Collision Theory
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Define Collision Theory
chemical reactions occur only if reactant atoms/molecules/ions collide
They must do this with correct orientation, and enough kinetic energy, Ek, to not only break bonds, but form new ones (the products) as well
What is Kinetic Energy? What is it proportional to?
movement of particles
proportional to temp in K
Average K.E is proportional to?
temp in K
Define Activation Energy
denoted by EA
minimum amount of energy colliding molecules must have for reaction to happen
helps overcome repulsion and weaken reactant bonds
How do reactions get to a transition state? What is formed as a result?
when enough energy reaches the activation energy
products!
What factors affect the Rate of Reactions? How do they affect the rate?
Temperature
Concentration
Particle Size
Pressure
Catalyst
they increase the # of successful collisions
What is Temperature? How does it affect the rate of a reaction?
average kinetic energy of particles
Increased temp. = increased kinetic energy = more energy for collisions = increased rate of collisions
small increases in temp. have big effects of rate (double rate for every 10°C)
What does a Maxwell-Boltzmann graph look like?
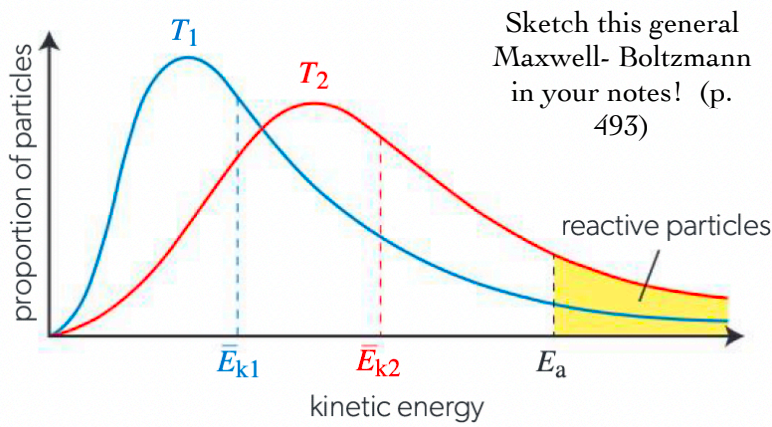
What is Concentration? How does it affect the rate of a reaction?
increased con.products = increased # of collisions = increased rate of reaction
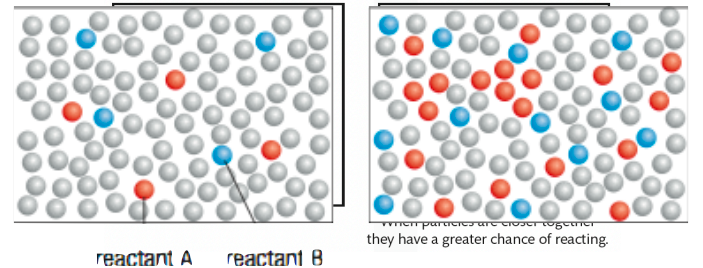
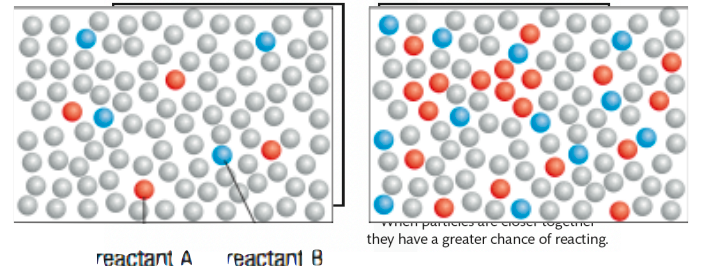
Which picture would have an increased rate of reaction? Why?
the right picture because it has a higher concentration of reactants
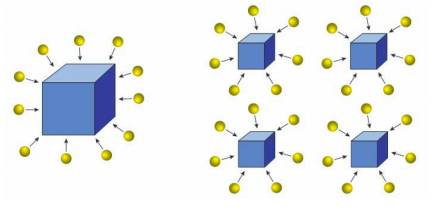
Which picture would have an increased rate of reaction? Why?
the right picture because it has a smaller particles for reactants
What is Particle Size? How does it affect the rate of a reaction?
Also known as Surface Area
decreasing particle size increases rate of reaction for heterogenous mixtures
Larger particles splitting into smaller parts increases surface area = increased contact = increased # of collisions
What can be the cause of industrial accidents?
Larger particles splitting into smaller parts increases surface area
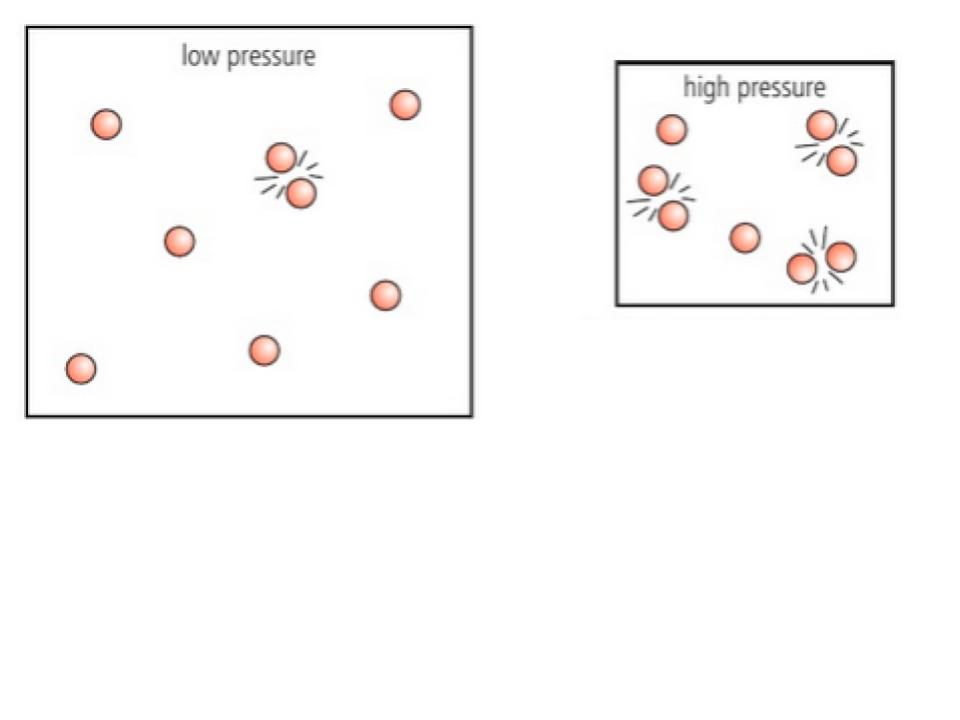
Which picture would have an increased rate of reaction? Why?
the picture on the right since with a higher pressure, the particles are closer together and are more likely to collide
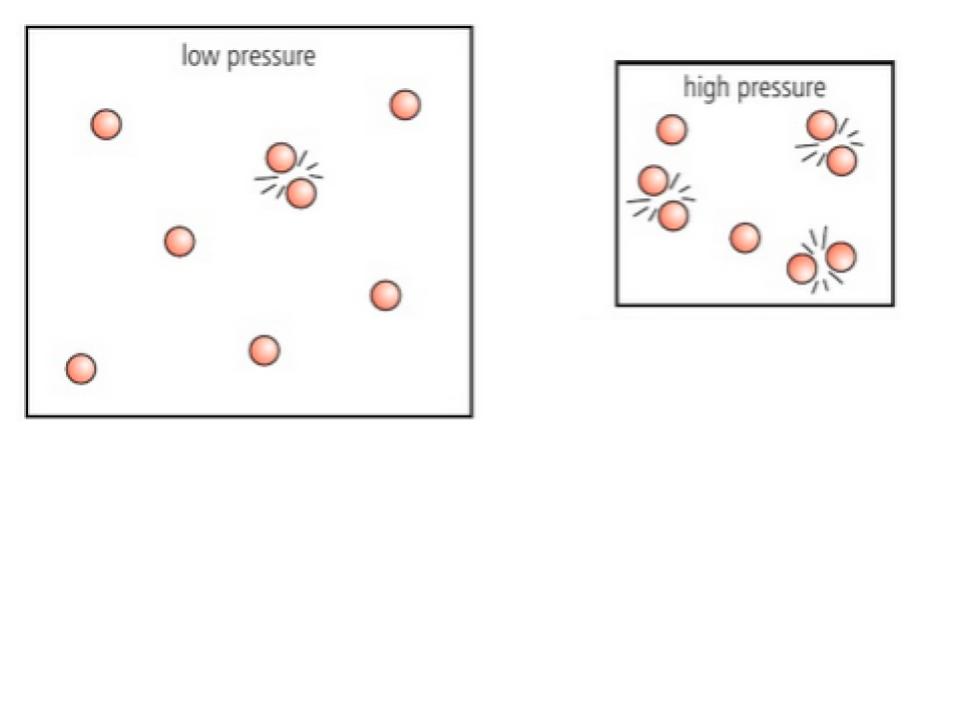
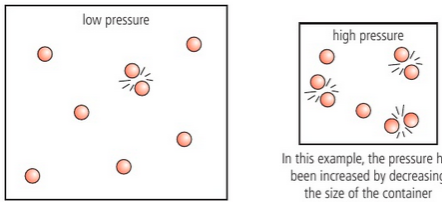
What is Pressure? How does it affect the rate of a reaction?
For GASES, increasing pressure increases rate of reaction
higher pressure compresses the gas increasing its concentration
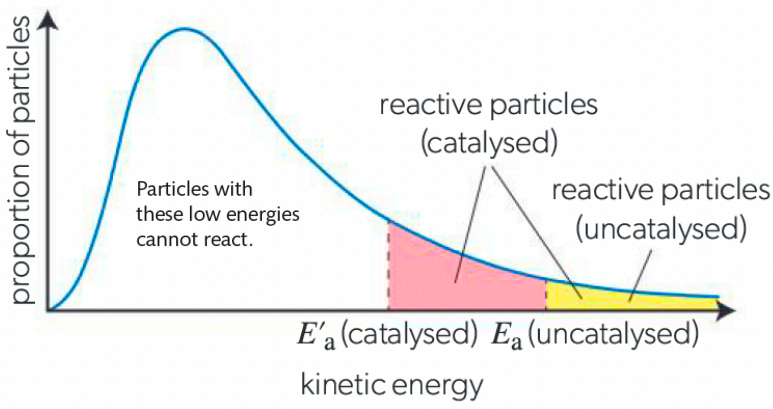
What are Catalysts and how do they affect the rate of reactions?
entities that increase rate of reaction by using alternative route
are not used up in reaction
Example of Maxwell-Boltzmann Diagram
What are some real life examples of catalysts?
Catalytic Converters | Enzymes | Green Chemistry |
device that converts toxic gas → less harmful ones | biological catalysts | Catalysis seeks to reduce negative impact of chemical processes on environment |