3. Food Packaging Interactions
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Migration
Monomers or additives moving into the food
Can cause off-flavor sand safety problems
Absorption
Aroma compounds, fat, organic acids and pigments moving into the polymer film
Can cause loss of aroma intensity, development of unbalanced flavor profile and damage to the package
Mechanism of migration
Migrant must diffuse through the polymer matrix
Partition across the polymer food interface
Diffuse through food
What accelerates migration?
Short-contact, high temperature situations (microwaving, pasteurization)
Factors influencing diffusivity on polymer side
Crystallinity - high crystallinity = low diffusion
High molecular weight = lower diffusion
Additive content
Higher thickness = lower diffusion
Polarity (polyester higher than polyolefinic)
Glass transition temperature (Tg)
Effect of Tg on migration
At room temperature:
>Tg: soft, elastic-rubber → high diffusion
<Tg: Stiff chains - glassy (e.g. PET, PC) → Low diffusion
Bisphenol A (BPA)
Used as a starting substance for polycarbonate (PC) plastics and epoxy resins
Toxic and harmful at very low concentration
PC plastic is used to manufacture refillable containers, but also in cans
Main concern was on baby bottles in microwave
When is BPA mobilized?
BPA from can epoxy coatings only mobilizes when the can is heated above the Tg of the resin (~105C)
Factors influencing migration on migrant side
Molecular size (smaller = faster)
Polarity (lipophilic migrants go into fatty foods much faster)
Volatility
Factors influencing migration on food side
Antioxidant additives can migrate intentionally (active packaging) or unintentionally
Butylhydroxytuluene (BHT) = a lipophilic compound used as an antioxidant food additive (E321)
Fatty foods extract lipophilic migrants far more efficiently than aqueous foods
This is why the EU introduces a “fat reduction factor” (FRF)
Key migrants
Glass packaging: silicates, heavy metals
Metal packaging: Tin dissolution from plain tinplate into tomato products is the classic example
Paper and board: benzophene (BP) photointiator migration from UV-cured inks through cardboard into food
Class of compounds that can migrate from plastic material to food
Residual monomer
Plasticizer
Antioxidants
Residual solvents
Low MW compounds
Decomposition products
Key migrants and their hazards in plastics (residual monomers)
Residual monomers!
Vinyl chloride monomer (VCM) from PVC
Correlation between VCM inhalation and lung cancer
Styrene monomer from PS: taste threshold in orange juice as low as 0.2-0.3 ppm
Key migrants and their hazards in plastics (Plasticizers)
Plasticizers are the second big category
Phthalates (from PVC) are endocrine distruptors
Plastic regulation laws
Migration measurement protocol
OML = overal migration limit (60 mg/kg food or 10 mg/dm2)
SML = specific migration limit (compound specific)
Picture shows specific food stimulants, e.g. acetic acid is for acidic foods

Why are simulants used (for plastic regulation laws) instead of real food?
Food is really complex, each food has a different composition
Simulants provide a standardization
EU legislation assumes that for packages with a capacity of 0.5-10L, 1 Kg of food is exposed to 6 dm2 surface area of packaging material
If this does not apply you have to use a correction factor
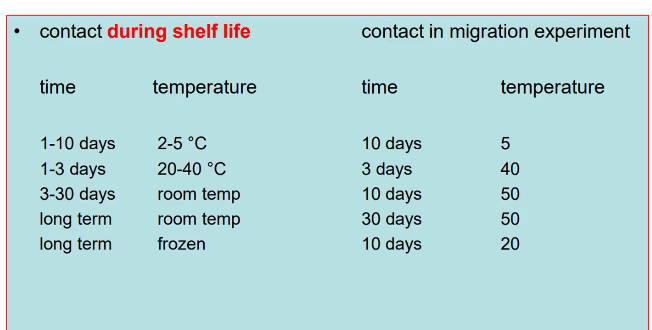
What conditions are standardized for migration experiments?
Contact during processing
Contact during shelf life (see picture)
Why does orange juice in high density poly ethylene taste different after storage?
From food to packaging
Adsorption or Scalping
Food components (mainly flavor compounds) are absorbed on packaging materials with consequent sever reduction of quality
occurs due to:
Partitioning between food and polymer
Sorption into amorphous polymer regions
What affects permeability of flavor components in scalping?
Mainly their polarity
Non-polar = better
Additionally the following factors also affect scalping
molecular size
polarity
polymer free volume