Dr M: Thyroid Gland and Thyroid Hormones
1/75
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
what are the names of the 3 hormones released from the thyroid
T3: triiodothyronine
T4: thyroxine
calcitonin
what are T3 and T4 important gfor?
growth and development, regulate energy metabolism, maintain body temperature
calcitonin function
important in regulating calcium metabolism
structure of the thyroid gland
consists of follicles (vesicles) made up of thyroid follicular cells and filled with thyrocolloid fluid
how are T3 and T4 stored
they're stored as amino acid residues in the thyroglobulin
what is thyroglobulin
a protein exclusively made by the thyroid gland that stores T3 and T4
constitutes the majority of thyroid follicular colloid content
iodothyronines, what are they?
iodinated tyrosine residues
where is thyroglobulin synthesized?
in the colloidal material fo cuboidal cells
thyroid hormone structure
made of a thyroglobulin with tyrosine residues attached
is T3 or T4 more potent
T3
binding areas of T3 and T4
both bind to specific cell surface receptors on sensitive target tissues which leads to increased uptake of glucose and amino acids
T3 also binds to cytoplasmic binding protein and nuclear receptor, leads to increased or decreased protein thesis
what does starvation do regarding thyroid hormones?
it decreases T3 and T4 receptors
iodide content and release in the thyroid
we have lots of iodide i the thyroid but only a small amount is released daily (to avoid Iodine deficiency)
what are the 6 steps to thyroid hormone synthesis
1. uptake of iodide (iodide trapping)
2. iodide organification (oxidation and iodination)
3. coupling of MIT and DIT
4. secretion of thyroid hormones
5. conversion of T4 to T3 (peripheral)
sodium iodide symptorter (NIS)
transports I with energy from Na/K ATPase
1 molecule of I brought in for every 2 molecules of Na brought in
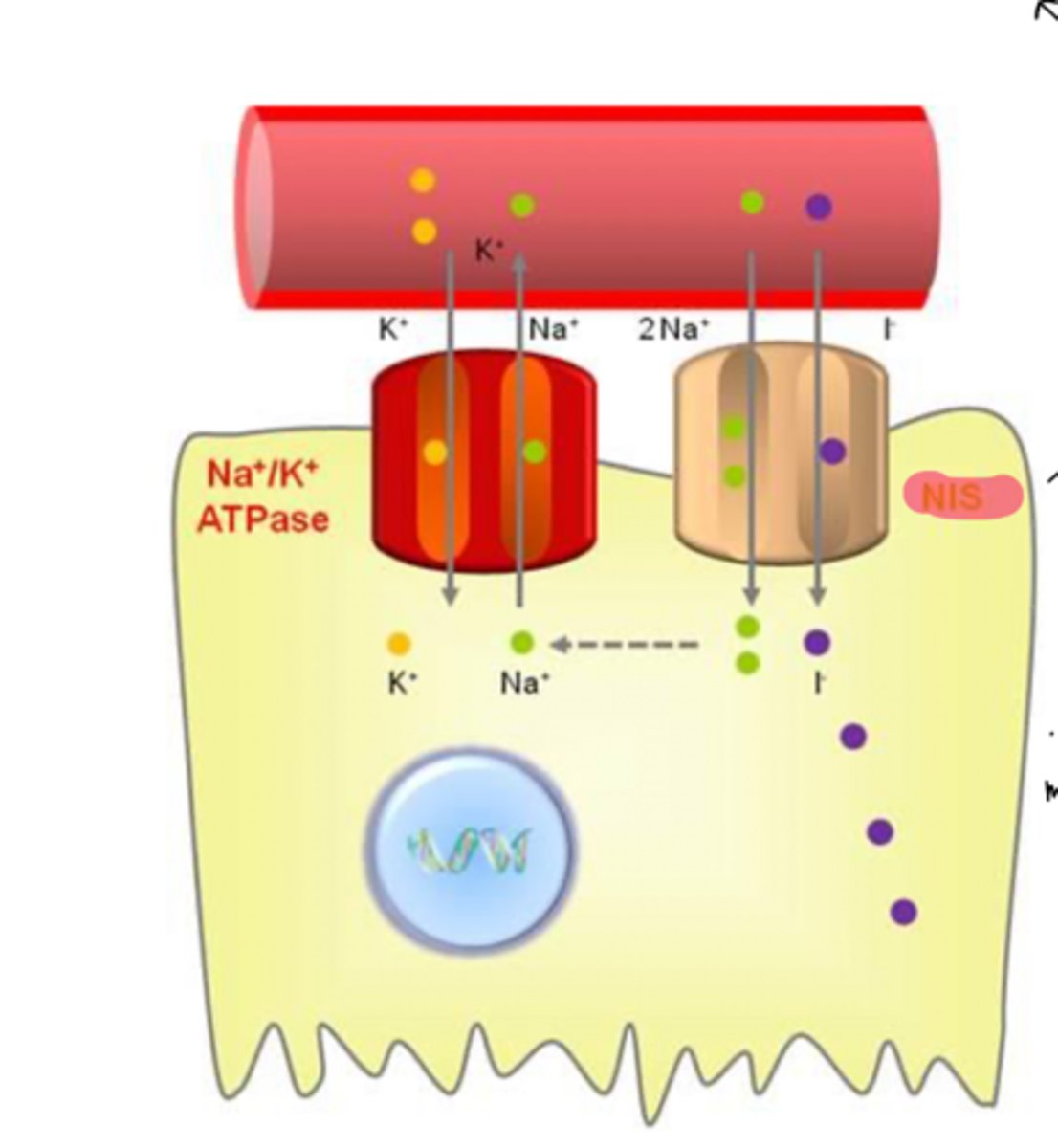
how does iodide trapping work (uptake of iodide)
- blood iodide levels are usually very low
-the thyroid gland actively concentrates and stores iodide
-uptake occurs from the blood into the thyroid follicular cell
auto-regulatory control of iodide uptake
-low iodide storage increases iodide uptake
-uptake is stimulated by TSH (AP)
-monovalent anions (perchlorate, thiocyanate, pertechnetate) inhibit uptake
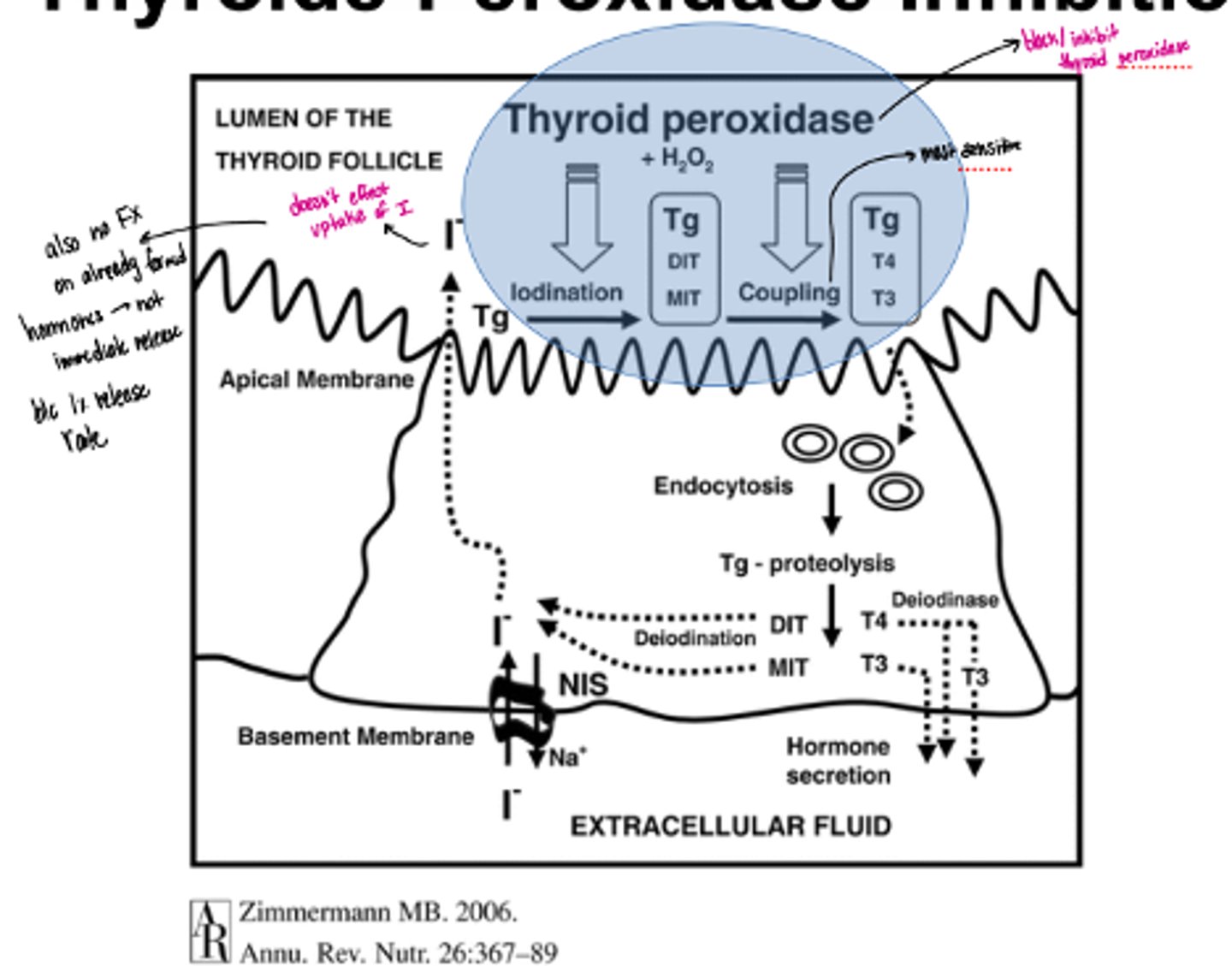
what enzyme is responsible for iodide organification
thyroid peroxidase (TPO)
thyroid peroxidase (TPO) function
-oxidizes iodide into iodine (blocked by thionamides)
-iodinates tyrosine residues in thyroglobulin to form MIT (mono-iodotyrosine) and DIT (di-iodotyrosine)
-coupling of MIT and DIT
what is the most sensitive step/ most blocked by drugs in thyroid hormone synthesis
coupling of MIT and DIT
what are the 2 parts that make triiodothyronine (T3)
MIT + DIT
what are the 2 parts that make thyroxine (T4)
DIT + DIT
where are T3 and T4 stored
in thyroglobulin in colloid matrix
ratio of T4:T3 synthesis
synthesis is 5 T4: 1 T3
mostly T4 is released
secretion of T4 and T3 in thyroid hormone synthesis
-endocytosis of TG (thyroglobulin) /colloid from follicular lumen
-fusion with lysosomal granules containing proteolytic enzymes
-breakdown of TG and release of T4 and T3
-MIT and DIT deiodinated; iodine re-utilized
-T4 and T3 reversibly bind to specific serum proteins thyroxine binding globulin (TGBG) and transthyretin; protect against metabolism and elimination
conversion of T4 to T3
-occurs peripherally
-de-iodination to active T3 and to reverse/inactive T4
thyroxine binding globuliin affinities
T4> T3
transthyretin
T4 and T3 binding protein but lower affinity than thyroxine binding globulin
autoregulation of the thyroid
increased iodine: decreased iodine uptake, conversion to iodine (oxidation) and organification
Graves Disease thyroid regulation
production of thryoid stimulating immunoglobulin (antibodies) by lymphocytes; mimics action of TSH; longer duration of action; autoimmune ddisease
tumors in thryoid regulation
thyrotoxicosis- excessive hormone production
what are 4 effects of thyroid hormones
-growth and development
-calorigenic effects
-CV effects
-metabolic effects
what are the 4 types of target tissues of thyroid homrone
-heart
-liver
-bone
-CNS
what is the "Wolff-Chaikoff Effect"
high levels of Iodine inhibit thyroid peroxidase and shut down glan uptake, synthesis, and release
can undergo escape and become hyperactive
whta happens when T3 binds to the nuclear thyroid receptor
creates a heterodimer with RXR
effects transcription of proteins
deiodinase (D2 ad D3)
removes iodine from iodothyronines (converts T4--> T3-->T2)
thyroid hormone functions in growth and development
o Promotes optimal growth and development in all body tissues
o Stimulates protein synthesis, likely by enhancing DNA transcription
o Critical for the health of the nervous system, skeletal system, and reproductive organs
what are the 2 causes of cretinism
-Iodine deficiency
-Improper development (or failure to develop) of the thyroid glad
what are 2 characteristics of cretinism
-Dwarfism
-Mental impairment
calorigenic effects of thyroid hormoen
Thyroid hormones increase resting or basal metabolic rate of whole organism
increase body temperature
what happens to thyroid hormone if you're cold
increased production of T3 and T4
what 4 tissues are most sensitive to the calorigenic effects of thyroid hormone
heart, skeletal muscle, liver, and kidneys
3 CV effects of thyroid hormone
· Increase HR
· Increase force of contraction
· Increased cardiac output
mechanism of the direct effect of thyroid hormone on CV
binding to thyroid hormone receptors in the heart; not just response to increased basal metabolism
mechanism of the indirect effect of thyroid hormone on CV
increased number of B-adrenergic receptors in the heart
what are 4 metabolic effects of thyroid hormone
· Stimulates metabolism of cholesterol to bile acids
· Increase binding of LDL by liver
· Increase carbohydrate metabolism
· Increase glucose uptake
What is Grave's disease?
diffuse toxic goiter caused by autoimmune antibodies (thyroid stimulating immunoglobulins) overstimulating the thyroid gland
Has prominent in-folding of the hyperplastic epithelium
key symptoms of Grave's disease
weight loss, rapid HR, nervousness/irritability, and protruding eyes (exophthalmos), goiter
TSI (thyroid stimulating immunoglobulins MOA in Grave's disease
they mimic TSH stimulation but are constant instead of pulsatile
what is Plummer's disease
a toxic nodular goiter caused by one or more hot/overactive nodules in the thyroid gland which produce excessive thyroid hormones
nodule is an adenoma (benign)
key symptoms of Plummer's disease/toxic nodular goiter
weight loss, rapid HR, heat intolerance, tremors, often without exophthalmos (unlike in Graves)
whta is the difference in symptoms between Grave's and Plummer's disease
Grave's presents with exophthalmos (bulging eyes) while Plummer's doesn't
what are the 3 MOA's of treating hyperthyroidism
· Agents that interfere with production of T3 and T4
· Agents that modify tissue responses to T3 and T4
Destructionof thyroid gland
what is a thyroid storm (thyrotoxic crisis)
life-threatening form of thyrotoxicosis
what is needed to treat a thyroid storm
immediate treatment with propaolol
goitrogens
· agents that decrease production of thyroid hormones
o Produce enlargement of the glands in patients with normal thyroid function
what are the 4 thionamides/thioureas
o Methimazole (10x more potent)
o Propylthiouracil (PTU)
o Carbimazole (UK)
o Ethylenethiourea or ETU & 3-aminotriazole
MOA of thionamides
accumulate in the thyroid and inhibit synthesis of T3 and T4 (inhibit thyroid peroxidase)
block Iodine organification and coupling of MIT and DIT
also decreases TSI tier
what effect do thionamides have in people with normal thyroids
· Inhibition of T4 and T3 synthesis- low levels of circulating hormones- increase in TSH release in normal people- sustained glandular stimulation- glandular enlargement (“non-productive”)
thyroid peroxidase inhibition
· Inhibits this enzyme (especially affects coupling)
· Doesn't affect the intake of iodine or already formed hormones
· Not immediate due to 1% release/day
monovalent anion inhibitor MOA
resemble iodide; competitive inhibitors inhibit transport of iodide into thyroid. can be overcome by increased Iodide
diagnostic use
what are the 4 anion inhibitors
§ Perchlorate (ClO4-)
§ Pertechnetate(TcO4-)
§ Thiocynanate (SCN-)
§ Cesium
Iodide MOA in hyperthyroidism treatment
· Inhibit iodide uptake, organification of iodide, and release of thyroid hormones into the bloodstream
· Decreased size and vascularity of hyperplastic gland
o Preoperative preparation of thyrotoxic patients
· Should not be used alone (thioamides)
how does reactive iodide get into the thyroid
is has sleective uptake into the thyroid and gets concentrated there because it's indistinguishable from normal iodide
MOA of reactive iodide (I-131)
emits beta radiation which destroys the thyroid gland
what is the 1 disadvantage of reactive iodide
can cause delayed hypothyroidism
adrenoreceptor blocking agents in hyperthyroidism
· Many symptoms of thyrotoxicosis mimic sympathetic stimulation
· Need to use agents that rapidly delete catecholamines or B-blockers
· Block physiological effects of sympathetic nervous system stimulation
propanolol and guanethidine
what can happen to the thyroid in nuclear disasters
· Radioactive iodine can get into the gland and cause glandular destruction
KI (potassium iodide) MOA
salt of stable, non-radioactive, iodine that blocks radioactive iodine from being absorbed by the thyroid gland, thus protecting this gland from radiation injury
o Shuts down gland so radioactive can't be absorbed
what is myxedema
a severe/advanced form of hypothyroidism
key symptoms of myxedema and Hashimoto's
weight gain, cold intolerance, lethargy/fatigue, slowed HR, dry skin
Hashimoto's also can have a goiter
what is Hashimoto's thyroiditis/ chronic lymphocytic thyroiditis
an autoimmune disorder in which the body's immune system attacks the thyroid gland
what is the most common cause of hypothyroidism in the US?
hashimoto's
what is endemic goiter
hypothyroidism due to deficiency in iodide intake
less common in developed countries
diagnosis of hypothyroidism
decreased T4 production; presence of anti-thyroid antibody (autoimmune)
treatment of choice for hypothyroidism
levothyroxine (T4)