Biochem exam 2 flashcards
1/334
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
335 Terms
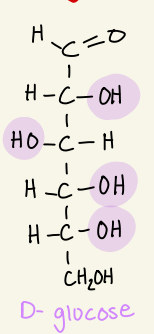
Draw the fischer projection of D- glucose
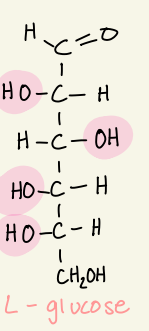
Draw the fischer projection of L- glucose
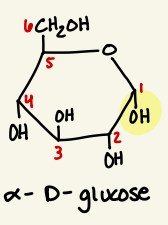
Draw alpha d glucose
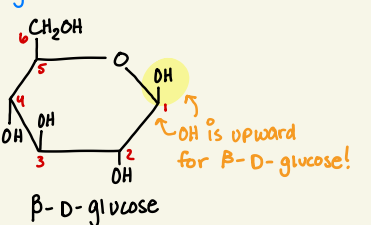
draw beta d glucose
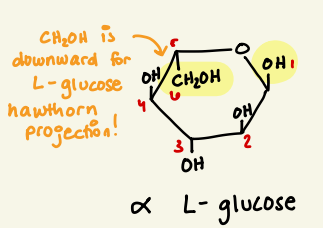
draw alpha L glucose
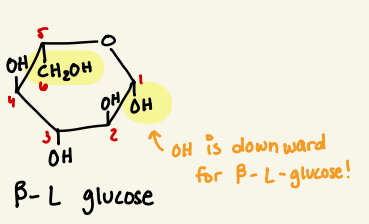
draw beta L glucose
Catalyst Affect the______ of a chemical reaction
Energy of activation
Enzymes can increase the rate constant of the chemical reaction by what?
A) Lowering the energy of the transition state
B) stabilizing the intermediate state
C) Promoting the catalytic event
T or F: An increase in temp can result in an increased reaction rate.
True
T or F: Lowering the free energy of the transition state can increase a reaction rate.
True
T or F: The free energy barrier in a chemical reaction must be overcome in order for products to form
True
T or F: Enzymes can selectively increase the rate constant either for the forward or the reverse direction of a chemical reaction.
False why?
Enzymes lower the activation energy for both the forward and reverse reactions equally, not just one direction. They speed up how fast equilibrium is reached, but they do not change which side is favored
Two enzymes, A and B, can catalyze the same reaction. For enzyme A Kcat=0.3s^-1, and for enzyme B Kcat=0.15s^-1. Which statement is TRUE.
A) Enzyme B is twice as fast as enzyme A in catalyzing the reaction.
B) We cannot use kcat to compare enzymatic activity without knowing the Km.
C) By adding enzyme A instead of B to the reaction we will obtain more product molecules faster.
D) The fastest reaction rate would be obtained by mixing 1 portion of A with 2 portions of B, instead of using 3 portions of enzyme A only.
Correct Answer: C) By adding enzyme A instead of B to the reaction we will obtain more product molecules faster.
*kcat tells how many substrate molecules one enzyme molecule converts into product per second when fully saturated.
A is wrong because it says B is faster, but A has the higher kcat.
B is wrong because kcat alone can compare turnover speed; Km is only needed when comparing catalytic efficiency (kcat/Km).
D is wrong because nothing proves that mixing enzymes beats simply using the faster enzyme A in equal total enzyme amount.
The lock and key model of substrate binding and enzymatic catalysis explains
Substrate specificity
Understand this question.
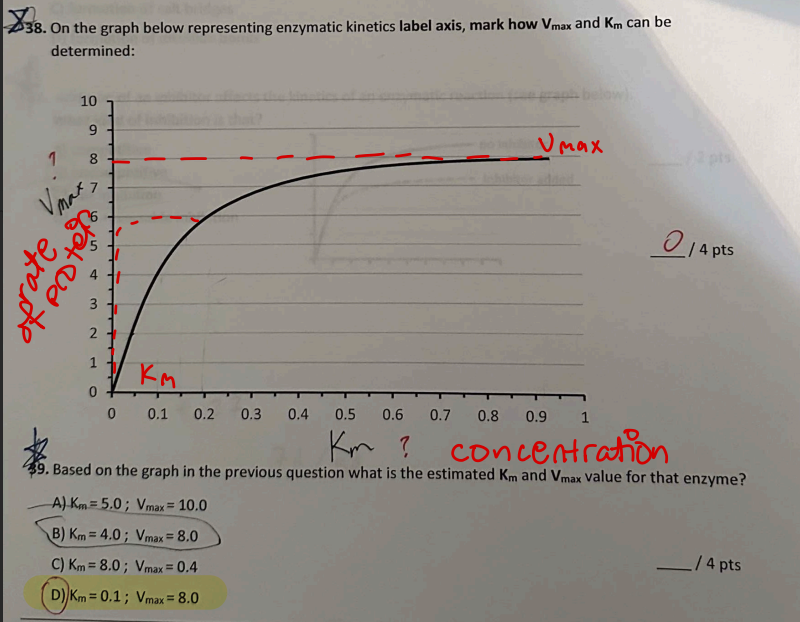
An inhibitor is added and the graph shows Vmax decreases while Km also decreases. What type of inhibition is this?
Uncompetitive inhibition
Reason: inhibitor binds only to ES complex, lowering both Km and Vmax.
An inhibitor is added and the graph shows same Vmax but higher Km. What inhibition is this?
Competitive inhibition
Reason: substrate can outcompete inhibitor at high concentration.
An inhibitor is added and the graph shows lower Vmax but same Km. What inhibition is this?
Noncompetitive inhibition
inhibitor binds enzyme regardless of substrate.
An inhibitor same Vmax, but Km increases. What inhibition is this?
Mixed inhibition
Which inhibition occurs when inhibitor binds the active site and competes with substrate?
Competitive inhibition
Which inhibitor binds only after substrate is already attached?
Uncompetitive inhibition
Which inhibitor binds both free enzyme and ES complex?
Mixed inhibition
Which inhibition can be overcome by adding more substrate?
Competitive inhibition
Which inhibition cannot be overcome by adding substrate because enzyme activity itself is reduced?
Noncompetitive inhibition
If both Km and Vmax decrease, what should you immediately think?
Uncompetitive inhibition
Memory: both go down together ⬇⬇
An inhibitor makes the enzyme appear to have higher affinity for substrate, but maximum rate drops. Which inhibition?
Uncompetitive inhibition
Why: lower Km means apparent higher affinity.
Graph trends for types of inhibitions
Competitive → Km ↑, Vmax same
Uncompetitive → Km ↓, Vmax ↓
Noncompetitive → Km same, Vmax ↓
Mixed → Vmax decreases, Km increases
The B-L-glucose is ________ .
A) a Hexose
B) an Aldose
C) a Pyranose
D) all of the above
All of the above
In comparison to B-L-glucose, A-L-glucose is:
A) a Stereoisomer
B) an Anomer
C) a Diastereoisomer
D) all of the above
All of the above
Fructose can form:
both pyranose and furanose rings
Cellulose is an example of a________
Polysaccharide
ATP:
A) is an example of the energy carrier molecule
B) is an abbreviation of the Adenosine Triphosphate
C) contains two high energy phosphate bonds
D) all of the above
All of the above
Feed-forward activation happens when a metabolite:
activates an enzyme further down the same metabolic pathway
As an important component of the cellular metabolism, NAD+
serves as an oxidizing agent in many reactions
The first step of Glycolysis:
A) is a phosphorylation reaction
B) is catalyzed by the enzyme hexokinase
C) is largely irreversible
D) all of the above
All of the above
Through glycolysis, one glucose molecule is converted into:
2 pyruvate molecules
2 ATP net (4 made, 2 used)
2 NADH
2 H₂O
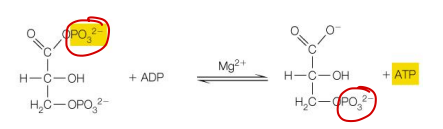
This reaction is
a substrate-level phosphorylation
This reaction shows:
ADP → ATP
A phosphate is transferred directly from the substrate to ADP
That is the definition of substrate-level phosphorylation:
A substrate directly donates phosphate to ADP to make ATP
✅ If ATP appears directly in one step, think substrate-level phosphorylation
Reaction of converting pyruvate to lactate regenerates_______
NAD+
The phosphofructokinase is a great example of how metabolism is regulated because
its activity is inhibited by ATP
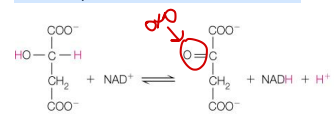
This TCA cycle reaction shows conversion of
L-malate to oxaloacetate
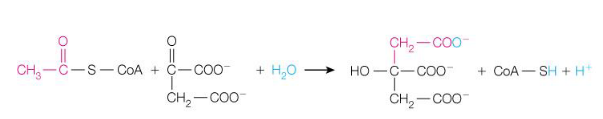
16. This important reaction is known as the committed step of the TCA cycle. Explain it.
it is the condensation of acetyl-CoA and oxaloacetate to citrate
A good example of a reaction catalyzed by enzyme dehydrogenase is:
A) conversion of L-malate to oxaloacetate
B) conversion of glyceraldehyde-3-phosphate to 1,3-bisphosphate glycerate
C) conversion of isocitrate to α-ketoglutarate
D) all of the above
D) all of the above
When drawing Glucose molecules be able to label:
1) Number carbons correctly
2) Recognize functional groups
3) Carbon which chirality defines the L/D Isomerization
Sucrose, commonly known as table sugar, is a disaccharide composed of:
a-D-glucose and B-D-Fructose
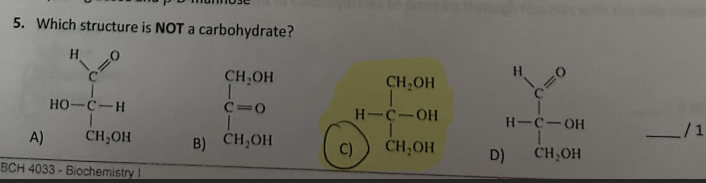
Why is that not a Carbohydrate?
A carbohydrate must have 1 carbonyl group either an aldehyde or ketone AND multiple hydroxyl groups.
(CH2O)n, roughly one oxygen per carbon
The intramolecular cyclization reaction of glucose:
Generates a new chiral center
Is a reaction between the aldehyde and the hydroxyl group
allows the formation of a glucopyranose
Pyrqanose rings are usually most stable when the ring adopts a________conformation with the bulkiest ring substituents in_______positions.
chair;equatorial
naturally occurring monosaccharides are predominantly what type of isomer
D-isomers
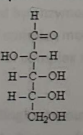
This molecule represents a carbohydrate, which is an
aldopentose
How do you classify a carbohydrate as aldose/ketose and pentose/hexose?
Look for carbonyl position:
Top carbonyl (CHO) = aldose
Middle carbonyl (C=O) = ketose
Count total carbons:
5 carbons = pentose
6 carbons = hexose
✅ Combine both names:
aldehyde + 5 carbons = aldopentose
ketone + 6 carbons = ketohexose
The storage polysaccharides are______ in animals and_____ in plants:
Glycogen, Starch. (Chitin for structure of animals and Cellulose in plants)
N-glycosidic bonds allow attachment of carbohydrates to proteins through reaction with the side chain of
Asparagine
To sustain life on earth the sun energy needs to be transformed into chemical energy by:
Autotrophs
A distinct set of metabolic reactions is called a:
Metabolic Pathway
Many biochemical processes are carried out via multi-step pathways rather than by single-step reactions (or only a few steps), because:
A) more control points to regulate biochemical processes.
B) Sharing of intermediates between pathways.
C) greater control over the amount of energy consumed or released.
Synthesis of complex molecules from simpler molecules
Anabolic process
Degredation of complex molecules into simpler molecules
Catabolic
The main energy-coupling compound in biochemical reactions that allows thermodynamically unfavorable processes to become favorable is:
ATP
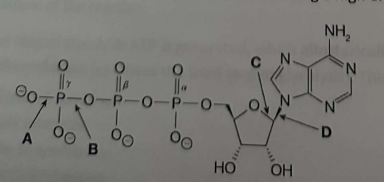
Which bond in ATP is primarily responsible for it being a high energy molecule?
B; Breaking the bond between phosphate groups relieves strong negative charge repulsion and releases usable energy.
The full bond is:
P–O–P
A substrate-level phosphorylation is a term given to reactions resulting in
ATP hydrolysis
Many metabolic reactions require an electron acceptor. Which molecule serves as one:
A) NAD+
B)NADP+
C)FAD
D)All of the above
NAD+
reactions in metabolic pathways that are subjected to regulation are those
displaced far from their chemical equilibrium
Which thermodynamic quantity is used to determine if a reaction will occur in the cell?
Delta G
The flow of matter through a metabolic pathway is called the
Flux
An enzyme that catalyzes a committed step in a metabolic pathway controls the:
Flux of that pathway
In the last step of glycolysis ATP is generated, which allosterically inhibits the enzyme phosphofructokinase (catalyzes the third step of glycolysis)/ This is an example of
Feedback inhibition
Phosphorylation of proteins (enzymes) at the expense of ATP is catalyzed by:
Kinases
The unidirectionality of Glycolysis is primarily due to
Three strongly exergonic, non-equilibrium reactions
How many ATP molecules are consumed during glycolysis for every one molecule of glucose?
2
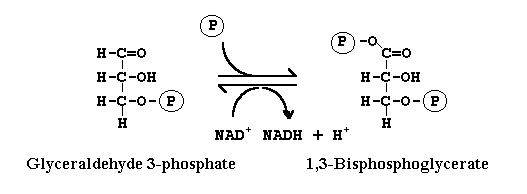
During glycolysis, conversion of glyceraldehyde-3-phosphate to 1,3-biphosphate glycerate is catalyzed by the enzyme:
Dehydrogenase
During glycolysis, isomerization occurs during the following:
Glucose 6- phosphate→ Fructose 6-phosphate
transfer of a high energy phosphoryl group to ADP, resulting in the production of ATP occurs during:
Substrate-level phosphorylation
examples:
1) 1,3-biphosphoglycerate→ 3-Phosphoglycerate
2) Phosphoenolpyruvate→ pyruvate
Glyceraldehyde 3-phosphate dehydrogenase causes:
reduction of NAD+ to NADH

In this reaction what happened
1,3 biphosphoglycerate is converted into 3-phosphoglycerate
In comparison to ATP, which compound has a greater free energy of hydrolysis?
Answer: 1,3-bisphosphoglycerate
Why correct: 1,3-bisphosphoglycerate has a higher phosphoryl-transfer potential than ATP, so it can donate phosphate to ADP to make ATP during glycolysis.
Pyruvate can be used to regenerate cytosolic pool of NAD in the processes of:
Answer: Fermentation
Why correct: In fermentation, pyruvate accepts electrons from NADH and is reduced (for example to lactate), which regenerates NAD⁺ so glycolysis can continue.
In process of Gluconeogenesis:
Answer: Pyruvate is used to generate glucose
Why correct: Gluconeogenesis is the pathway that builds glucose from non-carbohydrate precursors, and pyruvate is one of the main starting molecules.
In process of glycolysis, one glucose molecule is metabolized to generate (NET):
Answer: 2 pyruvate, 2 ATP, 2 NADH
Why correct: One glucose splits into two 3-carbon pyruvates, produces 4 ATP but uses 2 ATP, giving a net of 2 ATP, and forms 2 NADH.
Which of the following does NOT apply to the reaction catalyzed by the pyruvate dehydrogenase complex?
Answer: It requires different type of coenzymes
Why correct: Pyruvate dehydrogenase requires several coenzymes, but they work together as one coordinated enzyme complex, not as separate unrelated coenzyme types acting independently.
remember:
the process is highly exergonic and essentially irreversible in vivo
it is multistep reaction engaging three different enzymes
each reaction is catalyzed independently from the other reactions
List the components of the pyruvate dehydrogenase complex.
Answer:
Pyruvate dehydrogenase (E1)
Dihydrolipoamide transacetylase (E2)
Dihydrolipoamide dehydrogenase (E3)
What enzyme catalyzes a reaction of the citric acid cycle that does not produce reduced electron carriers (NADH or FADH2)?
Answer: Succinyl-CoA synthetase
Why correct: This step produces GTP (or ATP) by substrate-level phosphorylation instead of reduced electron carriers.
One substrate level phosphorylation occurs in the citric acid cycle in the reaction catalyzed by:
Answer: Succinyl-CoA synthetase
Why correct: It converts succinyl-CoA to succinate and directly forms GTP (or ATP).
Where are citric acid cycle enzymes located?
Answer: Mitochondrion
Why correct: Most citric acid cycle enzymes are in the mitochondrial matrix, with succinate dehydrogenase attached to the inner mitochondrial membrane.
Why does malate dehydrogenase proceed toward oxaloacetate even though ΔG°′ is positive?
Answer: the concentration of oxaloacetate is maintained at exceedingly low levels
Why correct: Oxaloacetate is immediately used in the next step, pulling the reaction forward.
Among different prosthetic groups utilized during the TCA cycle, enzyme that uses the Fe-S cluster is:
Answer: Aconitase
Why correct: Aconitase contains an Fe-S cluster used during citrate to isocitrate conversion.
What type of reaction is the conversion of fumarate to L-malate?
Answer: Hydration
Why correct: Water is added to fumarate to form malate.
What enzymes catalyze substrate-level phosphorylation reactions?
In glycolysis:
Phosphoglycerate kinase
Pyruvate kinase
In TCA cycle:
Succinyl-CoA synthetase
Most of the energy released in citric acid cycle reaction is conserved in form of:
Answer: NADH and FADH₂
Why correct: Most energy is stored in reduced electron carriers.
The ultimate product of complete oxidation of carbohydrates is:
Answer: pyruvate
Starting from glycolysis, through bridge reaction, till the end of the TCA cycle, metabolizing one glucose will generate:
Answer: 6x CO₂, 10x NADH/H⁺, 2x FADH₂ and 4x ATP
Why correct: This total combines glycolysis, pyruvate dehydrogenase, and TCA cycle yields.
Enzymes not only bind molecules called _______, they convert them chemically to a different _______.
substrates
Product
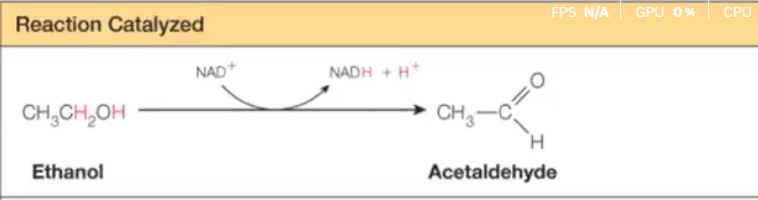
Oxidoreductases
Answer: Catalyze oxidation-reduction reactions (transfer electrons or hydrogen).
Example: Alcohol dehydrogenase
Transferases
Answer: Transfer a functional group from one molecule to another.
Example: Hexokinase
Hydrolases
Answer: Break bonds using water.
Example: Carboxypeptidase A
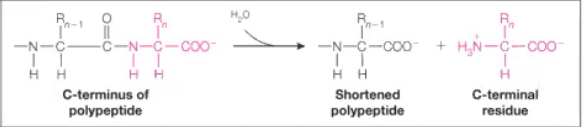
Lyases
Answer: Break or form bonds without water or redox reactions.
Example: Pyruvate decarboxylase
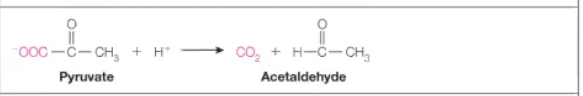
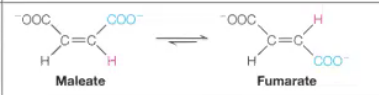
Isomerases
Answer: Rearrange atoms within the same molecule.
Example: Maleate isomerase
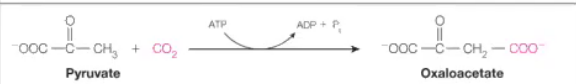
Ligases
Answer: Join two molecules using ATP.
Example: Pyruvate carboxylase

solve this

What 2 factors determine the velocity of an enzyme reaction?
The rate constant and the concentration of substrates and products
The observed rate of a chemical reaction can depend on:
The order of the reaction
the concentration of the reactants and products
temperature
the value of the rate constant for the reaction