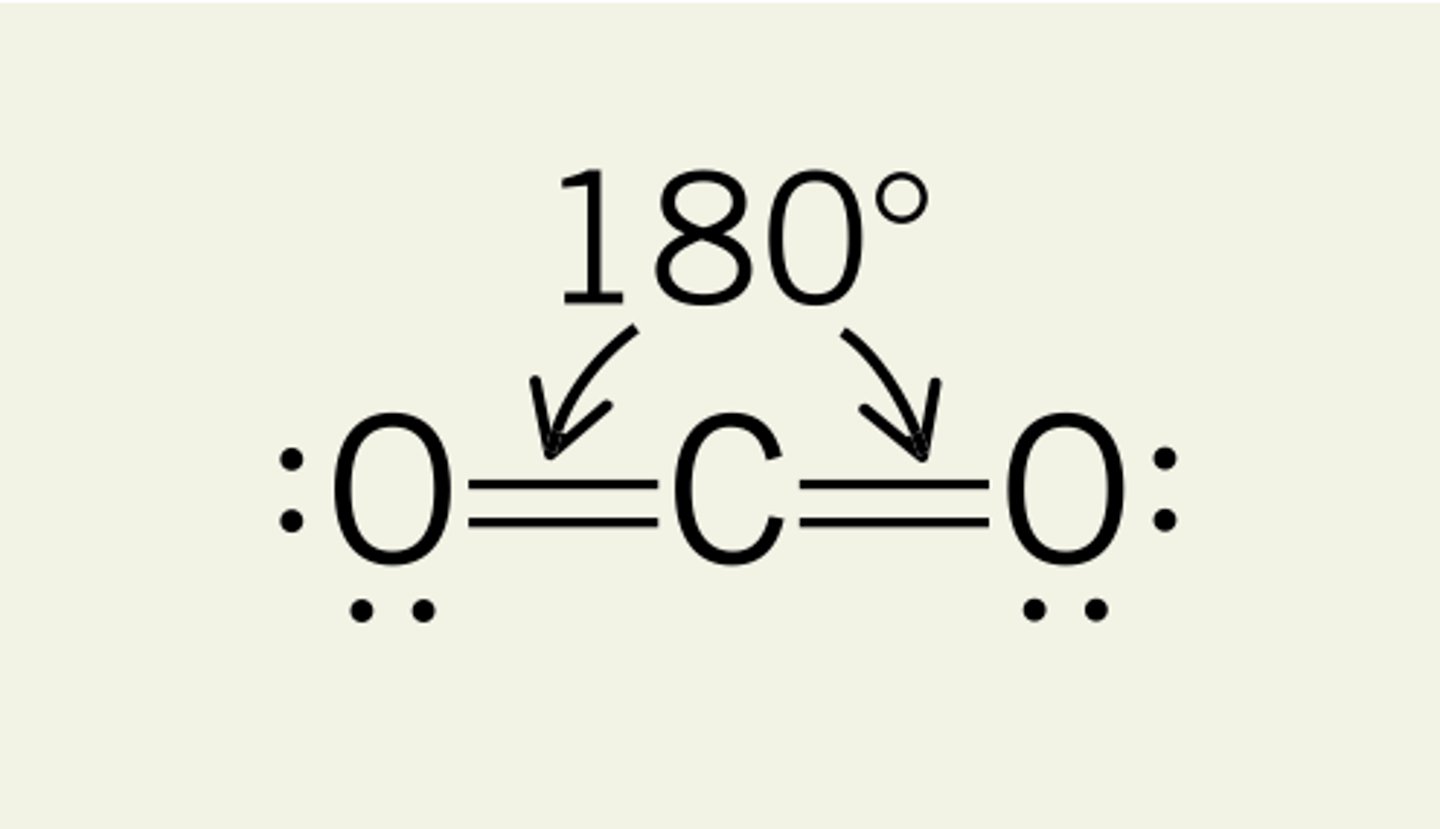
Linear shape
-180 degrees
-2 bonding pairs/regions
-no lone pairs on central atom
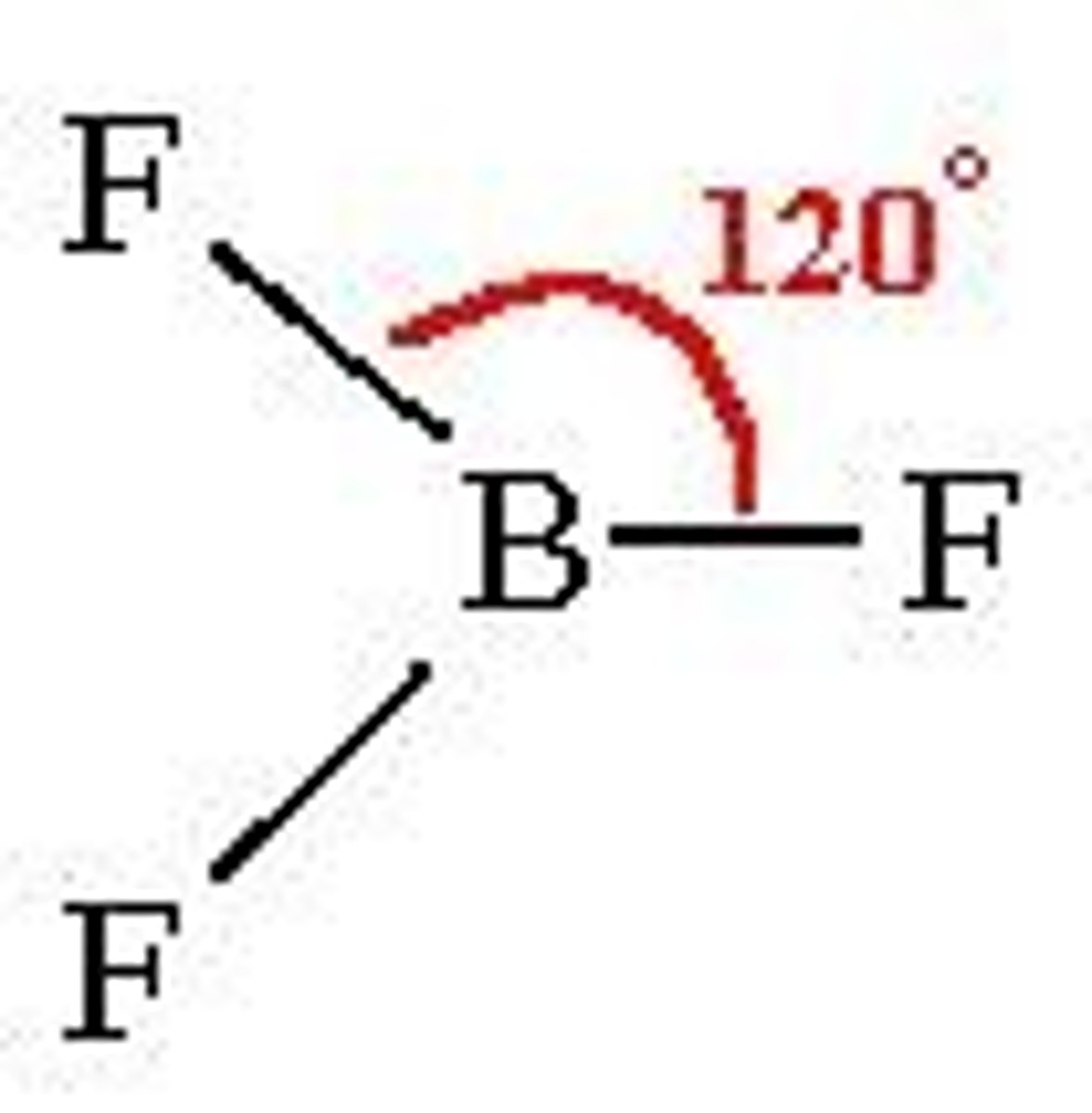
Trigonal planar shape
120 degrees
3 bonding pairs/regions
No lone pairs
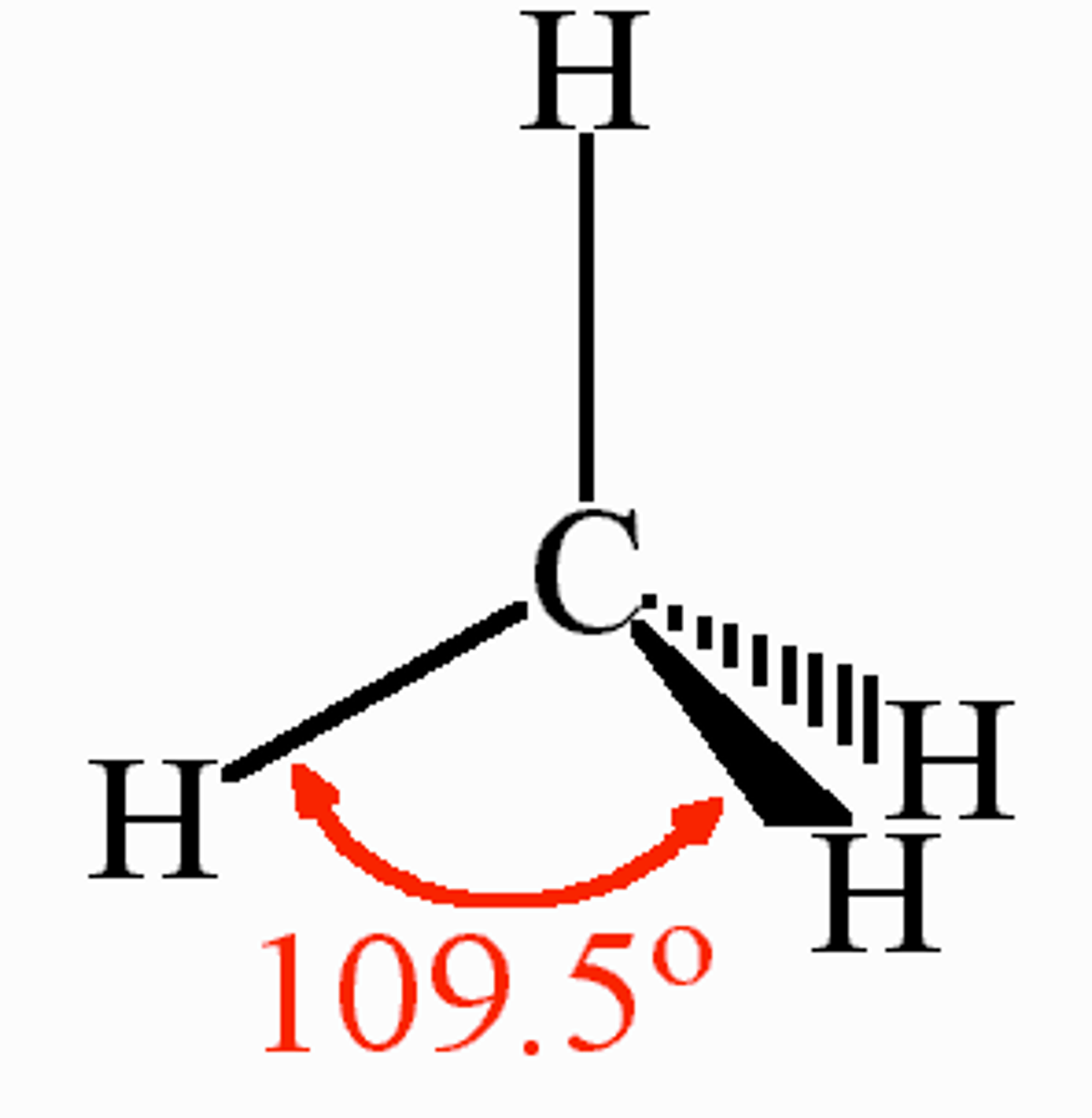
Tetrahedral shape
4 bonding regions/pairs
109.5 degrees
no lone pairs
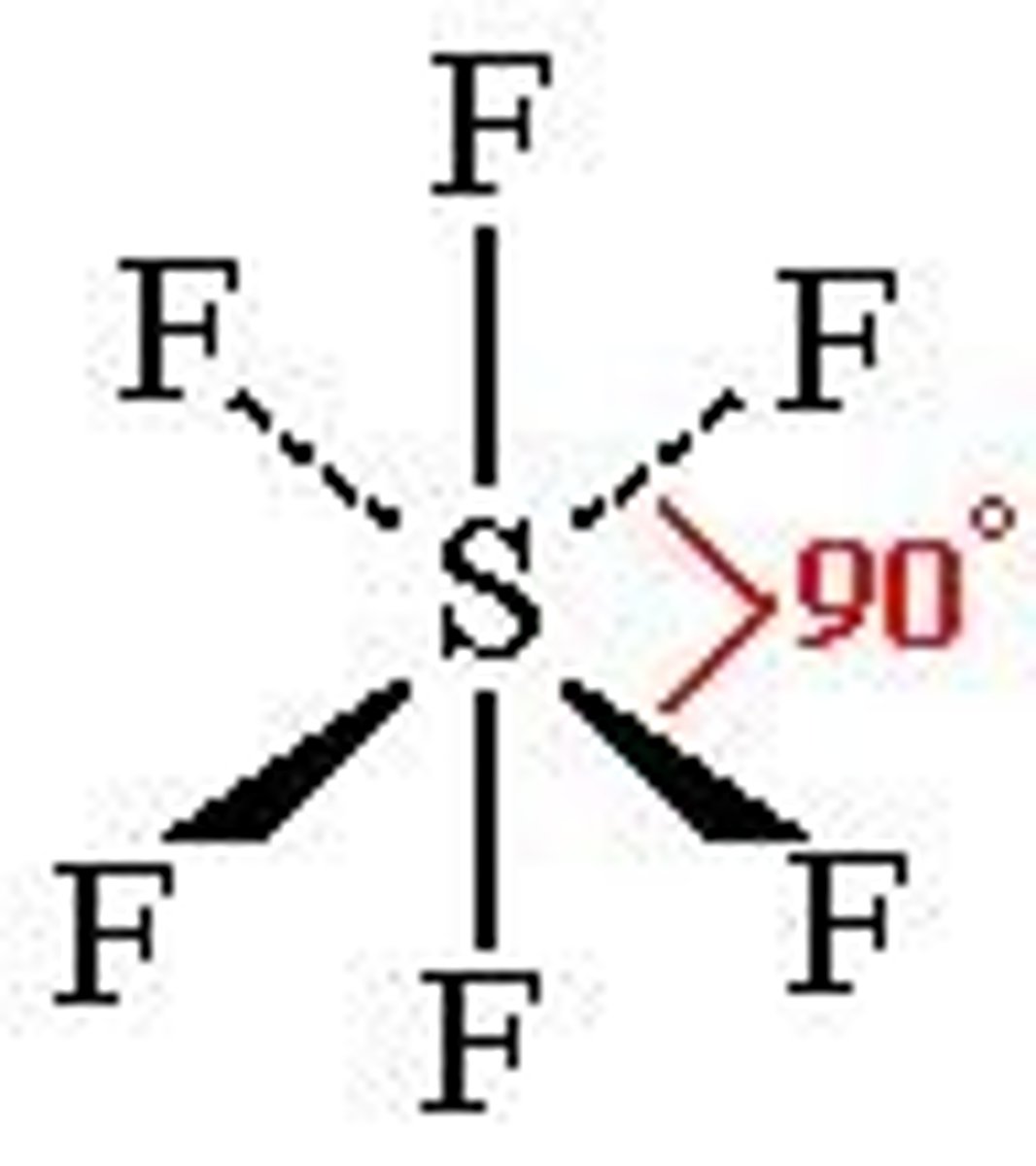
Octahedral shape
-6 bonding pairs/regions
- 90 degrees
No lone pairs
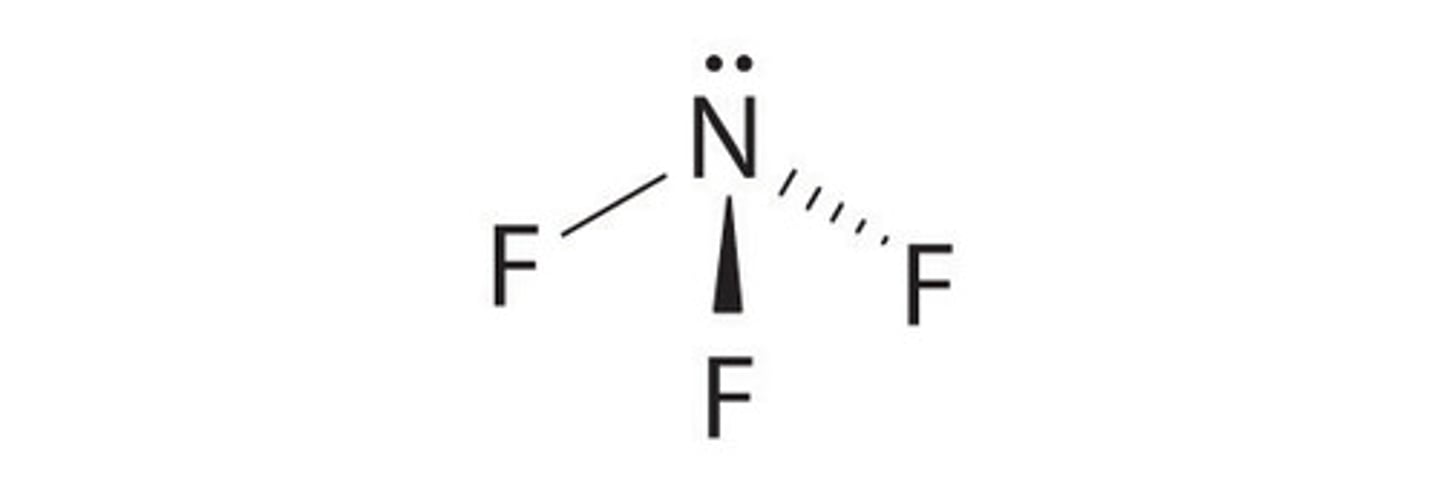
Pyramidal shape
3 bonding pairs/regions
107 degrees
1 lone pair
Non-linear shape
- 2 bonding regions/pairs
- 2 lone pairs on central atom
- 104.5 degrees