orgo exam 2 (edited)
1/124
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
125 Terms
Alkyl halide
A compound where a halogen (F, Cl, Br, I) is bonded to an sp3 carbon
Alcohol
A compound with an —OH group attached to an alkyl carbon
Thiol
A compound with an —SH (sulfhydryl) group attached to carbon
Phenol
A compound where —OH is bonded to an aromatic ring
Enol
A compound where —OH is attached to a double-bonded carbon
Ether
A compound with an oxygen bonded to two carbon groups (R–O–R)
Sulfide (thioether)
A compound with sulfur bonded to two carbon groups (R–S–R)
Alpha carbon
The carbon bonded directly to a functional group like halogen or OH
Primary (1°) carbon
A carbon attached to one other carbon
Secondary (2°) carbon
A carbon attached to two other carbons
Tertiary (3°) carbon
A carbon attached to three other carbons
Common naming (alkyl halides)
Name alkyl group + halide (e.g., methyl chloride)
IUPAC naming (alkyl halides)
Halogens treated as substituents (fluoro-, chloro-, bromo-, iodo-)
Common naming (alcohols)
Name alkyl group + "alcohol"
Principal functional group
The group that determines the suffix in IUPAC naming
Principal chain
The longest chain containing the principal functional group
Alkoxy group
An ether substituent (RO–)
Alkylthio group
A sulfide substituent (RS–)
Diol
A compound with two OH groups
Triol
A compound with three OH groups
Heterocyclic compound
A ring containing atoms other than carbon
Epoxide
A three-membered cyclic ether (oxirane)
sp3 hybridization
Tetrahedral geometry (~109.5° bond angle)
Noncovalent interactions
Attractions between molecules that do not involve covalent bonds
Intermolecular attraction
Attractive force between different molecules
Boiling point
Temperature where vapor pressure equals atmospheric pressure
Van der Waals forces
Weak attractions from temporary dipoles (dispersion forces)
Dispersion forces
Attraction from induced dipoles in molecules
Polarizability
How easily an electron cloud can be distorted
Dipole-dipole interactions
Attractions between molecules with permanent dipoles
Hydrogen bonding
Attraction between H bonded to O/N/F and lone pair on another atom
Hydrogen bond donor
Molecule providing hydrogen (O–H, N–H)
Hydrogen bond acceptor
Molecule with lone pairs that accepts hydrogen bond
Boiling point trend
Increases with molecular size and surface area
Branching effect
More branching lowers boiling point
Alcohol boiling points
High due to hydrogen bonding
Melting point
Temperature where solid becomes liquid
Symmetry effect
Higher symmetry → higher melting point
Solute
Substance being dissolved
Solvent
Substance doing the dissolving
Solution
Homogeneous mixture of solute and solvent
Entropy of mixing
Increase in disorder when substances mix
Free energy of solution (ΔGs)
Determines if solution formation is favorable
Favorable solution
ΔGs < 0
Unfavorable solution
ΔGs > 0
Protic solvent
Solvent that can donate hydrogen bonds (water, alcohols)
Aprotic solvent
Solvent that cannot donate hydrogen bonds
Polar solvent
Solvent with large dipole moment
Nonpolar solvent
Solvent with little or no dipole moment
Dielectric constant
Measure of solvent’s ability to separate charges
"Like dissolves like" rule
Polar dissolves polar; nonpolar dissolves nonpolar
Miscible liquids
Liquids that mix in all proportions
Hydrophobic interaction
Nonpolar molecules cluster together in water
Solvation shell
Layer of solvent molecules surrounding solute
Ion pair
Two oppositely charged ions associated in solution
Dissociated ions
Ions fully separated and surrounded by solvent
Charge-dipole interaction
Attraction between ion and polar molecule
Xenobiotic
Foreign substance in a biological system
Amphipathic molecule
Molecule with both polar and nonpolar regions
Phospholipid
Molecule with polar head and nonpolar tails forming membranes
Phospholipid bilayer
Double layer forming cell membranes
Hydrophilic
Water-attracting (polar)
Hydrophobic
Water-repelling (nonpolar)
Ionophore
Molecule that binds and transports ions
Crown ether
Cyclic compound that binds metal cations
Ion channel
Protein that allows ions to pass through membranes
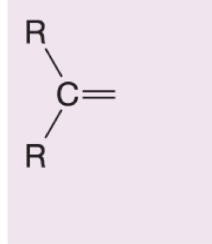
What happens when DMS is added to this alkene during ozonolysis?
Becomes a ketone
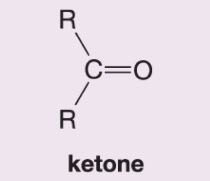
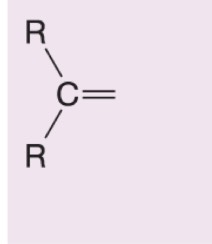
What happens when hydrogen peroxide and water (H2O2/H2O) are added to this alkene during ozonolysis?
Also becomes a ketone
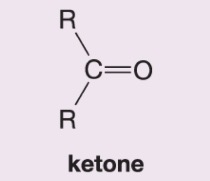
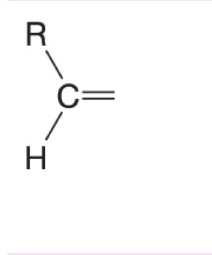
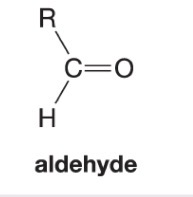
What happens when DMS is added to this alkene during ozonolysis?
Forms an aldehyde
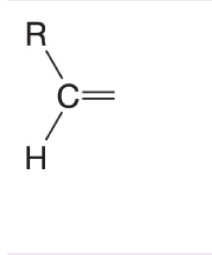
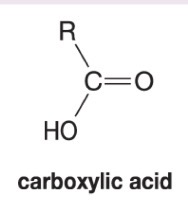
What happens to when hydrogen peroxide and water (H2O2/H2O) are added to this alkene during ozonolysis?
Forms carboxylic acid
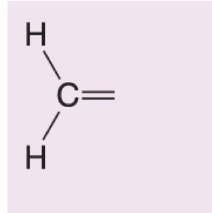
What happens when DMS is added to this alkene during ozonolysis?
Forms formaldehyde
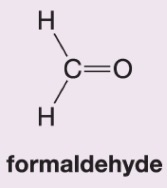
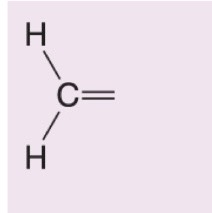
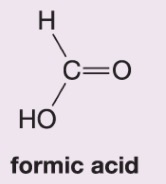
What happens when hydrogen peroxide and water (H2O2/H2O) are added to this alkene during ozonolysis?
Forms formic acid
What happens during HBr, HCl and HI addition to an alkene?
Halogen goes to the more substituted C
What happens during acid-catalyzed hydration to an alkene?
OH goes to more substituted C
Addition of Cl or Br and OH-OH does what to an alkene?
Goes to the more substituted C if it has two alkyl substituents
Oxymercuration-reduction to an alkene
No rearrangements; adds H and OH; OH goes to the more substituted C
Hydroboration-oxidation to an alkene
Adds H and OH; Anti-markovnikov (H goes to the MORE substituted C while OH goes to the LESS substituted C)
Ozonolysis in an alkene
Cycloaddition to double bond forms ozonide (C-C double bond is broken)
Catalytic hydrogenation to an alkene
adds H2
HBr, HCl, and HI addition to an alkyne
can occur once or twice to give vinylic halide or geminal dihalide
Hg2+ catalyzed hydration in an alkyne?
Adds H and OH; OH goes to the more substituted C; resulting enols convert to ketones
Hydroboration-oxidation in alkynes
Adds H and OH; Anti-Markovnikov (OH is added to the LESS substituted C); resulting enols convert to aldehydes/ketones
Catalytic hydrogenation to an alkyne
H added to each C if poisoned catalyst (Lindlar’s) is used; two H’s to each if no poison present
Achiral
congruent mirror images
Chiral
enantiomers and noncongruent mirror images
Asymmetric carbon
A carbon that has 4 different groups attaches. CHIRAL DOES NOT MEAN ASYMMETRIC CARBON!
What happens if a sample rotates a plane of polarized light clockwise?
Sample is considered dextrorotatory (+) [not related to R/S]
What happens if a sample rotates a plane of polarized light counterclockwise?
Sample is considered levorotatory (-) [not related to R/S]
What can be said about a substance that rotates a plane of polarized light?
The substance is optically active and chiral
Biot’s law
a = [a]cl (optical rotation = specific rotation*concentration*path length)
Racemic mixture
Type of enantiomer mixture that s 50:50; 0 specific rotation; NOT optically active; NOT ACHIRAL
Enantiomeric ratio (ER)
moles of major enantiomer/moles of minor enantiomer
Enantiomeric Excess (EE)
% of the major enantiomer - % of the minor enantiomer
Specific rotation [a] of mixture used to calculate EE
100% x ([a] mixture/[a] pure)
Stereochemical correlation example
Ozonolysis does not break any bonds (absolute configuration is retained); therefore the absolute configuration can be assumed because they have the same relative positions
Diastereomers
Molecule has two or more asymmetric carbons; not mirror images; differ in all physical properties; do not have identical physical properties
Isomers
Have the same molecular formula
Constitutional isomers
different atomic connectivities
Stereoisomers
have identical atomic connectivities
enantiomer properties
noncongruent mirror images