Alkenes
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Alkene definition
Unsaturated hydrocarbons containing a C=C double bond
General formula
CₙH₂ₙ
Bond type
One double bond (C=C) + single bonds
Unsaturated meaning
Can undergo addition reactions because of C=C
Homologous series
Alkenes form a homologous series with similar properties
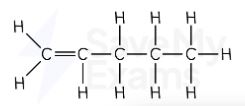
Ethene formula
C₂H₄
Propene formula
C₃H₆
Butene formula
C₄H₈
Boiling point trend
Increases with chain length
Flammability trend
Decreases with chain length
Reactivity
More reactive than alkanes due to C=C bond
Combustion
Burns with smoky flame due to incomplete combustion
Addition reaction definition
Atoms add across the double bond
Hydrogenation
Alkene + hydrogen → alkane (Ni catalyst, 150°C)
Halogenation
Alkene + bromine → dibromoalkane
Bromine water test
Decolourises from orange to colourless with alkenes
Hydration
Alkene + steam → alcohol (H₃PO₄ catalyst)
Polymerisation
Many alkene molecules join to form addition polymers
Example polymerisation
Ethene → poly(ethene)
Key idea
Alkenes are reactive due to the C=C bond and undergo addition reactions
ethene displayed formula
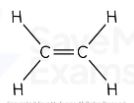
propene displayed formula
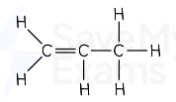
but-1-ene displayed formula
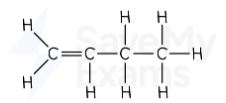
pent-1-ene displayed formula