DAT Organic Chemistry - Mike's Videos
1/346
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
347 Terms
2 electron domains: geometry, bond angle, hybridization
Linear
180
sp
3 electron domains: geometry, bond angle, hybridization
Trigonal planar
120
sp2
4 electron domains: geometry, bond angle, hybridization
Tetrahedral
109.5
sp3
Condensed formula
Can be written in a single line of print
# of bonds: single bond
one sigma
# of bonds: double bond
one sigma + one pi
# of bonds: triple bond
one sigma + two pi
Pi bonds are formed by which orbitals?
2 p-orbitals
How would one best describe the orbital required to form a σ bond? Select all that apply.
I. Overlap of two s-orbitals
II. Overlap of one s- and one p-orbital
III. Overlap of two p-orbitals
IV. Parallel overlap of two p-orbitals
I, II, III
For resonance structures, only ________________ move
Electrons (pi electrons, lone-pair electrons, or negative charges)
For resonance structures, you can move electrons toward/into an atom that _____________________
Does not have a full octet
If an atom already has a full octet, then you can move electrons into it only if _____________________
You push electrons out the opposite side
For resonance structures, do not move or break _________________
Sigma bonds
Major resonance contributor rules
Most stable resonance structure will have:
1. A full octet on every atom
2. The smallest possible number of charges
3. Negative charges on the most EN atoms and positive charges on the least EN atoms
Allyl carbocation
Carbocation that is one position away from a double bond
Allyl carbanion
Carbanion that is one position away from a double bond
Order of stability in Newman Projections from most to least
Staggered > Gauche > Eclipsed
Gauche interaction
Occur whenever 2 substituents larger than hydrogen are adjacent to each other in a staggered conformation
Most stable cycloalkane
Cyclohexane
Which is more stable: axial or equatorial? Why
Equatorial
1, 3-diaxial interactions
How to achieve the greatest stability in cyclohexane rings
Placing the largest substituents in the equatorial positions
Trans vs. cis cyclohexanes
Trans: 2 substituents going in opposite directions
Cis: 2 substituents going in same direction
Bronsted acid
Substance that donates H+ ions (protons)
Bronsted base
Substance that accepts H+ ions
Lewis acid
Substance that accepts electrons
Lewsi base
Substance that donates electrons
Conjugate acid =
Base + an H + 1 charge
The stronger/more reactive the acid, the _________________ its conjugate base
Weaker/more stable
The stronger/more reactive the base, the _________________ its conjugate acid
Weaker/more stable
Increase Ka = ________________ pKa = ________________ acid strength
Decrease
Increase
pKa of H2O
16
pKa of H3O+
-2
pKa of H3CCOOH
5
pKa of NH4+
10
pKa of H3CCOCH3
20
pKa of C6H5OH
10
Increase Kb = _________________ pKb = _________________ base strength
Decrease
Increase
Mnemonic for ranking acids and bases
Charge
Atom
Resonance
Dipole Induction
Orbital hybridization
The more positively-charged a compound is = the more ________________
Acidic
The more negatively-charged a compound is = the more ________________
Basic
H's acidity increases as the atom that it's bonded to
1. Goes left to right across a row on the periodic table (increasing EN)
2. Goes down a column on the PT (increasing size)
A resonance-stabilized conjugate base will be a _______________ acid
Stronger
Electron withdrawing groups ________________ acidity
Increase
Electron donating groups ________________ acidity
Decrease
Electron-withdrawing appendages (not involving aromatic rings) include
EN atoms or groups (F, Cl, Br, I, N, or O)
Electron donors (not involving aromatic rings) are generally
H or C groups
Electron withdrawers (bonded to aromatic rings) include
Halogen atoms
Groups with double/triple bonds
Electron donors (bonded to aromatic rings)
An atom with at least one lone pair e-
Atoms bonded by single bonds
An alkyl or aryl group
Withdrawing groups __________________ basicity because ___________________
Decrease
They make basic electrons less available
Donating groups __________________ basicity because ____________________
Increase
They make basic electrons more available
S-orbitals tend to be _____________ EN than P-orbitals
More
The more "s-character" an atom has, the ________________ the acid
Stronger
Order the following from least to most acidic: sp3, sp2, sp
sp3 < sp2 < sp
IUPAC names for simple hydrocarbon parent chains 1-10
Methane
Ethane
Propane
Butane
Pentane
Hexane
Heptane
Octane
Nonane
Decane
Isopropyl
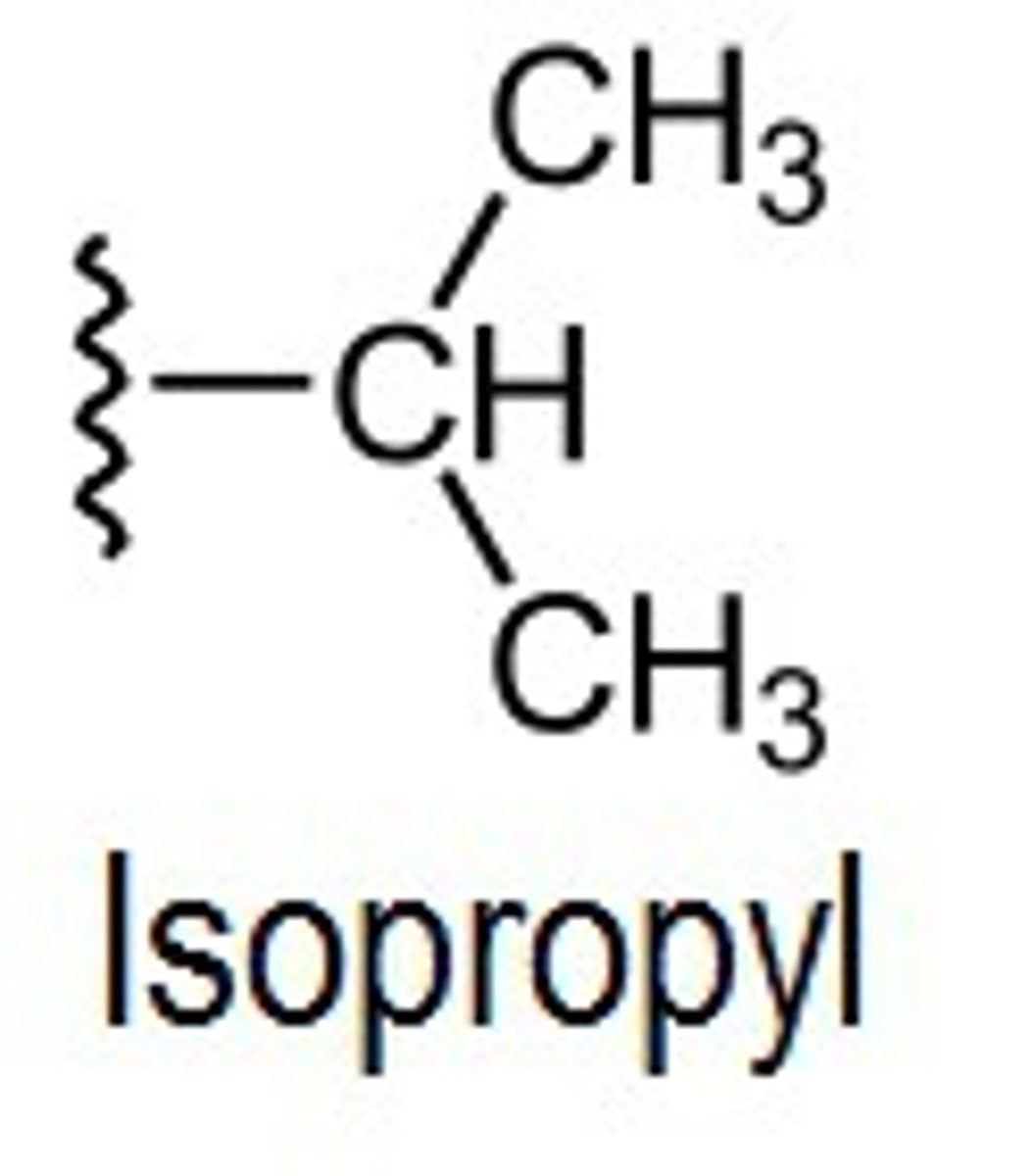
Sec-butyl
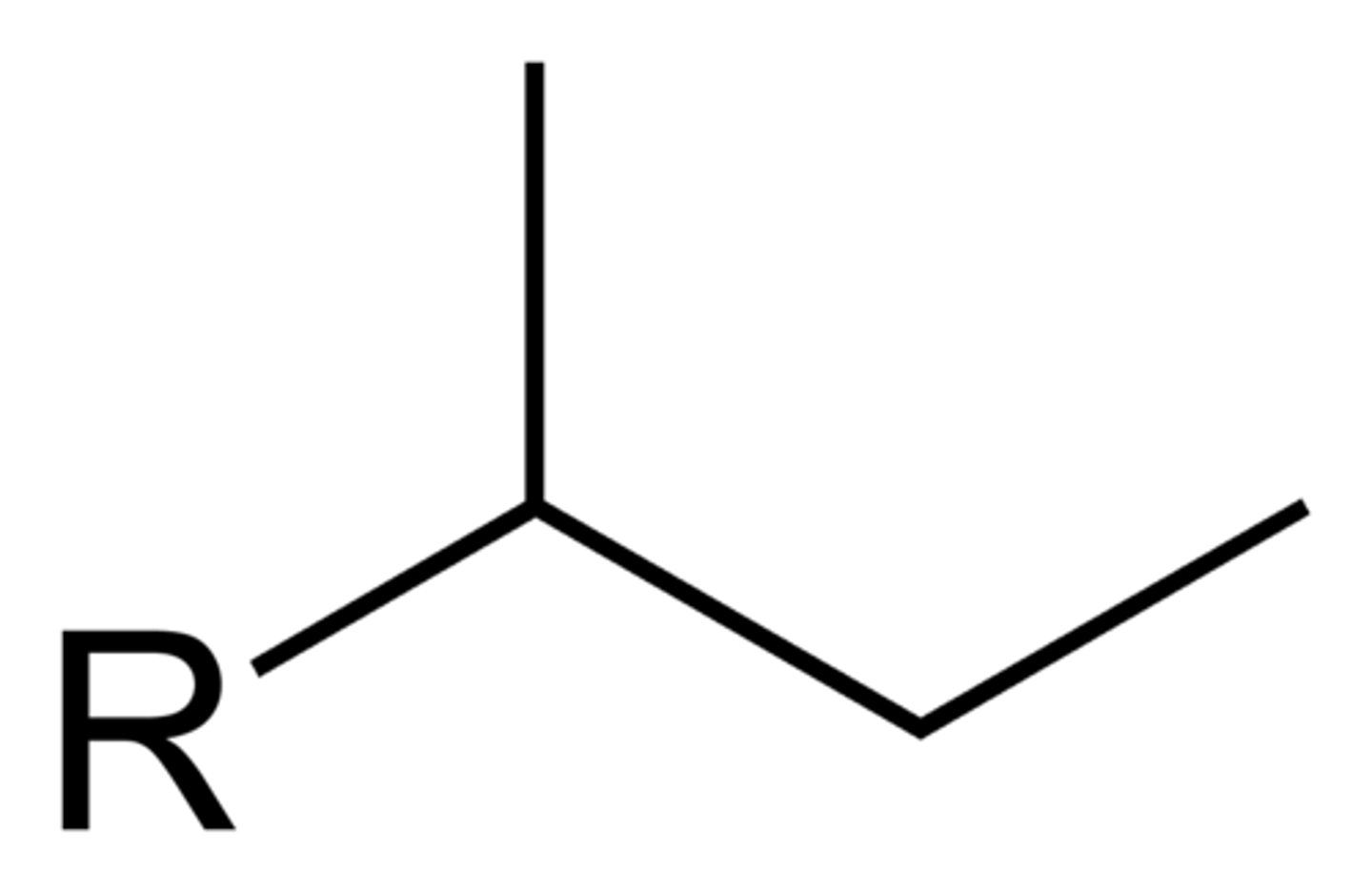
Isobutyl
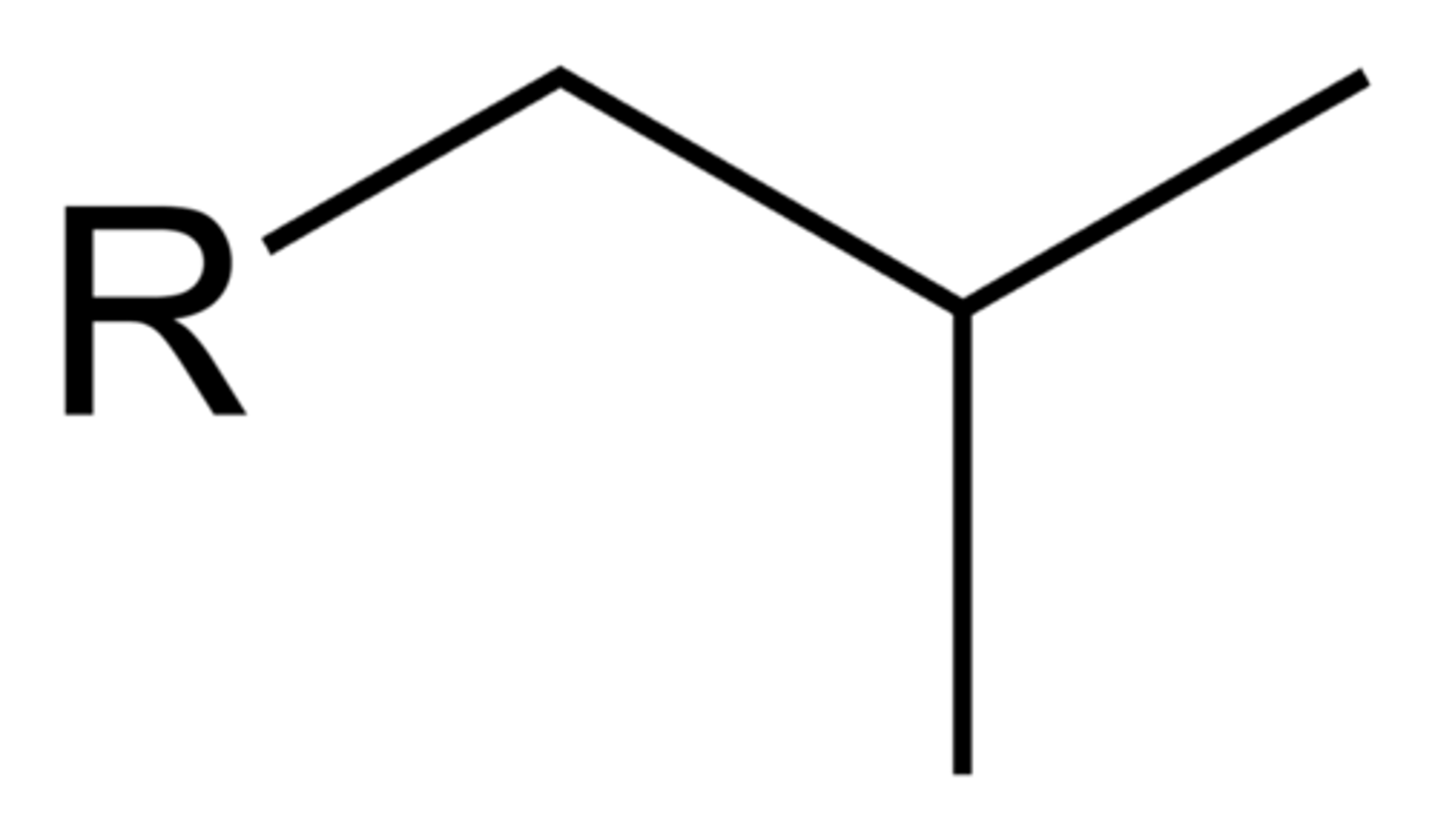
tert-butyl
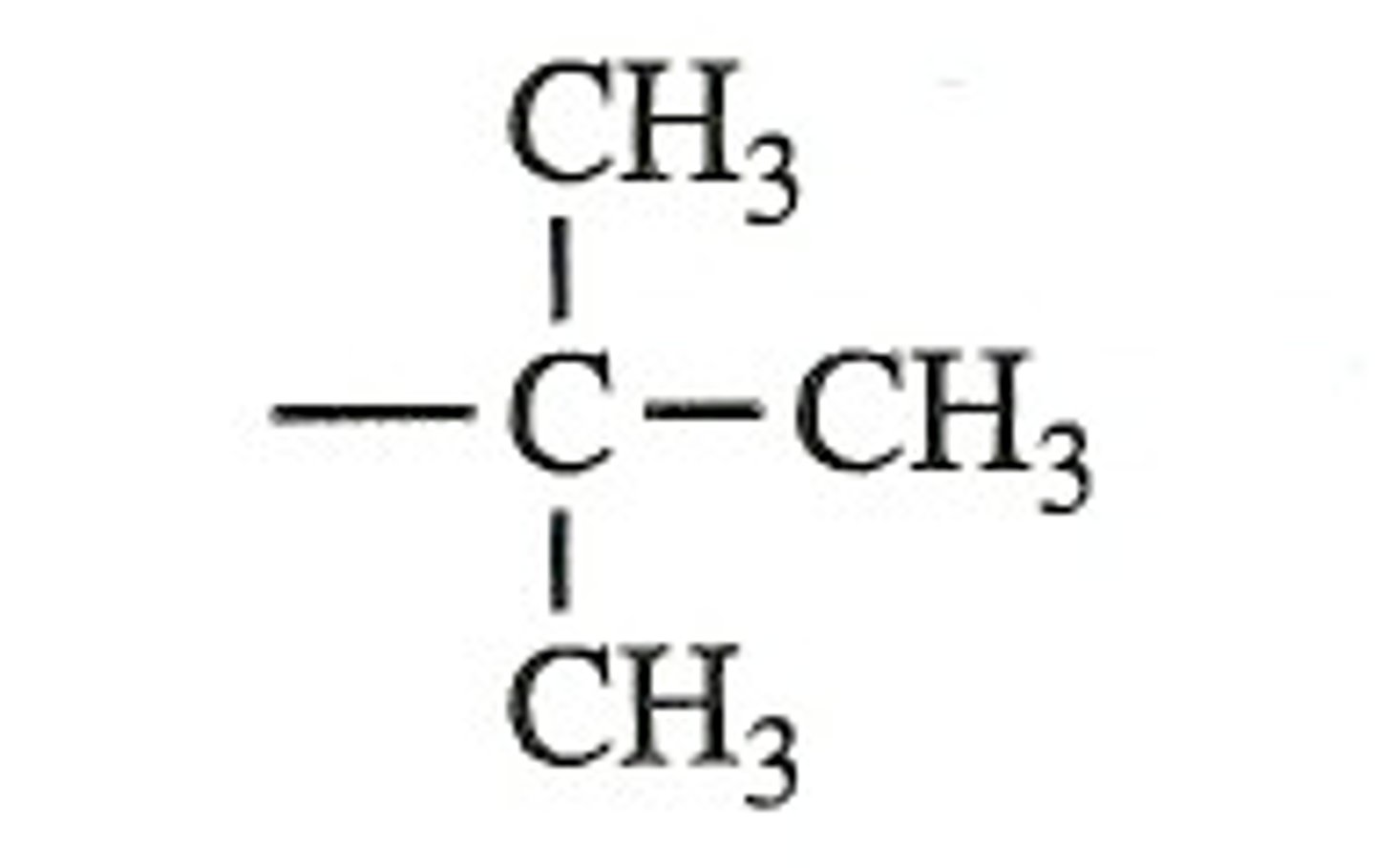
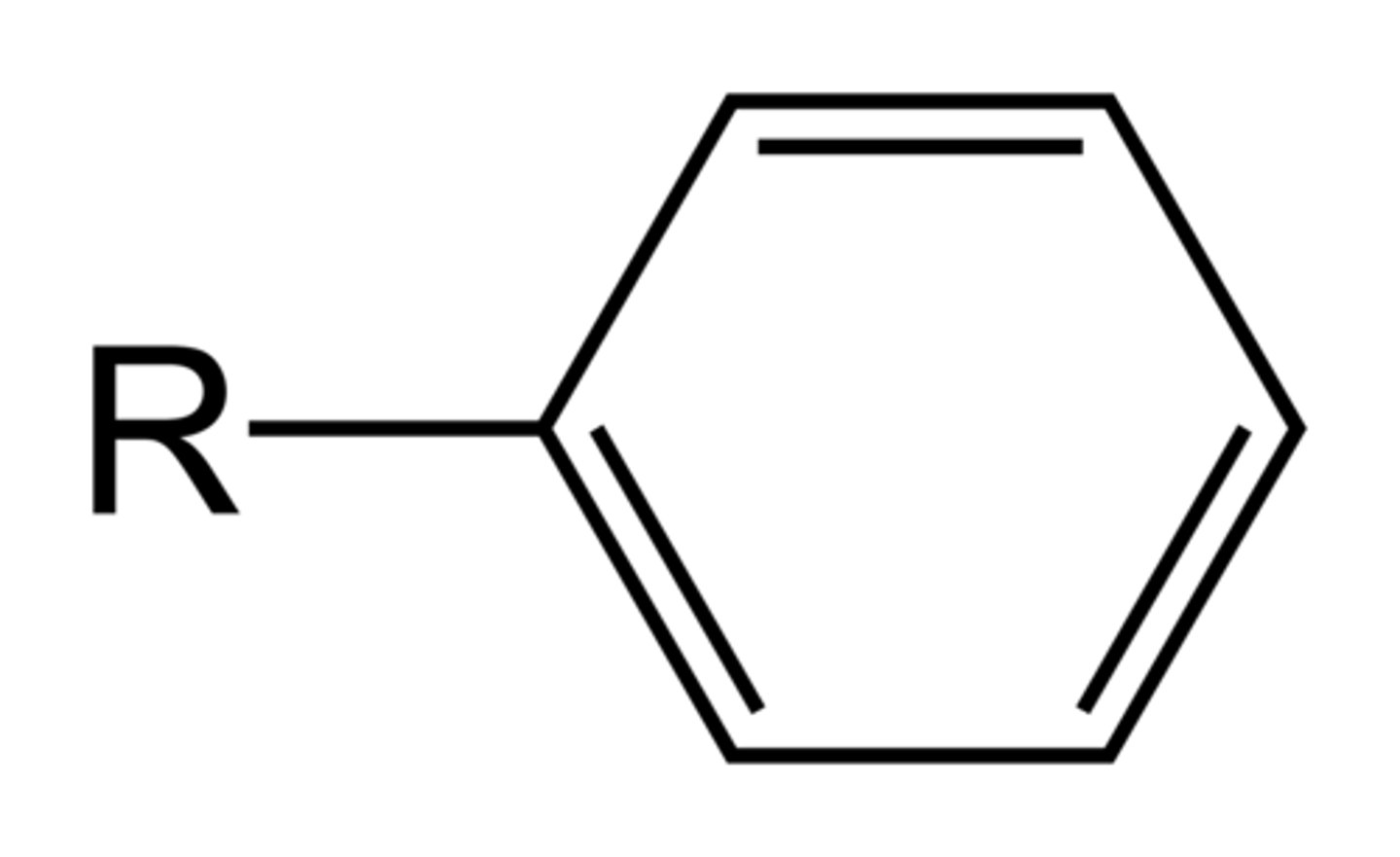
Phenyl
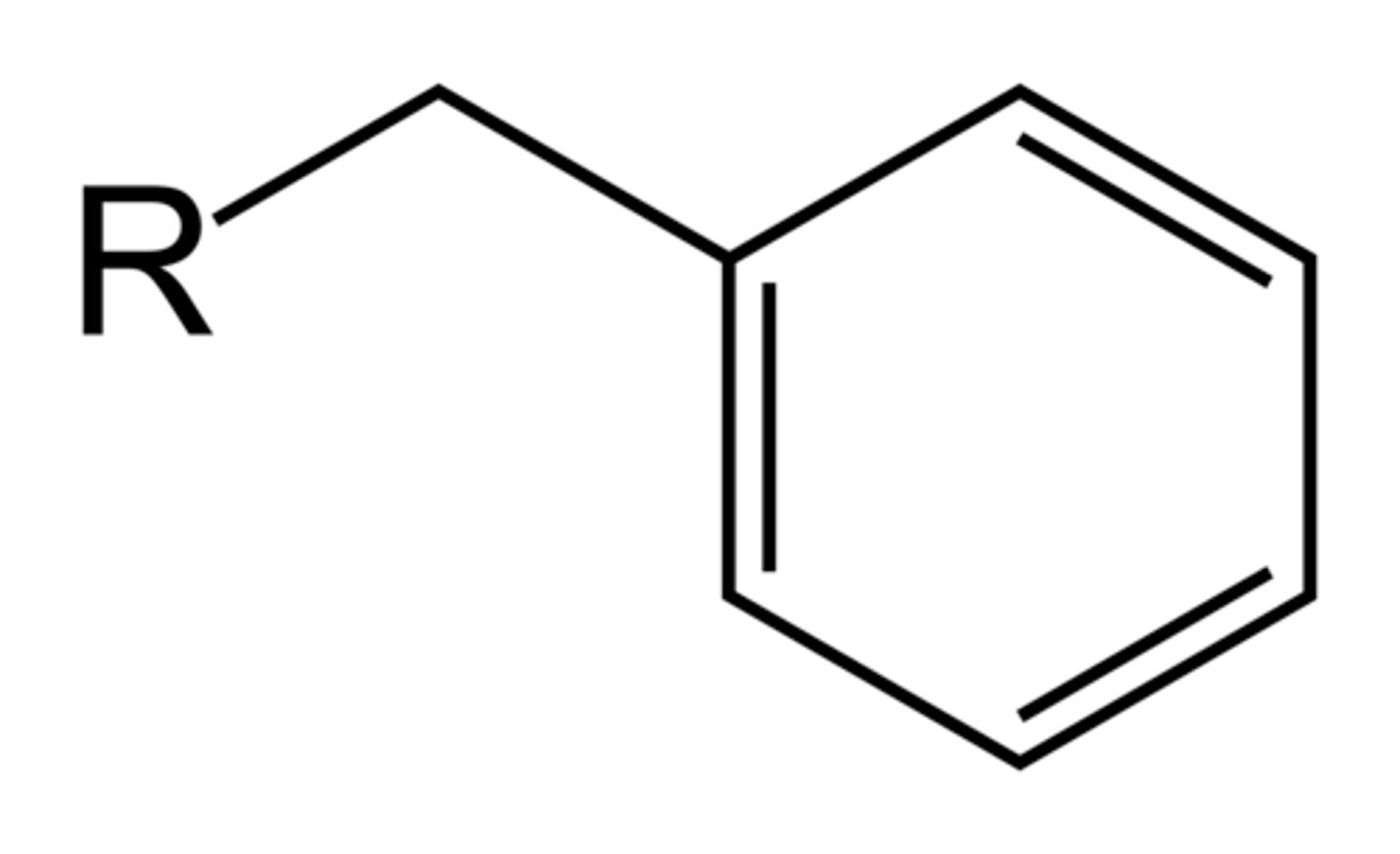
Benzyl
Steps for naming alkanes
1. Find parent chain (longest C chain) with the smallest substituent number at the first point of difference
2. Count number of C atoms in the parent chain
3. Identify and number the substituents
4. Write name as a single word with substituents in order
-Substituents (replace "ane" with "yl") + parent chain
When there are 2 substituents on a cyclic molecule, direction is indicated by
Cis: same side
Trans: opposite side
If there are more than 2 substituents, their direction is indicated by
Stereochemical configurations (R/S)
Is the cyclic portion of an alkane the parent chain or the noncyclic portion?
Cyclic
Naming alkyl halides
Can follow the same rules for naming alkanes, using prefixes "fluoro-", "bromo-", "chloro-", "iodo-"
OR can use suffixes "-yl fluoride", etc.
Naming alkenes
1. Use suffix "-ene"
2. Number the double bound (LOWEST possible)
3. Add prefixes "cis" or "trans" if there is one H on each of the 2 alkene C=C carbons
OR
3. Use E/Z naming system (E: opposite sides, Z: same sides)
Determining priority for alkenes
1. Highest atomic number = highest priority
2. If there is a tie, move to adjacent carbons until the tie is broken
3. Multiple-bonded atoms are counted as the same number of single-bonded atoms
Naming alkynes
1. Use suffix "yne"
2. Add number at the start of the triple bond
NO cis/trans E/Z because the bond is linear
Naming alcohols
1. Parent chain is the longest chain with the hydroxyl group
2. Give the smallest number to the C bonded to the hydroxyl group (higher priority than cycloalkanes, amines, alkenes, ethers, and alkyl halides)
4. Change suffix "-e" to "-ol"
Naming ethers
1. Name the 2 alkyl groups as substituents with "ether" at the end
OR
2. Consider the longest carbon chain to be the parent chain and the alkoxy group to be a substituent
Naming primary amines
Add suffix "amine" to the name of the organic substituent
Naming symmetrical and secondary amines
Add "di-" or "tri-" to the alkyl group
Naming asymmetrical amines
Name as N-substituted primary amines, with the largest alkyl group as the parent chain
Naming aldehydes
1. Number parent chain in direction that gives highest priority to the aldehyde carbon
2. Replace "-e" with "-al"
Naming ketones
1. Name parent chain with priority given to the ketone carbon
2. Replace "-e" with "-one"
3. Carbonyl carbon in a cyclic ketone is assumed to be the #1 carbon
Naming carboxylic acids
1. Number parent chain with the carboxylic group at #1 C
2. Replace "-ane" for "-oic acid"
Naming acid halids
1. Number parent chain with the acid halide at #1 C
2. Replace "-ane" for "-oyl halide"
Naming esters
1. Alkyl group attached to the ester oxygen is listed first with the suffix "-yl"
2. Parent chain follows and the suffix is replaced with "-oate"
Naming amides
1. Name alkyl groups attached to the N as "N-methyl/ethyl/etc."
2. Parent chain starts at the carbonyl carbon and is counted moving away from the amide
Naming acid anhydrides
1. Determine length of the chain on either side of the bridging oxygen
2. List both lengths alphabetically, replacing suffix "-e" with "-oic"
3. Write "anhydride" at the end
Naming nitriles
1. Parent chain is the longest C chain that involves the nitrile C
2. Number parent chain in direction that gives the smallest number (always 1) to the nitrile C
3. Add the suffix "nitrile" to the parent name
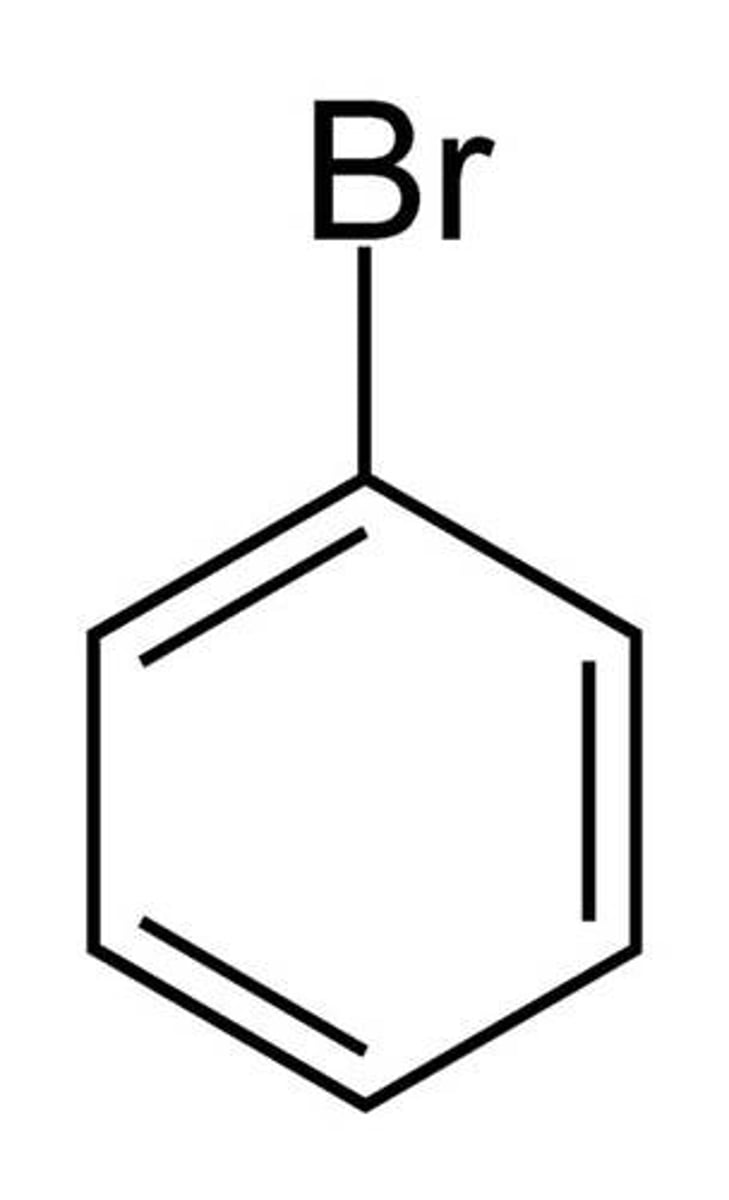
Bromobenzene
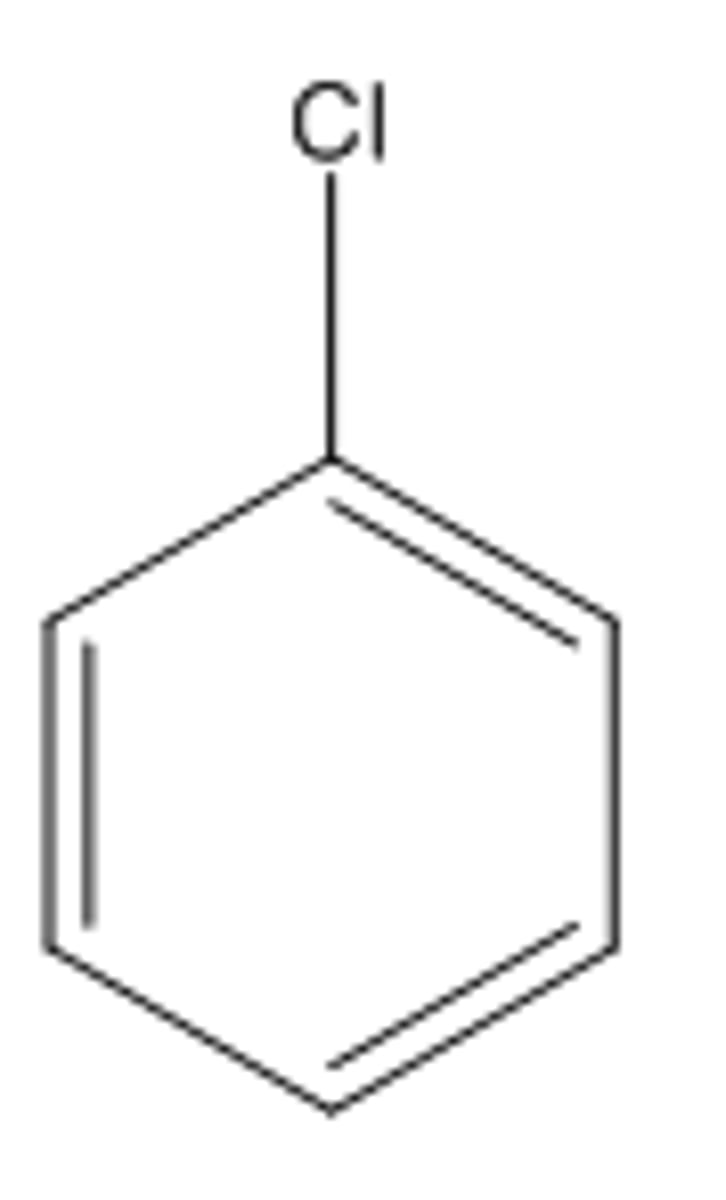
Chlorobenzene
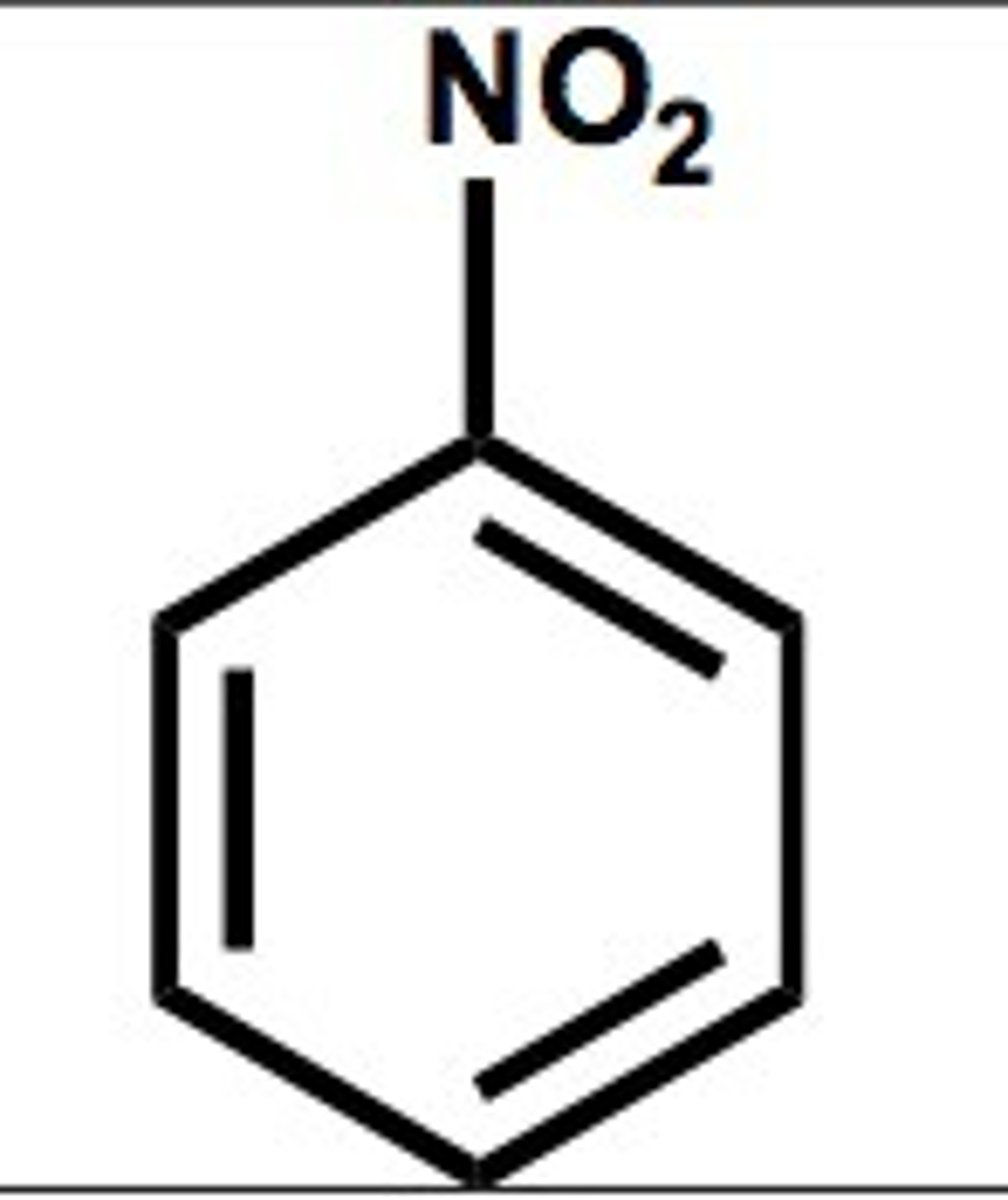
Nitrobenzene
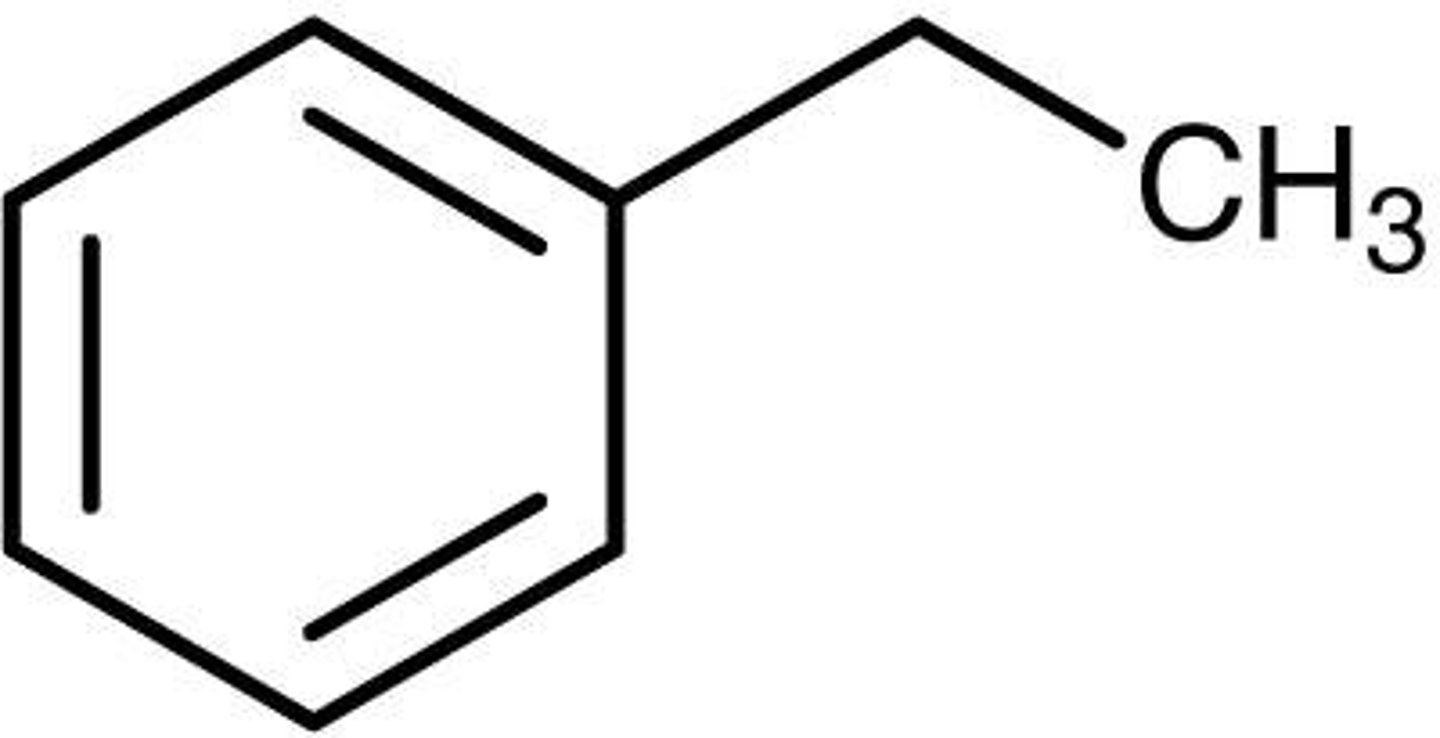
Ethylbenzene
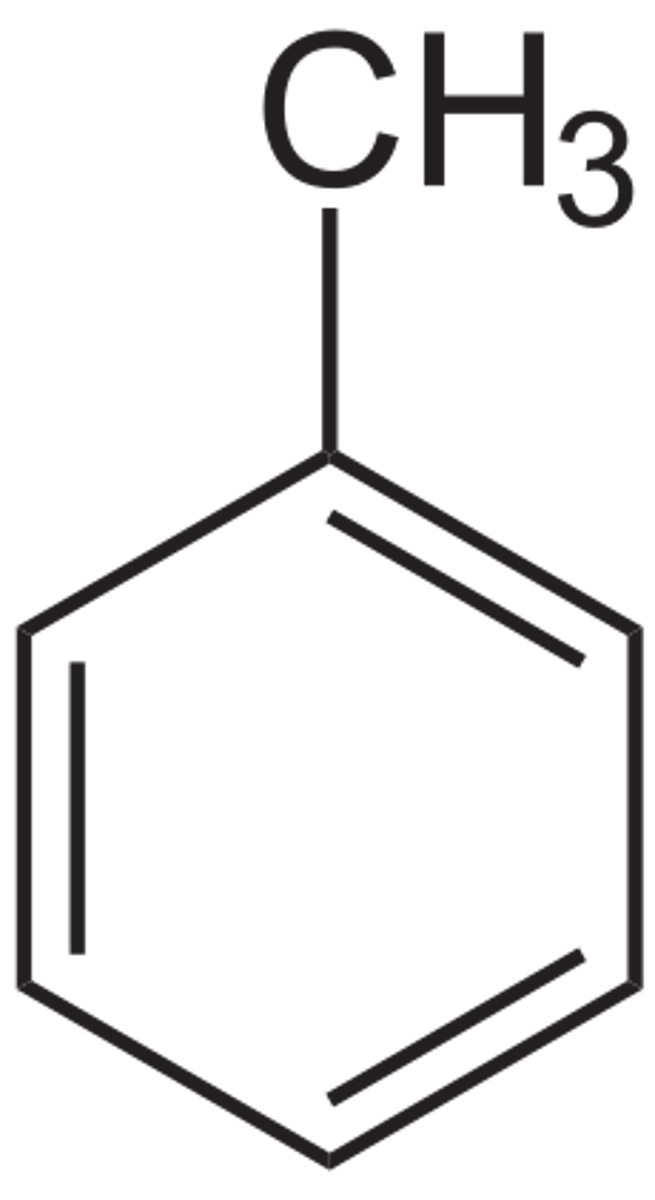
Toulene
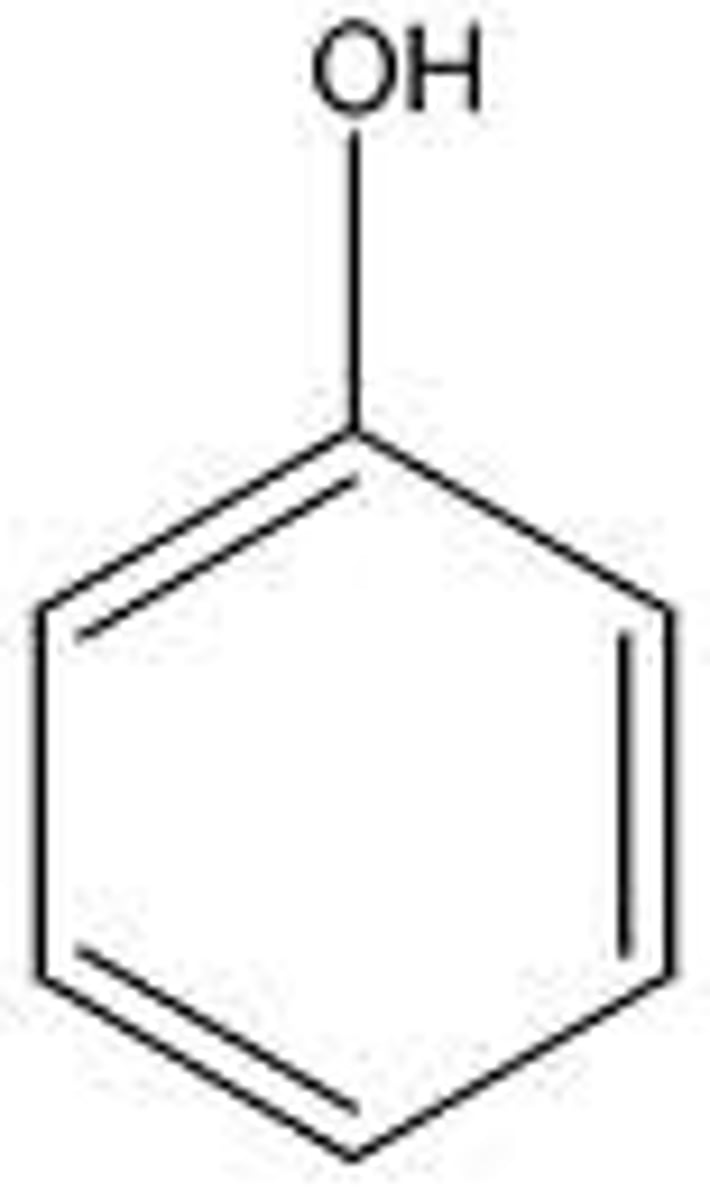
Phenol
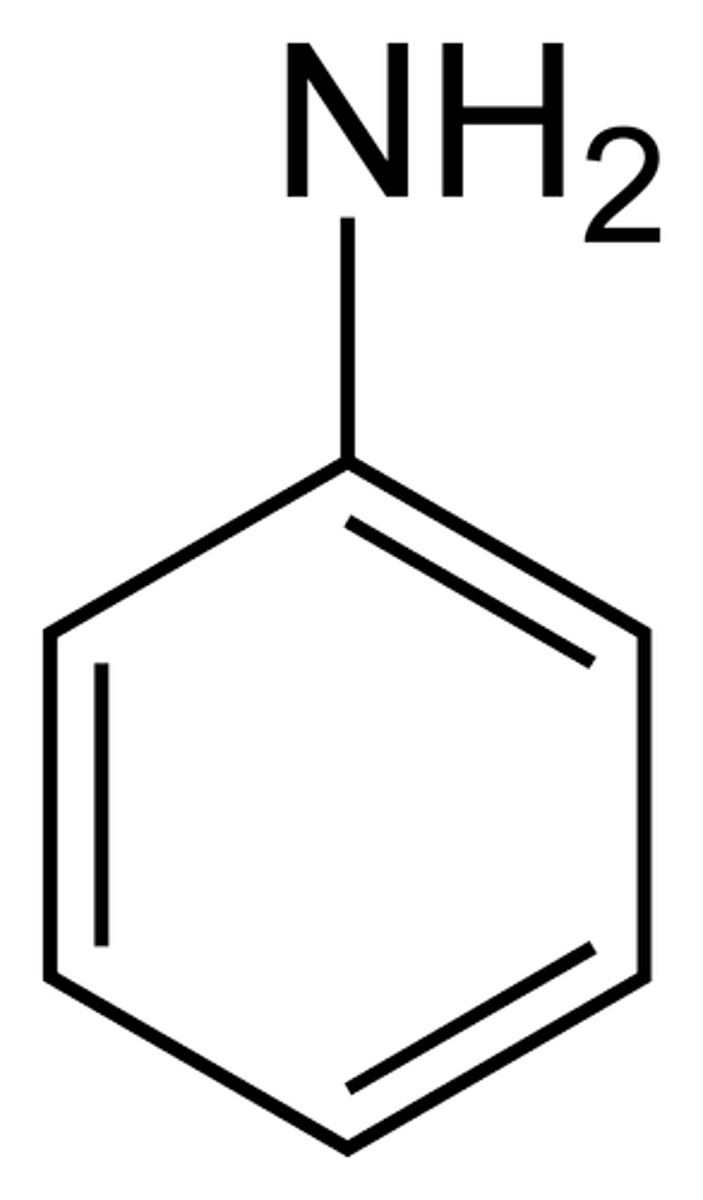
Aniline
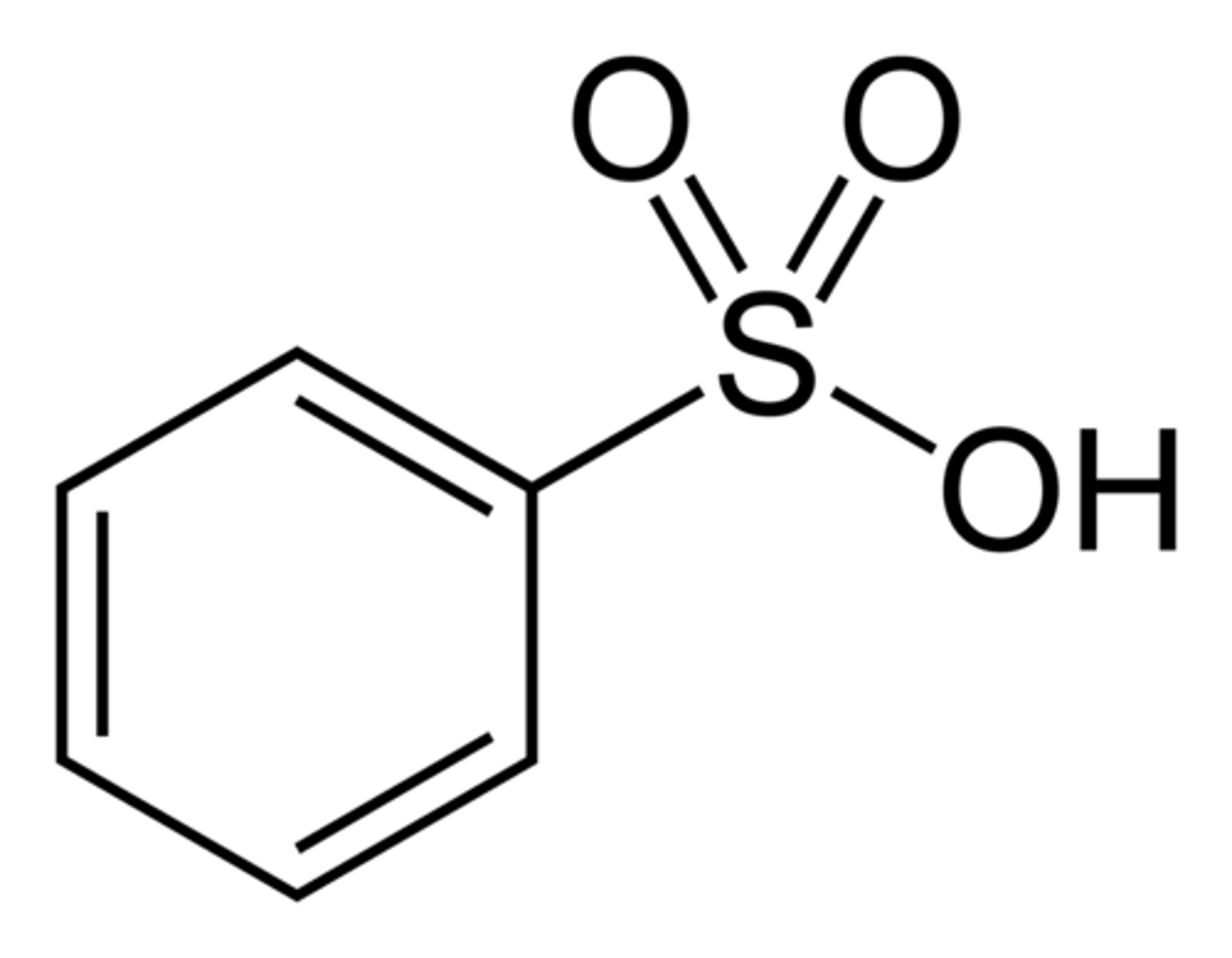
Benzenesulfonic acid
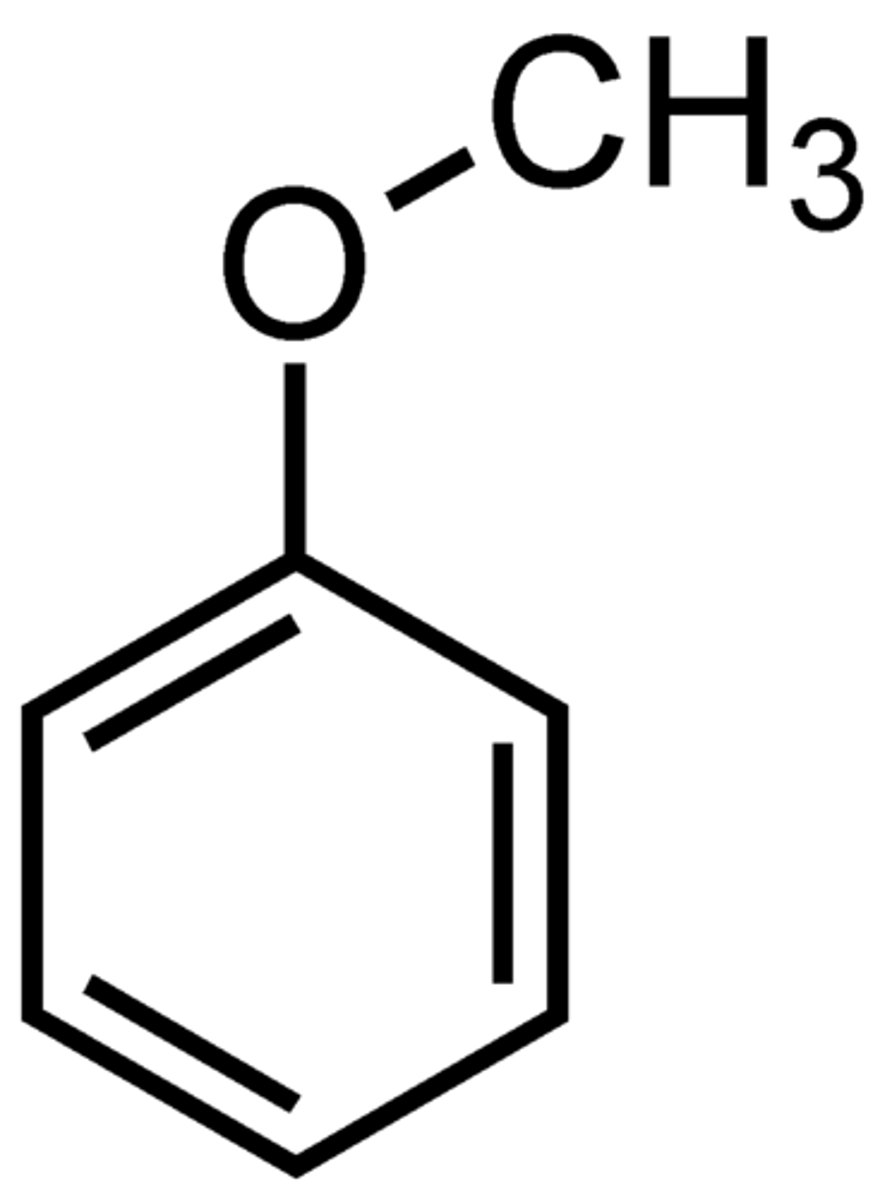
Anisole
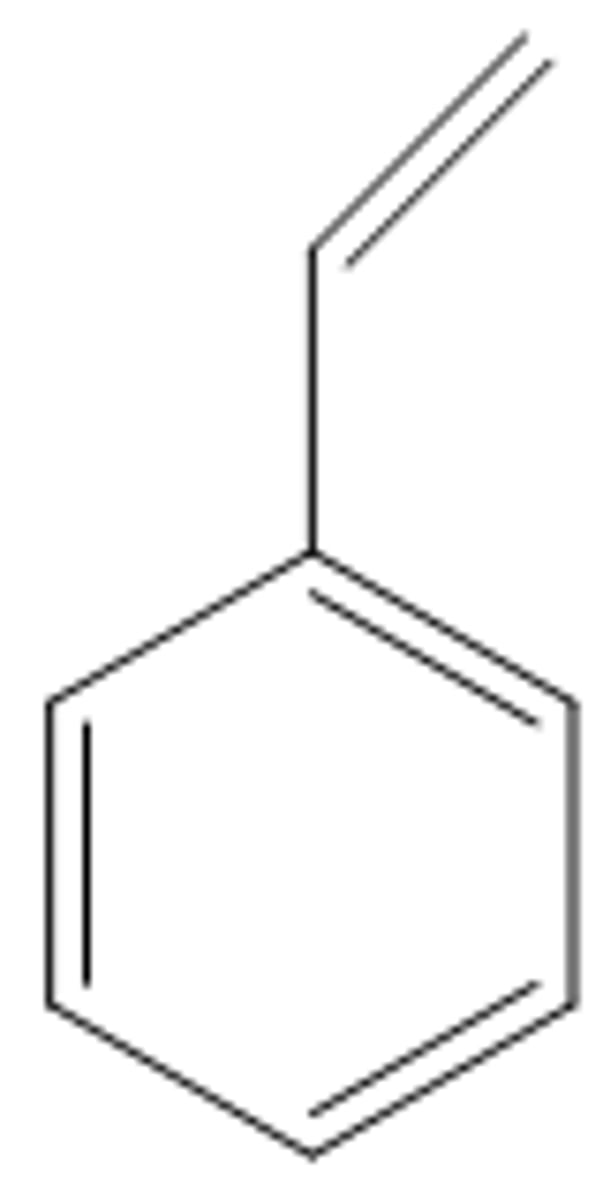
Styrene
Benzaldehyde
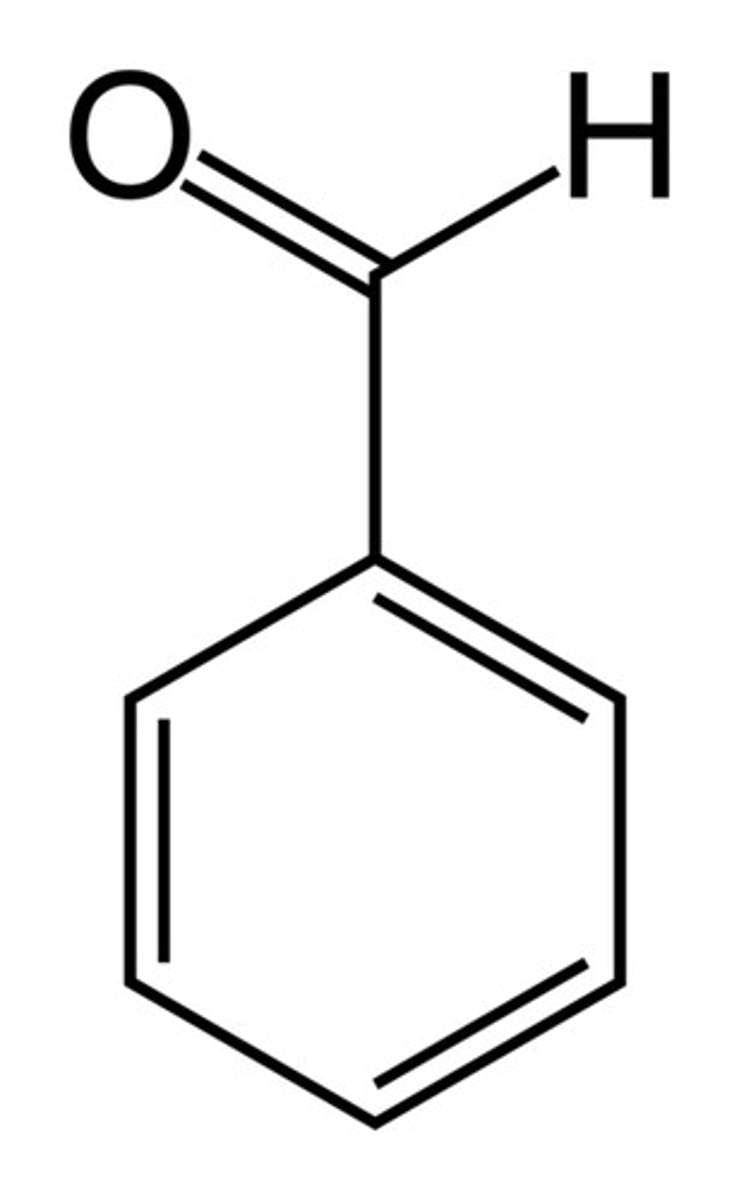
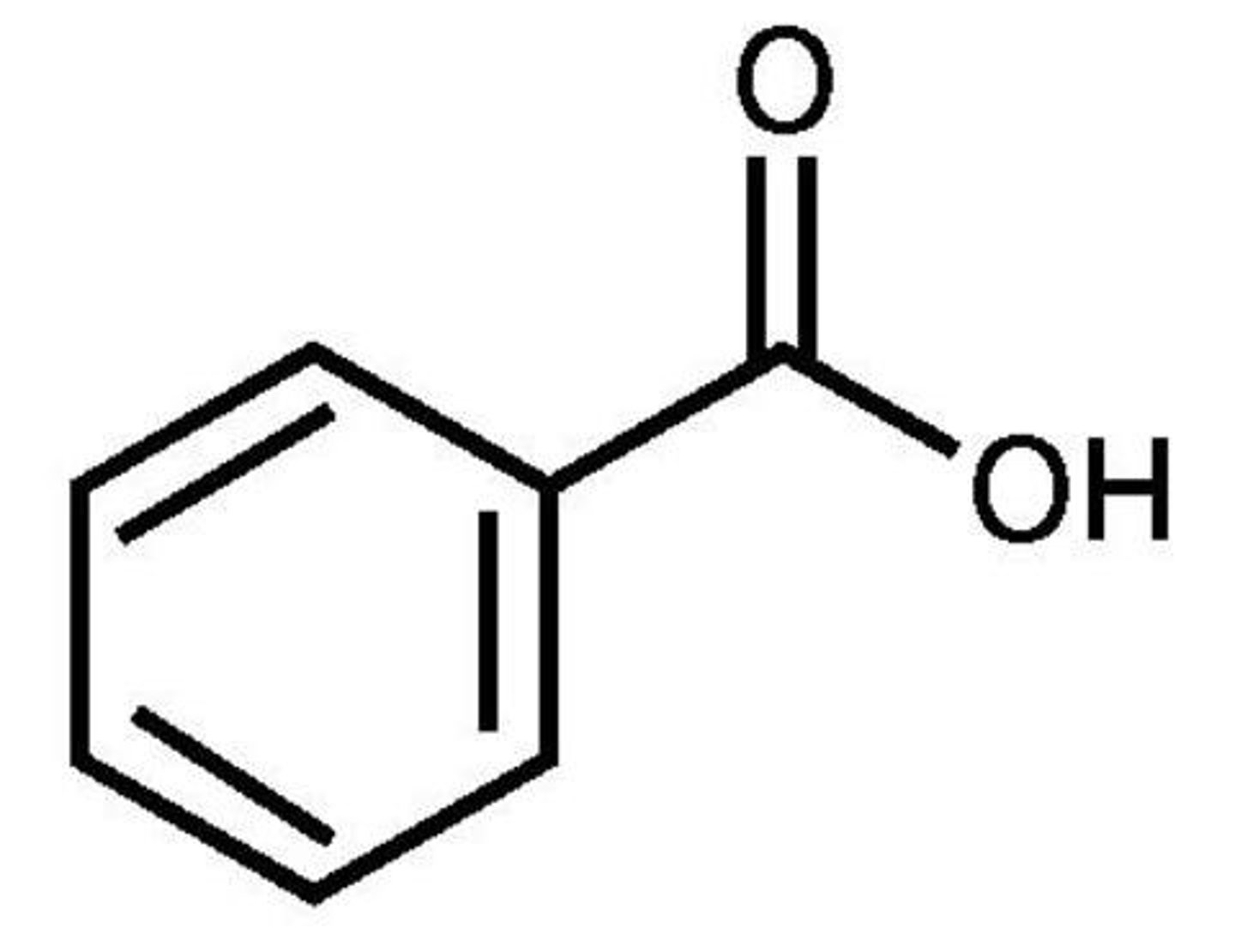
Benzoic acid
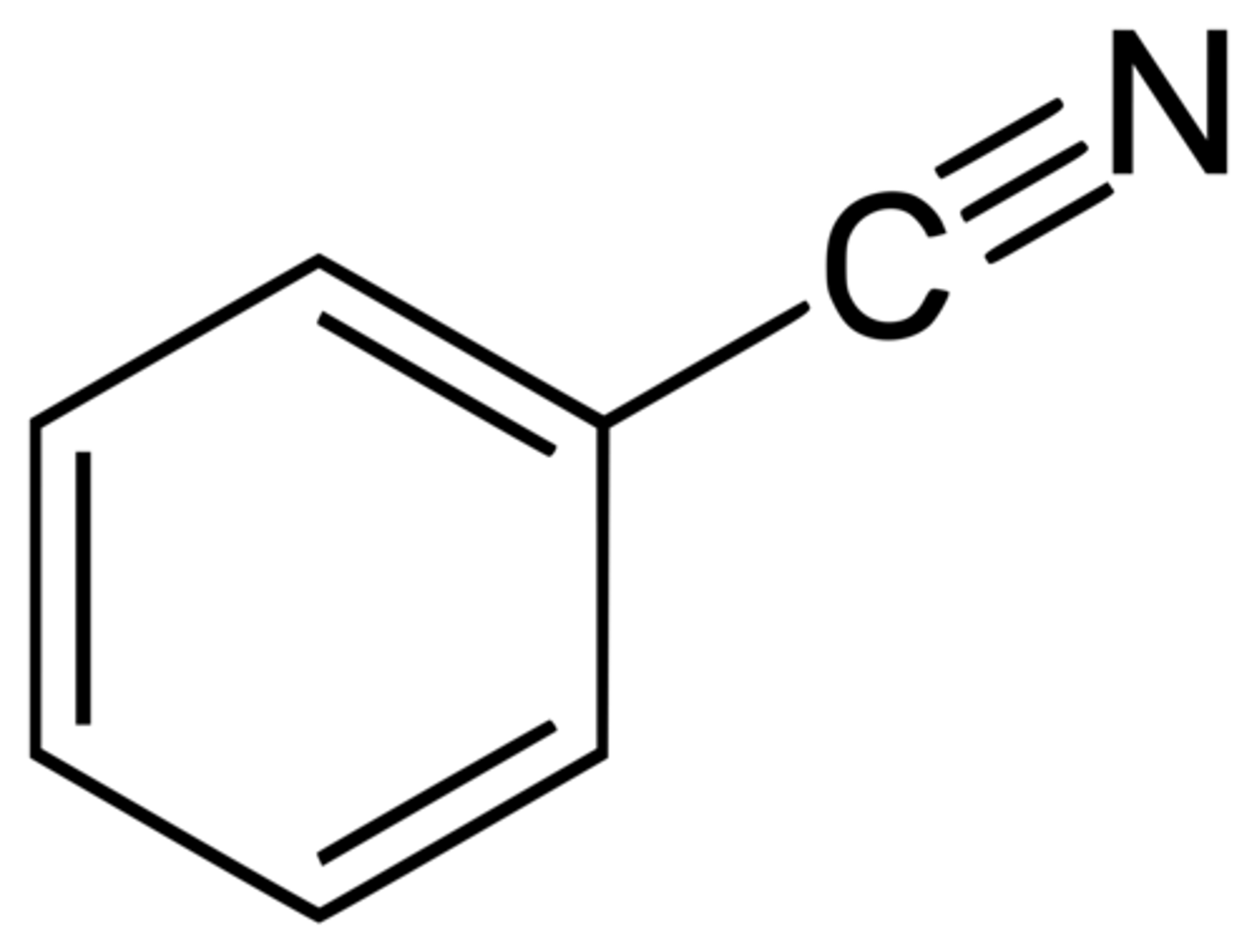
Benzonitrile
Naming substituted benzenes
1. Identify the parent chain with the highest-priority functional group
2. C atom that is attached to the priority functional group is #1
3. Number around the ring clockwise/counter-clockwise to obtain the lowest number at the first point of difference
Naming spiro alkanes
1. Count TOTAL number of carbons across the entire molecule (parent name)
2. Count number of carbons to the left + right of the spiro-carbon center (a and b, from lowest to highest)
3. Write the final name as spiro[a.b]parent name
Naming bicyclic alkanes
1. Count TOTAL number of carbons across the entire molecule (parent number)
2. Count number of carbons to the left/right/above the bridgehead carbons (written as a, b, and c from highest to lowest)
3. Write final name as bicyclo[a.b.c]parentname
Spiro vs. bicyclic alkane
Spiro: fused at a single C atom
Bicyclic: fused across a bridge
Enantiomers
Same chemical formula, same bonding connections
Non-superimposable mirror-image molecules
Diastereomers
Same chemical formula, same bonding connections
NOT non-superimposable mirror-image molecules