UNIT 6 (HOMEOSTASIS)
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
Chapter 44: Fluid Imbalances
Maintained by balance of volume, osmolality, electrolytes, and pH
Body fluid content
Adults: 55%–60% of body weight
Older adults: ~50%–55% (decreased muscle mass, ↓ thirst)
Fluid movement between compartments via
Diffusion
Osmosis
Filtration
Active transport
Fluid balance regulated by intake vs output
Intake influenced by thirst, habits, social factors
Output via kidneys (primary regulator), skin, lungs, GI tract
Types of Fluid Imbalances
Volume imbalances
Too little or too much isotonic fluid
Osmolality imbalances
Hypertonic or hypotonic states
Examples
Hypernatremia (water deficit)
Hyponatremia (water excess or intoxication)
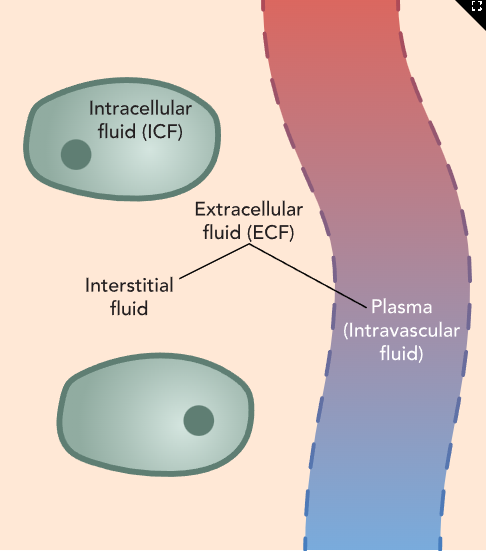
Body Fluid Compartments
Intracellular Fluid (ICF)
Two-thirds of total body water
Fluid inside cells
Extracellular Fluid (ECF)
One-third of total body water
Fluid outside cells
Subdivisions
Intravascular: plasma (liquid part of blood)
Interstitial: between cells and blood vessels
Transcellular: epithelial secretions
CSF
Pleural
Peritoneal
Synovial
Intracellular / Extracellular / Plasma (image)
Dehydration
Deficit of body fluid from inadequate intake or excessive loss
Types
Actual dehydration
Total body water loss
Relative dehydration
Fluid shifts from intravascular to interstitial space
Hypovolemia (isotonic dehydration)
Loss of water and electrolytes
↓ circulating blood volume
Also called fluid volume deficit
Dehydration Risk Factors
Causes of Isotonic Fluid Volume Deficit (Hypovolemia)
GI losses
Vomiting
Diarrhea
NG suctioning
Skin losses
Diaphoresis without fluid replacement
Renal losses
Diuretics
Kidney disease
Adrenal insufficiency
Third spacing
Burns
Blood or plasma loss
Hemorrhage
Decreased intake
Anorexia
Nausea
Impaired swallowing
Confusion
NPO status
Causes of Dehydration
Hyperventilation or excessive perspiration without fluids
Prolonged fever
Diabetic ketoacidosis
Inadequate water intake
Enteral feedings without free water
↓ thirst sensation
Aphasia
Diabetes insipidus
Osmotic diuresis
Excessive sodium intake
Salt tablets
Hypertonic IV fluids
Dehydration Findings
Vital Signs
Tachycardia (compensatory)
Hypotension
Orthostatic hypotension
Thready pulse
↓ central venous pressure
Tachypnea (compensation)
Hypothermia
Neuromusculoskeletal
Dizziness
Syncope
Confusion
Weakness
Fatigue
Gastrointestinal
Thirst
Dry, furrowed tongue
Nausea
Vomiting
Anorexia
Acute weight loss
Renal
Oliguria (↓ urine output and concentration)
Other Findings
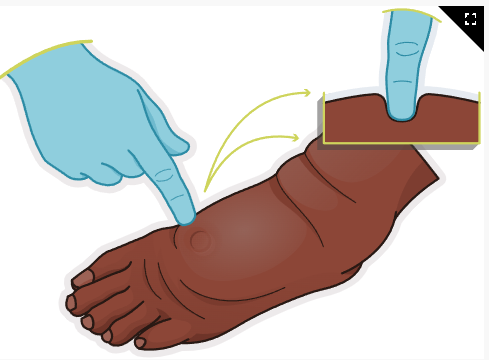
Delayed capillary refill
Cool, clammy skin
Diaphoresis
Sunken eyeballs
Flattened neck veins
Poor skin turgor, tenting
Weight loss
↓ CVP
Older adults at higher risk due to
↓ skin elasticity
↓ GFR and urine-concentrating ability
↓ muscle mass
↓ thirst reflex
Severe dehydration
Elevated temperature
Risk of seizures
Dehydration Labs
Hematocrit: ↑ in hypovolemia
BUN: ↑ (>25 mg/dL) from hemoconcentration
Urine specific gravity: >1.030
Serum sodium: >145 mEq/L (with dehydration)
Serum osmolality: >295 mOsm/kg (dehydration, hypernatremia)
Note
With hemorrhage, hemoconcentration does not occur initially
Dehydration Care
Administer oral or IV rehydration
Monitor I&O
Monitor vital signs
Orthostatic BP
Heart rate
Monitor mental status (early sign of worsening imbalance)
Weigh every 8 hours during fluid replacement
Assess gait stability
Use call light
Assist with ambulation (fall risk)
Instruct slow position changes
Interprofessional Care
Collaborate on
Fluid replacement strategy
Oxygen management
Client Education
Encourage adequate fluid intake
Identify dehydration causes
Vomiting
Diarrhea
Large draining wounds
Excessive ostomy output
Dehydration Complications
Hypovolemic Shock
Results from severe fluid loss
↓ MAP → ↓ tissue perfusion
↓ oxygen delivery to cells
Nursing Actions
Administer oxygen
O₂ saturation <70% = emergency
Remain with unstable client
Monitor vital signs every 15 minutes
Fluid replacement
Colloids
Whole blood
Packed RBCs
Plasma
Synthetic plasma expanders
Crystalloids
Normal saline
Lactated Ringer’s
Administer medications as ordered
Vasoconstrictors
Dopamine
Norepinephrine
Phenylephrine
Inotropes
Dobutamine
Milrinone
Vasodilators for perfusion support
Sodium nitroprusside
Perform hemodynamic monitoring
The nurse is assessing the client who reports nausea, vomiting, and weakness. Which of the following findings nurse are manifestations of fluid volume deficit?
Select all that apply.
a
Potassium level
b
Urine specific gravity
c
Heart rate
d
Temperature
e
Oxygen saturation
b
Urine specific gravity
c
Heart rate
d
Temperature
Overhydration
Excess fluid in the body from excessive intake or ineffective removal
Key Concepts
Fluid overload
Excess water or fluid
Includes water intoxication
Causes hemodilution (↓ blood components like cells and electrolytes)
Hypervolemia
Excess water and electrolytes in correct proportions
Example: excessive sodium intake → water retention
Increased risk for
Pulmonary edema
Congestive heart failure
Older adults at higher risk due to
↓ kidney function
Age-related regulatory changes
Health Promotion and Disease Prevention
For clients with heart disease or kidney impairment
Consume low-sodium diet
Follow prescribed dietary restrictions
Restrict fluid intake as ordered
Overhydration Risk Factors
Causes of Hypervolemia
Compromised regulatory systems
Heart failure
Kidney disease
Cirrhosis
Excessive fluid administration
Oral
Enteral
IV
Fluid shifts after burns
Prolonged corticosteroid use
Severe stress
Hyperaldosteronism
Causes of Overhydration
Water replacement without electrolytes
Excessive water intake
Forced intake
Psychogenic polydipsia
SIADH (syndrome of inappropriate antidiuretic hormone)
Excessive IV fluids
Hypotonic solutions used for irrigation
Overhydration Findings
Vital Signs
Tachycardia
Bounding pulse
Hypertension
Tachypnea
↑ central venous pressure
Neuromuscular
Weakness
Visual changes
Paresthesias
Altered level of consciousness
Seizures (severe, rapid hyponatremia from water excess)
Gastrointestinal
Ascites
↑ GI motility
Hepatomegaly
Respiratory
Crackles
Cough
Dyspnea
Other Findings
Peripheral edema
Pulmonary congestion
Weight gain
Distended neck veins
↑ urine output
Skin warm, pale, taut
Overhydration Exams
Laboratory Findings
↓ Hematocrit and hemoglobin (hemodilution)
↓ Serum osmolality
↓ Urine sodium
↓ Urine specific gravity
↓ BUN (plasma dilution)
Diagnostic Procedures
Chest X-ray
Possible pulmonary congestion
Overhydration Care
Monitor intake and output
Monitor daily weight
1 kg (2.2 lb) change in 24 hr = ~1 L fluid
Assess lung sounds
Monitor peripheral edema
Maintain sodium-restricted diet as prescribed
Maintain fluid restriction if ordered
Encourage rest
Monitor response to diuretics
Review OTC medications for sodium content
Positioning
Semi-Fowler’s or Fowler’s
Reposition regularly to prevent skin breakdown
Use pressure-reducing mattress
Assess bony prominences
Monitor serum sodium and potassium
Interprofessional Care
Consult respiratory therapy for oxygen management
Consult pulmonology if fluid shifts into lungs
Client Education
Weigh daily
Report 1–2 lb gain in 24 hr
Report 3 lb gain in 1 week
Follow low-sodium diet
Read food labels
Track daily sodium intake
Adhere to fluid restrictions
Divide daily fluid allowance evenly over 24 hr
Overhydration Complications
Pulmonary Edema
Caused by severe fluid overload
Manifestations
Anxiety
Tachycardia
Distended neck and hand veins
PVCs
Dyspnea at rest
Altered LOC
Restlessness, lethargy
Moist crackles
Frothy, pink-tinged sputum
Nursing Actions
Position in high-Fowler’s to improve ventilation
Administer oxygen
Prepare for positive airway pressure or intubation if needed
Administer medications as prescribed if BP adequate
Morphine
Nitrates
Diuretics
Pitting edema (image)
A nurse is planning care for a client who has fluid volume excess. Which of the following interventions should the nurse include in the plan?
Select all that apply.
a
Check the client’s weight 2 times per week
b
Place the client in a semi-Fowler’s position
c
Monitor the client’s breath sounds
d
Change the client’s position every 4 hrs
e
Assess the client for peripheral edema
Fluid volume deficit
Sunken eyeballs
Poor skin turgor
Fever
Fluid volume excess
Bounding pulse
Crackles heard in lung fields
Distended neck veins
A nurse is planning care for a client who has fluid volume excess. Which of the following interventions should the nurse include in the plan?
Select all that apply.
a
Check the client’s weight 2 times per week
b
Place the client in a semi-Fowler’s position
c
Monitor the client’s breath sounds
d
Change the client’s position every 4 hrs
e
Assess the client for peripheral edema
b
Place the client in a semi-Fowler’s position
c
Monitor the client’s breath sounds
e
Assess the client for peripheral edema
Chapter 45: Electrolyte Imbalances
Electrolytes are charged ions dissolved in body fluids.
Cations are positively charged. Anions are negatively charged.
Electrolytes are distributed between intracellular fluid (ICF) and extracellular fluid (ECF).
Differences in electrolyte concentration between ICF and ECF maintain cell excitability and nerve impulse transmission.
Body fluids remain electrically neutral overall.
Electrolytes conduct electrical current.
Imbalances occur from altered intake or output.
Older adults and ill clients are at higher risk.
Serum labs reflect plasma levels only, not intracellular levels.
Common Electrolytes
Cations: sodium, potassium, calcium, magnesium, hydrogen
Anions: chloride, bicarbonate, phosphate, sulfate, proteins
Expected Reference Ranges
Sodium: 136–145 mEq/L
Potassium: 3.5–5.0 mEq/L
Calcium: 9.0–10.5 mg/dL
Magnesium: 1.3–2.1 mEq/L
Chloride: 98–106 mEq/L
Phosphorus: 3.0–4.5 mg/dL
Sodium Imbalances
Physiology
Sodium (Na⁺) is the primary ECF cation.
Regulates ECF osmolarity and fluid balance.
ICF sodium is low (≈14 mEq/L).
Sodium gradients are critical for muscle contraction, cardiac function, and nerve transmission.
Water follows sodium concentration.
Kidneys regulate sodium via aldosterone, ADH, and natriuretic peptides.
Hyponatremia
Serum sodium less than 136 mEq/L.
Caused by sodium loss or excess free water.
Pathophysiology
Slowed membrane depolarization.
Water shifts from ECF into ICF, causing cellular edema.
Risk of cerebral edema.
Common Causes
Fluid imbalance leading to sodium loss.
Compensatory renal excretion of sodium-free water.
Diagnostic Clues
Urine sodium helps distinguish renal vs nonrenal losses.
Hyponatremia Risk Factors
Actual Sodium Deficit
Excessive sweating
Diuretic use
Wound drainage, especially GI
NG tube suction of isotonic gastric contents
Decreased aldosterone secretion
Kidney disease
Hyperlipidemia
Inadequate sodium intake or NPO status
Hyperglycemia
Low-sodium diet
Cerebral salt wasting syndrome
Relative Sodium Deficit Due to Dilution
Excess hypotonic fluids (psychogenic polydipsia, forced intake, hypotonic irrigation)
Freshwater submersion
Kidney failure or nephrotic syndrome
Heart failure
SIADH
Medications: SSRIs, desmopressin
Older adult status with chronic illness and diuretic use
Hyponatremia Findings
General
Manifestations depend on ECF volume status: euvolemic, hypovolemic, or hypervolemic.
Hypervolemic Hyponatremia
Bounding pulse
Blood pressure normal or elevated
Vital Signs in Hypovolemia
Hypothermia
Tachycardia
Rapid, thready pulse
Hypotension
Orthostatic hypotension
Diminished peripheral pulses
Neuromuscular
Headache
Confusion
Lethargy
Muscle weakness
Fatigue
Decreased deep tendon reflexes
Dizziness or lightheadedness
Seizures
Possible respiratory compromise
Gastrointestinal
Increased GI motility
Hyperactive bowel sounds
Abdominal cramping
Nausea
Hyponatremia Exams
Serum sodium: less than 136 mEq/L
Serum osmolality: decreased
Urine sodium:
Less than 20 mEq/L with sodium loss
Greater than 20 mEq/L in SIADH
Urine specific gravity:
Decreased (1.002–1.004) with sodium loss
Increased in SIADH
Hyponatremia Care
Encourage oral sodium intake if tolerated.
Provide high-sodium foods and fluids such as broth or tomato juice.
Administer isotonic IV fluids such as 0.9% normal saline or lactated Ringer’s.
Correct sodium slowly.
Do not exceed 12 mEq/L increase in 24 hours (prevents osmotic demyelination).
Restrict fluids if fluid overload is present.
Monitor intake and output.
Obtain daily weights.
Monitor vital signs and level of consciousness.
Interprofessional Care
Nephrology consultation for electrolyte management.
Respiratory therapy for oxygen support if needed.
Nutrition services for sodium-rich diet planning and fluid restriction.
Client Education
Weigh daily.
Report weight gain of 1–2 lb in 24 hours or 3 lb in one week.
Follow prescribed high-sodium diet.
Read food labels for sodium content.
Track daily sodium intake.
Hyponatremia Complications
Severe Hyponatremia
Seizures
Coma
Respiratory arrest
Requires immediate treatment
Nursing Actions
Goal is to raise serum sodium to reduce neurologic symptoms.
Maintain airway and monitor vital signs.
Implement seizure precautions.
Monitor level of consciousness.
Administer hypertonic oral or IV fluids as prescribed.
Administer 3% sodium chloride cautiously with frequent sodium monitoring.
Target sodium level not greater than 125 mEq/L during hypertonic therapy.
Hypernatremia
Serum sodium greater than 145 mEq/L.
Increased sodium causes hypertonicity of blood.
Water shifts out of cells, leading to cellular dehydration.
Can cause severe neurologic, endocrine, and cardiac disturbances.
Hypernatremia Risk Factors
Actual Sodium Excess
Kidney failure
Cushing’s syndrome
Hyperaldosteronism
Medications such as glucocorticoids
Excessive oral sodium intake
Relative Sodium Excess Due to Decreased Fluid Volume
Water deprivation or NPO status
Hypertonic enteral feedings without adequate free water
Diabetes insipidus
Heatstroke
Hyperventilation
Watery diarrhea
Burns
Excessive sweating
Hypernatremia Findings
Thirst
Intense thirst (cellular dehydration)
Vital Signs
Hyperthermia
Tachycardia
Orthostatic hypotension
Neuromuscular
Restlessness
Irritability
Muscle twitching progressing to weakness
Decreased or absent deep tendon reflexes
Seizures
Coma
Possible respiratory muscle weakness
Gastrointestinal
Dry mucous membranes
Nausea
Vomiting
Anorexia
Occasional diarrhea
Hypernatremia Exams
Laboratory Findings
Serum sodium greater than 145 mEq/L
Serum osmolality greater than 300 mOsm/L
Urine specific gravity increased
Urine osmolality increased
Hypernatremia Care
Monitor level of consciousness and ensure safety.
Monitor vital signs and cardiac rhythm.
Auscultate lung sounds.
Provide oral hygiene and comfort measures for thirst.
Monitor intake and output.
Notify provider of decreased urine output.
Monitor potassium levels if diuretics are administered.
Fluid Management
Based on Serum Osmolality and Hemodynamic Status
Dextrose 5% in 0.45% sodium chloride
Hypertonic before infusion
Becomes hypotonic after glucose metabolism
0.45% sodium chloride
Hypotonic solution
Gradual sodium reduction
Preferred if severe hyperglycemia is present
Dextrose 5% in water or 0.9% sodium chloride
Isotonic solutions
Management of Excess Sodium
Encourage water intake.
Restrict sodium intake.
Administer loop diuretics if renal excretion is impaired.
Interprofessional Care
Nutrition services for low-sodium diet planning and fluid management.
Client Education
Weigh daily.
Report weight gain of 1–2 lb in 24 hours or 3 lb in one week.
Follow a low-sodium diet.
Read food labels for sodium content.
Track daily sodium intake.
Adhere to prescribed fluid intake.
Avoid over-the-counter medications containing sodium bicarbonate.
Hypernatremia Complications
Severe Hypernatremia
Seizures
Convulsions
Death if untreated
Nursing Actions
Maintain airway patency.
Monitor vital signs closely.
Implement seizure precautions.
Monitor level of consciousness frequently.
Potassium Imbalances
Physiology
Potassium (K⁺) is the major intracellular cation.
About 98% of potassium is inside cells.
Essential for cell metabolism, nerve impulse transmission, and cardiac, lung, and skeletal muscle function.
Maintains acid–base balance.
Has a reciprocal relationship with sodium.
Small potassium changes cause significant clinical effects.
Hypokalemia
Serum potassium less than 3.5 mEq/L.
Caused by potassium loss or shift of potassium into cells.
Hypokalemia Risk Factors
Actual Potassium Deficit
Overuse of diuretics, digitalis, corticosteroids
Increased aldosterone secretion
Cushing’s syndrome
GI losses: vomiting, diarrhea, prolonged NG suction
Excessive laxative or tap water enema use (hypotonic fluid with isotonic GI losses)
NPO status
Kidney disease with impaired potassium reabsorption
Relative Potassium Deficit
Alkalosis (potassium shifts into cells)
Hyperinsulinism
Hyperalimentation
Total parenteral nutrition
Water intoxication
Older adults with frequent diuretic or laxative use
Hypokalemia Findings
Vital Signs
Decreased blood pressure
Weak, thready pulse
Orthostatic hypotension
Neurologic
Altered mental status
Anxiety
Lethargy progressing to confusion or coma
Cardiac
Dysrhythmias
ECG Changes
Flattened or inverted T waves
Prominent U waves
ST depression
Prolonged PR interval
Gastrointestinal
Hypoactive bowel sounds
Nausea
Vomiting
Constipation
Abdominal distention
Possible paralytic ileus
Muscular
Muscle weakness
Decreased deep tendon reflexes
Respiratory
Shallow respirations
Risk for respiratory compromise
Hypokalemia Exams
Laboratory Findings
Serum potassium less than 3.5 mEq/L
Diagnostic Procedures
Electrocardiogram
Inverted or flat T waves
ST depression
Elevated U waves specific to hypokalemia
Possible dysrhythmias
Hypokalemia Care
Administer prescribed potassium replacement.
Never give potassium IM or subcutaneously (tissue necrosis risk).
Monitor urine output before potassium administration.
Monitor respiratory status and breath sounds.
Continuous cardiac monitoring as indicated.
Monitor clients receiving digoxin (increased toxicity risk).
Monitor level of consciousness and ensure safety.
Assess bowel sounds and abdominal distention.
Monitor oxygen saturation above 95%.
Assess hand grasps for weakness.
Assess deep tendon reflexes.
Implement fall precautions.
Potassium Replacement
Oral Replacement
Encourage potassium-rich foods:
Avocados
Broccoli
Dairy products
Dried fruit
Cantaloupe
Bananas
Fruit juices
Melon
Lean meats
Milk
Whole grains
Citrus fruits
Salt substitutes are high in potassium.
Administer oral potassium supplements as prescribed.
IV Potassium Supplementation
Never give IV push or bolus (cardiac arrest risk).
Maximum rate: 10 mEq per hour.
Maximum concentration: 1 mEq per 10 mL solution.
Monitor IV site for phlebitis (tissue irritation).
Interprofessional Care
Nephrology for electrolyte management.
Respiratory therapy for oxygen support.
Nutrition services for potassium-rich diet planning.
Cardiology for dysrhythmia management.
Client Education
Identify potassium-rich foods.
Avoid excessive use of diuretics and laxatives unless prescribed.
Adhere to prescribed potassium therapy.
Hypokalemia Complications
Respiratory Failure
Due to respiratory muscle weakness
Nursing Actions
Maintain airway patency.
Monitor vital signs.
Monitor for hypoxemia and hypercapnia.
Assist with intubation and mechanical ventilation if indicated.
Cardiac Arrest
Due to severe dysrhythmias
Nursing Actions
Continuous cardiac monitoring.
Treat dysrhythmias promptly.
Hyperkalemia
Serum potassium greater than 5.0 mEq/L.
Caused by increased potassium intake, movement of potassium out of cells, or impaired renal excretion.
High risk for life-threatening dysrhythmias and cardiac arrest.
Rare in clients with normal kidney function.
Hyperkalemia Risk Factors
Actual Potassium Excess
Older adults (↓ renin and aldosterone; ↑ use of salt substitutes, ACE inhibitors, potassium-sparing diuretics)
Excess intake of high-potassium foods or salt substitutes
Excessive or rapid potassium replacement (oral or IV)
RBC transfusions
Adrenal insufficiency
ACE inhibitors
Potassium-sparing diuretics
Kidney failure
Relative Potassium Excess (Shift Out of Cells)
Decreased insulin production
Metabolic acidosis (diabetic ketoacidosis)
Tissue damage (sepsis, trauma, surgery, fever, myocardial infarction)
Hyperuricemia
Hyperkalemia Findings
Vital Signs
Slow, irregular pulse
Hypotension
Neuromuscular
Restlessness
Irritability
Muscle weakness progressing to ascending flaccid paralysis
Paresthesia
Cardiac
Dysrhythmias
ECG Changes
Peaked T waves
Widened QRS
Prolonged PR
Ventricular dysrhythmias
Possible ventricular fibrillation or asystole
Gastrointestinal
Increased motility
Diarrhea
Hyperactive bowel sounds
Renal
Oliguria
Hyperkalemia Exams
Laboratory Findings
Serum potassium greater than 5.0 mEq/L
Hemoglobin and hematocrit
Increased with dehydration
Decreased with kidney failure
BUN and creatinine increased with kidney failure
Arterial blood gases
Metabolic acidosis (pH less than 7.35) with kidney failure
Diagnostic Procedures
Electrocardiogram
Peaked T waves
Widened PR and QRS
Absent or flattened P waves
ST depression
Possible heart block, ventricular fibrillation, or asystole
Hyperkalemia Care
Priority is prevention of falls, early recognition of cardiac complications, and health teaching.
Monitor cardiac rhythm continuously.
Monitor intake and output.
Assess muscle strength and weakness.
Monitor for GI symptoms such as nausea and intestinal cramping.
Stop potassium infusions immediately if potassium is elevated.
Maintain IV access.
Discontinue potassium supplements.
Promote potassium-restricted diet.
Monitor serum potassium levels frequently.
Monitor for hypokalemia during treatment.
Severe Management
Administer calcium gluconate as prescribed (cardiac membrane stabilization).
Dialysis may be required for severe or chronic hyperkalemia.
Shift Potassium Into Cells
IV dextrose with regular insulin (drives potassium into cells).
Sodium bicarbonate if metabolic acidosis is present.
Prevention
Avoid whole blood or packed RBC transfusions in clients with impaired renal function when possible.
Avoid high-potassium foods:
Citrus fruits
Legumes
Whole grains
Lean meats
Milk
Eggs
Cocoa
Some cola beverages
Teach clients to read food labels for potassium content.
Low-potassium foods include:
Fruits: apples, cranberries, grapes, canned peaches, cranberry or grape juice
Vegetables: lettuce, cabbage, cucumbers, green peppers, sweet onions, green peas, green beans
Leaching vegetables reduces potassium content (peel, soak, drain, boil)
Refined grains instead of whole grains
Beverages: brewed tea, coffee, ginger ale, root beer
Other: applesauce, angel food cake, butter, margarine, hard candy, sugar, honey
Clients with kidney disease on potassium-sparing diuretics should avoid potassium supplements and salt substitutes.
Interprofessional Care
Nephrology for dialysis and electrolyte management.
Nutrition services for potassium-restricted diet planning.
Cardiology for dysrhythmia management.
Client Education
Identify low-potassium food choices.
Read food labels for potassium content.
Avoid salt substitutes containing potassium.
Hold potassium supplements unless directed by the provider.
Hyperkalemia Meds
To Increase Potassium Excretion
Loop Diuretics (Furosemide)
Use only if kidney function is adequate.
Increase renal potassium loss.
Monitor intake and output.
Cation-Exchange Resins
Sodium polystyrene sulfonate
Binds potassium in the GI tract and excretes it in stool.
Dialysis may be required if potassium remains critically high.
Beta-2 Agonists (Albuterol)
Shifts potassium into cells.
Monitor for tachycardia and chest pain.
Patiromer
Binds potassium in the GI tract.
Used for chronic hyperkalemia.
IV Insulin and Glucose
Shifts potassium into cells.
Hyperkalemia Complications
Cardiac Arrest
Nursing Actions
Treat dysrhythmias immediately.
Maintain continuous cardiac monitoring.
A nurse is teaching a class about electrolyte imbalances. The nurse should include that which of the following conditions places a client at risk for hyperkalemia?
a
Diabetic ketoacidosis
b
Heart failure
c
Cushing’s syndrome
d
Thyroidectomy
a
Diabetic ketoacidosis
Other Electrolyte Imbalances
Calcium: hypocalcemia, hypercalcemia
Chloride: hypochloremia, hyperchloremia
Magnesium: hypomagnesemia, hypermagnesemia
Phosphorus: hypophosphatemia, hyperphosphatemia
Hypocalcemia and hypomagnesemia have high clinical risk due to neuromuscular and cardiac effects.
Hypocalcemia
Total serum calcium less than 9.0 mg/dL.
Hypocalcemia Risk Factors
Actual Calcium Deficit
Inadequate calcium intake
Lactose intolerance
Malabsorption syndromes
Chronic diarrhea or steatorrhea
Inadequate vitamin D intake
End-stage kidney disease
Wound drainage
Relative Calcium Deficit
Alkalosis
Acute pancreatitis
Hyperproteinemia
Hyperphosphatemia
Immobilization
Calcium-chelating treatments
Citrate
Mithramycin
Sodium cellulose phosphate
Penicillamine
Pamidronate
Parathyroid gland removal or damage
Prolonged immobility
Hypocalcemia Findings
Neuromuscular
Tetany (increased neuromuscular excitability)
Paresthesia of fingers and lips
Muscle twitching
Painful muscle spasms at rest, especially calves or feet
Seizures
Hyperactive deep tendon reflexes
Positive Chvostek’s sign
Positive Trousseau’s sign
Risk Indicators
History of thyroid or upper neck surgery
History of neck or chest irradiation
Cardiovascular
Prolonged QT interval
Prolonged ST segment
Risk for torsades de pointes
Decreased myocardial contractility
Bradycardia and hypotension in severe cases
Gastrointestinal
Hyperactive bowel sounds
Diarrhea
Abdominal cramping
Hypocalcemia Exams
Laboratory Findings
Serum calcium less than 9.0 mg/dL
Low albumin can falsely lower total calcium
Ionized calcium reflects true calcium status when albumin is low
Diagnostic Procedures
ECG showing prolonged QT and ST intervals
Hypocalcemia Care
Administer oral or IV calcium supplements as prescribed.
Administer vitamin D to improve calcium absorption.
Implement seizure and fall precautions.
Reduce environmental stimulation.
Keep emergency equipment available.
Encourage calcium-rich foods:
Dairy products
Yogurt
Canned salmon
Sardines
Fresh oysters
Dark leafy green vegetables
Treat severe hypocalcemia with IV calcium gluconate or calcium chloride.
Dilute IV calcium in dextrose 5% in water.
Administer slowly via infusion pump.
Monitor cardiac rhythm closely.
Rapid administration can cause cardiac arrest.
Interprofessional Care
Endocrinology for electrolyte regulation.
Respiratory therapy for oxygen support if needed.
Nutrition services for calcium-rich diet planning.
Cardiology for dysrhythmia management.
Client Education
Consume calcium-rich foods such as milk and yogurt.
Read food labels to increase dietary calcium intake.
A nurse is teaching a class about electrolyte imbalances.
Match the electrolyte imbalance with the associated manifestations.
Hyponatremia
Hypocalcemia
Hyperkalemia
Hypokalemia
Hypoactive bowel sounds
ECG with tall, peaked t-waves
Positive Trousseau’s sign
Bounding pulse
Hypoactive bowel sounds
Hypokalemia
ECG with tall, peaked t-waves
Hyperkalemia
Positive Trousseau’s sign
Hypocalcemia
Bounding pulse
Hyponatremia
The nurse is assessing the client for Chvostek’s sign. Which of the following actions should the nurse take?
a
Apply a blood pressure cuff to the client’s arm.
b
Place a stethoscope bell over the client’s carotid artery.
c
Ask the client to lower their chin to their chest.
d
Tap lightly on the client’s cheek.
d
Tap lightly on the client’s cheek.
The nurse is assessing the client who reports nausea, vomiting, and weakness. Which of the following findings are manifestations of hypocalcemia?
Select all that apply.
a
Tingling in fingers
b
Poor skin turgor
c
Abdominal pain
d
Elevated temperature
e
Muscle twitching
a
Tingling in fingers
c
Abdominal pain
e
Muscle twitching
Hypomagnesemia
Serum magnesium less than 1.3 mg/dL.
Hypomagnesemia Risk Factors
Celiac disease or Crohn’s disease (malabsorption)
Malnutrition or inadequate magnesium intake
Chronic alcohol use (increased renal magnesium excretion)
Diarrhea, steatorrhea, or chronic laxative use
Citrate exposure from blood products
Myocardial infarction or heart failure
Concurrent hypokalemia or hypocalcemia
Medications:
Aminoglycoside antibiotics
Cisplatin
Cyclosporine
Amphotericin B
Hypomagnesemia Findings
Cardiovascular
Increased risk for dysrhythmias
Possible hypertension
ECG changes:
Premature ventricular contractions
Flat or inverted T waves
ST depression
Prolonged PR interval
Widened QRS
Neuromuscular
Increased nerve impulse transmission
Hyperactive deep tendon reflexes
Paresthesia
Muscle tetany
Seizures
Positive Chvostek’s sign
Positive Trousseau’s sign
Gastrointestinal
Hypoactive bowel sounds
Constipation
Abdominal distention
Paralytic ileus
Neuropsychiatric
Depressed mood
Apathy
Agitation
Hypomagnesemia Care
Correct concurrent electrolyte imbalances, especially potassium and calcium.
Encourage magnesium-rich foods for mild deficiency:
Dark green vegetables
Nuts
Whole grains
Seafood
Peanut butter
Cocoa
Discontinue magnesium-depleting medications when possible:
Loop diuretics
Osmotic diuretics
Phosphorus-containing medications
Administer oral magnesium for mild hypomagnesemia.
Administer IV magnesium sulfate for severe hypomagnesemia.
Use an infusion pump.
Do not exceed 150 mg per minute or 67 mEq over 8 hours.
Monitor deep tendon reflexes hourly during IV administration.
Monitor clients taking digoxin closely (low magnesium increases digoxin toxicity risk).
Keep calcium gluconate available to reverse magnesium toxicity if needed.
Chapter 46: Acid-Base Imbalances
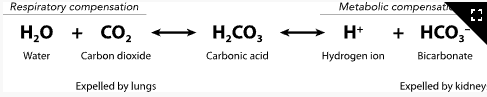
Acid–base balance maintains hydrogen ion (H⁺) homeostasis in body fluids
Small changes in H⁺ significantly affect cellular function
Arterial pH reflects combined respiratory and renal function
Normal arterial pH: 7.35–7.45
Relationship
↑ H⁺ = ↓ pH (acidic)
↓ H⁺ = ↑ pH (alkaline)
ABGs assess acid–base status
CO₂ regulated by lungs
HCO₃⁻ regulated by kidneys
Maintenance of Acid–Base Balance
Chemical Buffers (First Line of Defense)
Bicarbonate system and intracellular buffers
Protein buffers (albumin, globulins)
Bind or release H⁺ as needed
Rapid response to pH changes
Respiratory Buffers (Second Line of Defense)
Control H⁺ via CO₂ elimination
Chemoreceptors sense CO₂ changes and adjust respirations
Hyperventilation
Triggered by ↑ CO₂
↓ H⁺ via CO₂ exhalation
Hypoventilation
Triggered by ↓ CO₂
↑ H⁺ via CO₂ retention
Renal Buffers (Third Line of Defense)
Slowest but most effective and longest lasting
Regulate bicarbonate reabsorption or excretion
Can generate new bicarbonate
Responses
pH < 7.35: ↑ H⁺ → reabsorb and produce HCO₃⁻
pH > 7.45: ↓ H⁺ → excrete HCO₃⁻
Compensation
Body attempts to normalize pH using lungs and kidneys
Full compensation: pH returns to 7.35–7.45
Partial compensation: pH remains abnormal
Types of Acid–Base Imbalances
Respiratory acidosis
Respiratory alkalosis
Metabolic acidosis
Metabolic alkalosis
Health Promotion and Disease Prevention
Balanced diet and regular physical activity
Limit alcohol intake
Adequate hydration (6–8 cups water daily)
Maintain healthy weight
Smoking cessation
Compensation (image)
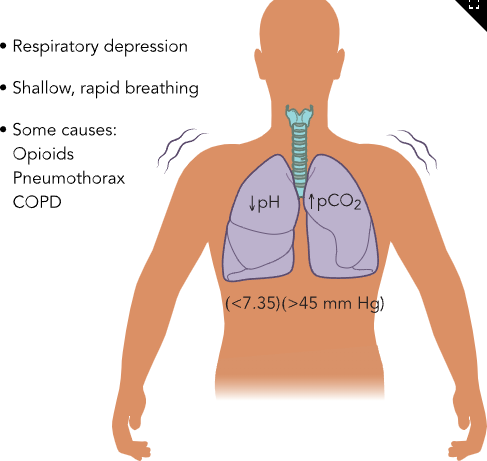
Respiratory Acidosis (Hypoventilation)
Causes
Respiratory depression (opioids, anesthetics, poisons)
CNS disorders (stroke, brain tumor, trauma, neuromuscular disease)
Impaired chest expansion (obesity, pneumothorax, hemothorax, flail chest)
Airway obstruction (edema, mucus, foreign body, lymph node enlargement)
Alveolar-capillary impairment (pulmonary embolism, ARDS, pulmonary edema)
Inadequate mechanical ventilation
ABG Findings
↑ CO₂
↑ or normal H⁺ (↓ pH)
Manifestations
Vital signs: early tachycardia and hypertension, late bradycardia and hypotension
Dysrhythmias (ventricular fibrillation possible with anesthesia)
Neurologic: anxiety, confusion → lethargy, coma
Respiratory: shallow, rapid, ineffective breathing
Skin: pale or cyanotic
Nursing Care
Oxygen therapy
Maintain patent airway
Improve ventilation and gas exchange
Positioning, breathing techniques
Ventilatory support, bronchodilators, mucolytics
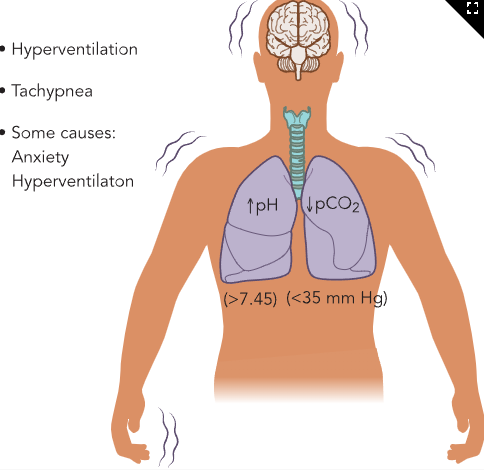
Respiratory Alkalosis (Hyperventilation)
Causes
Anxiety, fear, pain
Intracranial trauma
Salicylate toxicity
Excessive mechanical ventilation
Hypoxemia (high altitude, shock, early asthma, early pneumonia)
ABG Findings
↓ CO₂
↓ or normal H⁺ (↑ pH)
Manifestations
Vital signs: tachypnea
Neurologic: dizziness, tingling, numbness, tinnitus, possible LOC
Cardiovascular: tachycardia, atrial or ventricular dysrhythmias
Respiratory: rapid, deep respirations
Nursing Care
Treat underlying cause
Oxygen if hypoxemic
Anxiety reduction
Rebreathing techniques
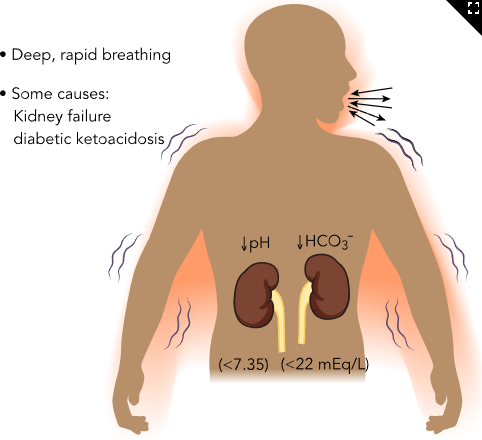
Metabolic Acidosis
Causes
Excess acid production
Diabetic ketoacidosis
Starvation
Lactic acidosis (exercise, seizures, hypoxia)
Excess acid ingestion
Ethanol, methanol
Aspirin toxicity
Decreased acid excretion
Kidney failure
Decreased bicarbonate production
Kidney failure
Pancreatitis
Loss of bicarbonate
Diarrhea
Impaired liver or pancreatic function
ABG Findings
↓ HCO₃⁻
↑ H⁺ (↓ pH)
Manifestations
Dysrhythmias
Vital signs: bradycardia, hypotension, weak pulses, tachypnea
Neurologic: headache, confusion, drowsiness
Respiratory: Kussmaul respirations (deep, rapid)
Skin: warm, dry, pink
Nursing Care
Treat cause
DKA: administer insulin
GI loss: antidiarrheals and rehydration
Severe low HCO₃⁻: sodium bicarbonate (1 mEq/kg)
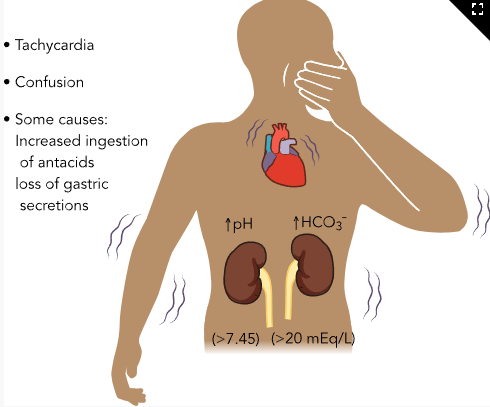
Metabolic Alkalosis
Causes
Base excess
Excess antacid intake
IV bicarbonate, blood transfusions, TPN
Acid loss
Prolonged vomiting
NG suction
Potassium depletion
Thiazide diuretics
Laxative overuse
Hyperaldosteronism, Cushing syndrome
Increased digitalis toxicity
ABG Findings
↑ HCO₃⁻
↓ H⁺ (↑ pH)
Manifestations
Vital signs: tachycardia, normotension or hypotension
Dysrhythmias (worsen as pH increases)
Neurologic: numbness, tingling, tetany, muscle weakness, confusion, seizures
Respiratory: hypoventilation from muscle weakness
Nursing Care
Treat cause
GI losses: antiemetics, IV fluids, electrolyte replacement
Potassium depletion: stop causative agent and replace electrolytes
Diagnostic Procedures for Acid–Base Imbalances
Step 1: Assess pH
pH < 7.35: Acidosis
pH > 7.45: Alkalosis
Step 2: Assess PaCO₂ and HCO₃⁻ Together
Identify which value is within normal range and which is abnormal
The abnormal value indicates the primary imbalance
PaCO₂
Normal: 35–45 mm Hg
< 35 or > 45 indicates respiratory origin
HCO₃⁻
Normal: 22–26 mEq/L
< 22 or > 26 indicates metabolic origin
Step 3: Name the Imbalance
Combine pH result with the abnormal PaCO₂ or HCO₃⁻ value
Step 4: Assess Oxygenation
Evaluate PaO₂ and SaO₂
Values below expected range indicate hypoxemia
Step 5: Determine Compensation
Uncompensated
pH abnormal
Either PaCO₂ or HCO₃⁻ abnormal
Partially compensated
pH abnormal
PaCO₂ and HCO₃⁻ both abnormal
Fully compensated
pH normal
PaCO₂ and HCO₃⁻ both abnormal
Use pH direction to identify the primary problem
pH < 7.40: acidosis
pH > 7.40: alkalosis
Respiratory Acidosis (image)
Respiratory Alkalosis (image)
Metabolic Acidosis (image)
Metabolic Alkalosis (image)
A nurse is teaching a group of nurses about conditions that can cause metabolic acidosis. Which of the following conditions should the nurse include?
a
Diabetic ketoacidosis
b
Myasthenia gravis
c
Asthma
d
Laxative overuse
a
Diabetic ketoacidosis
A nurse is reviewing ABGs on a client who has vomited for 24 hr. Which of the following acid‑base imbalances should the nurse expect?
a
Respiratory acidosis
b
Respiratory alkalosis
c
Metabolic acidosis
d
Metabolic alkalosis
d
Metabolic alkalosis
Classic ABG Patterns
Normal (Homeostasis)
pH: 7.35–7.45
PaCO₂: 35–45
HCO₃⁻: 22–26
Respiratory Acidosis
pH < 7.35
PaCO₂ > 45
HCO₃⁻ normal
Metabolic Acidosis
pH < 7.35
PaCO₂ normal
HCO₃⁻ < 22
Respiratory Alkalosis
pH > 7.45
PaCO₂ < 35
HCO₃⁻ normal
Metabolic Alkalosis
pH > 7.45
PaCO₂ normal
HCO₃⁻ > 26
Acid-Base Imbalance Care
Treat the underlying cause of the imbalance
Tailor interventions to the client’s condition and severity
Interprofessional Care
Respiratory therapy
Oxygen therapy
Breathing treatments
ABG monitoring
Pulmonology consultation for complex respiratory management
Client Education
Follow prescribed diet and dialysis regimen if renal disease present
Daily weights
Report 1–2 lb (0.5–0.9 kg) gain in 24 hours
Report 3 lb (1.4 kg) gain in one week
Smoking cessation counseling
Take medications as prescribed
COPD management adherence
Arrange home oxygen if indicated
Acid-Base Imbalance Complications
Seizures
Coma
Respiratory arrest
Nursing Actions
Implement seizure precautions as needed
Initiate life-support measures when indicated
A nurse is teaching a group of nurses about acid-base imbalances. Match the acid-base imbalance with the ABG result.
Respiratory alkalosis
Metabolic alkalosis
Respiratory acidosis
Metabolic acidosis
pH 7.30 PaCO 2 48 mm Hg HCO 3 − 26 mEq/L
pH 7.50 PaCO 2 28 mm Hg HCO 3 − 24 mEq/L
pH 7.32 PaCO 2 35 mm Hg HCO 3 - 18 mEq/L
pH 7.50 PaCO 2 38 mm Hg HCO 3 − 30 mEq/L
pH 7.30 PaCO 2 48 mm Hg HCO 3 − 26 mEq/L
Respiratory acidosis
pH 7.50 PaCO 2 28 mm Hg HCO 3 − 24 mEq/L
Respiratory alkalosis
pH 7.32 PaCO 2 35 mm Hg HCO 3 - 18 mEq/L
Metabolic acidosis
pH 7.50 PaCO 2 38 mm Hg HCO 3 − 30 mEq/L
Metabolic alkalosis
A nurse is caring for a client who was in a motor‑vehicle accident and reports chest pain and difficulty breathing. A chest x‑ray reveals the client has a pneumothorax. Which of the following ABG results should the nurse expect?
a
pH 7.25 PaCO2 52 mm Hg HCO3− 24 mEq/L
b
pH 7.42 PaCO2 38 mm Hg HCO3− 23 mEq/L
c
pH 7.30 PaCO2 36mm Hg HCO3− 18mEq/L
d
pH 7.50 PaCO2 29 mm Hg HCO3− 26 mEq/L
a
pH 7.25 PaCO2 52 mm Hg HCO3− 24 mEq/L