MAT E 202 MIDTERM
0.0(0)
Studied by 10 peopleCard Sorting
1/175
Earn XP
Description and Tags
Last updated 7:47 PM on 10/17/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
176 Terms
1
New cards
Metals
Atoms are surrounded by a "sea of free e-"
non-directional bonding
non-directional bonding
2
New cards
Ceramics and Glasses
Can have covalent or ionic bonding
3
New cards
Covalent bonding
Valence electrons are shared
directional bond
electronegativity determines the amount of covalent bonding
directional bond
electronegativity determines the amount of covalent bonding
4
New cards
Ionic Bonding
Occurs between a cation and an anion
Requires transfer of electrons
Non-directional bond
Requires transfer of electrons
Non-directional bond
5
New cards
Polymers
two types of bonding (intra-chain bonding and secondary bonding)
6
New cards
Intra-chain bonding
bonding between atoms in polymer chain
7
New cards
Secondary bonding (polymers)
Van der Waals bonding between chains on polymers
8
New cards
What does bonding affect in a material
-strength
-elastic modulus
-ductility
-performance
-thermal and electrical properties
-elastic modulus
-ductility
-performance
-thermal and electrical properties
9
New cards
Different types of stress
-tension
-compression
-shear
-compression
-shear
10
New cards
Uniaxial tension
loading occurs in one direction
11
New cards
Biaxial tension
loading occurs in multiple directions
12
New cards
Hydrostatic compression
compression due to water pressures
highly complex
highly complex
13
New cards
For metals, what is assumed about it's tensile properties
tensile properties are the same as the compressive
(commonly not true)
(commonly stronger in compression)
(commonly not true)
(commonly stronger in compression)
14
New cards
Bauschinger effect
effect that the tensile and compressive properties of a material are not the same
15
New cards
Uniaxial tensile test
-most common mechanical property tested
-must obtain a representative section of material
-specimens can be rectangular of cylindrical
-must obtain a representative section of material
-specimens can be rectangular of cylindrical
16
New cards
Standard tensile test conditions
- room temperature
-loaded at a very slow rate
-strain rate of (5.75*10^-5 - 5.75*10^-6)
-loaded at a very slow rate
-strain rate of (5.75*10^-5 - 5.75*10^-6)
17
New cards
Strain rate formula
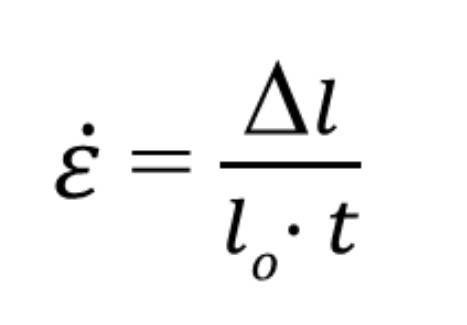
18
New cards
True stress
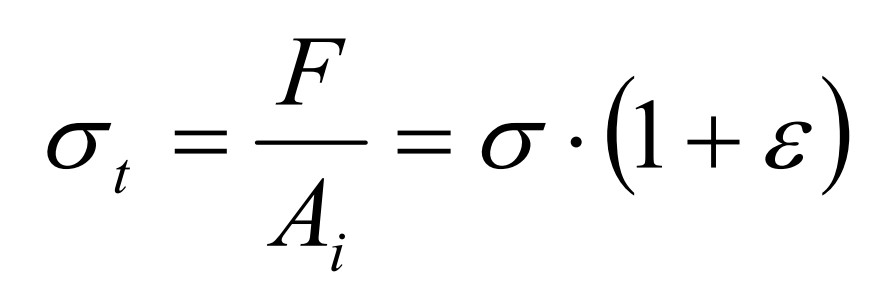
19
New cards
True strain
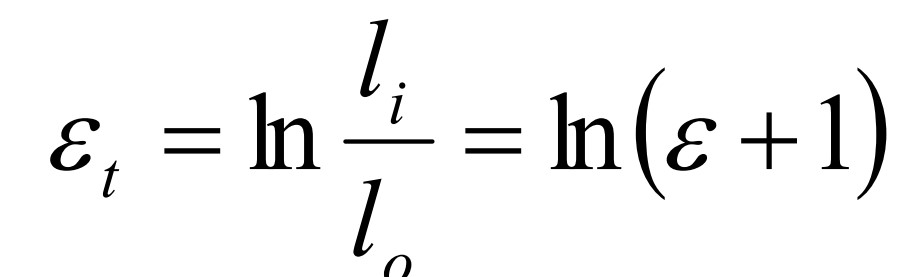
20
New cards
When are true stress/strain values used
When large strains are present
21
New cards
Ceramic stress strain curve
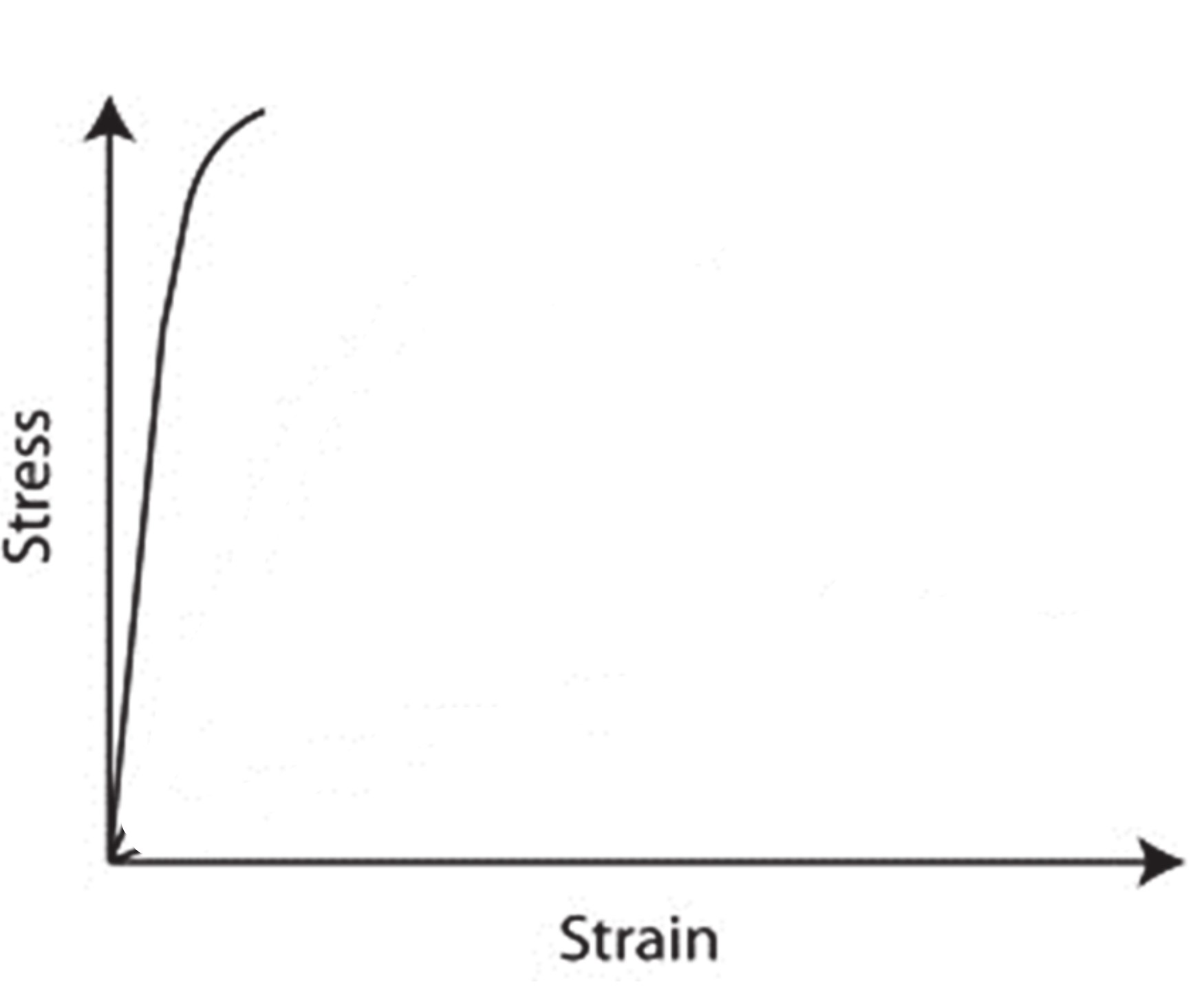
22
New cards
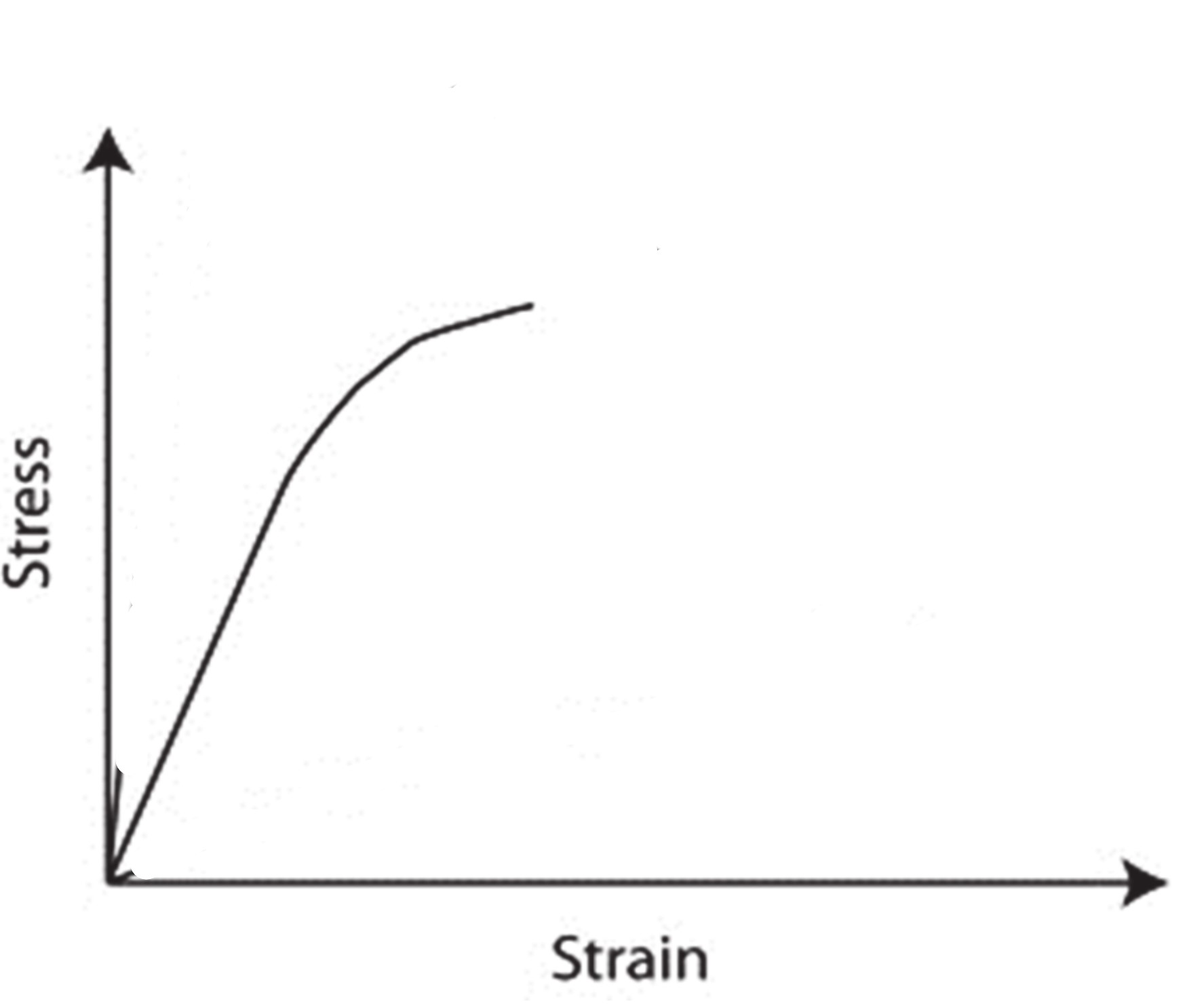
Metal stress strain curve
23
New cards
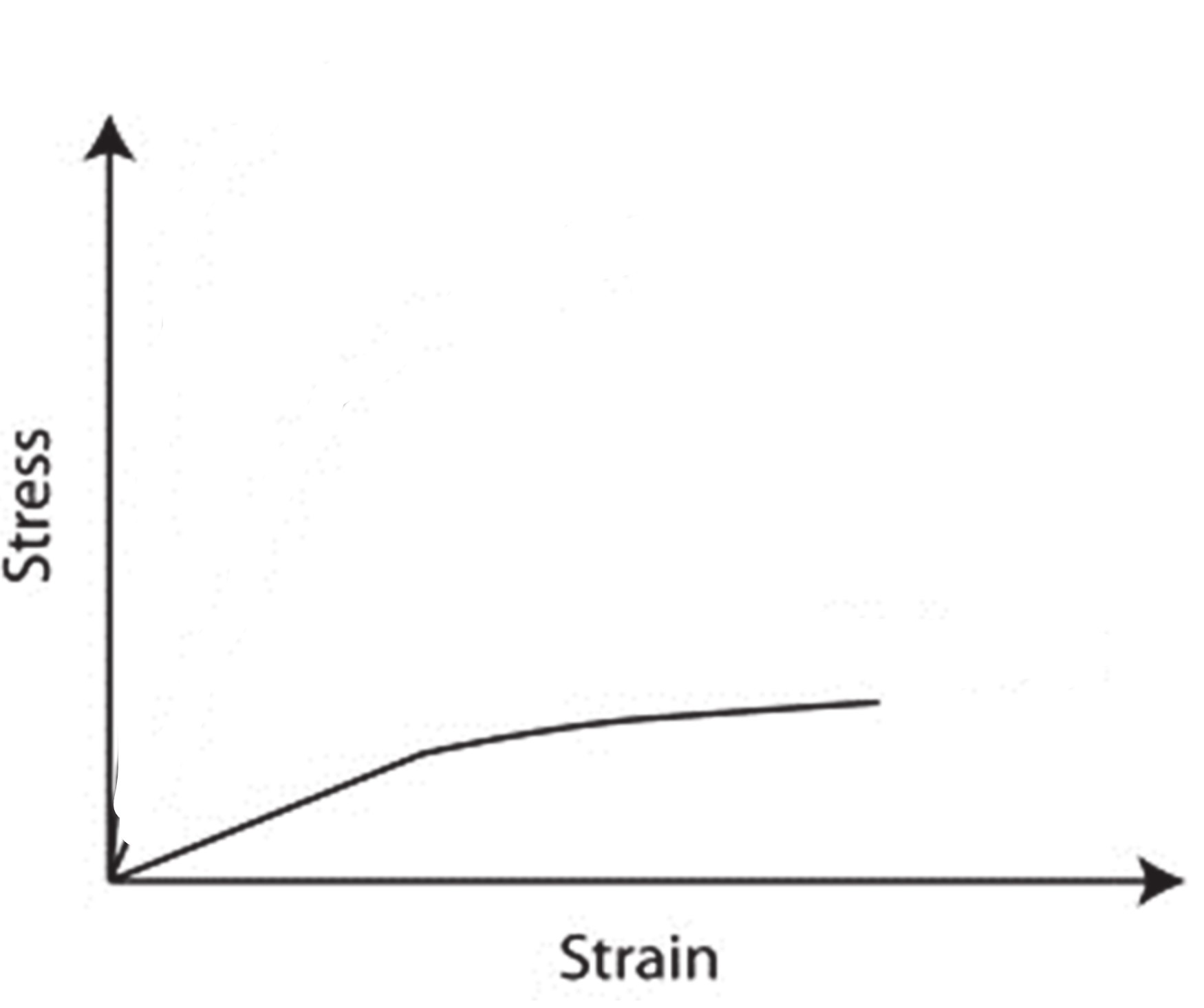
Polymer stress strain curve
24
New cards
Poisson's ratio
Ratio of lateral strain over vertical strain
25
New cards
Poisson's ratio meaning
The larger the Poisson's ratio the larger the elastic deformation of a material
26
New cards
Atomic bonding
forces that bind two atoms together
27
New cards
Repulsive forces
negative electron shells repel each other
28
New cards
Attractive force
positive nucleus with negative shell of neighboring atom attract
29
New cards
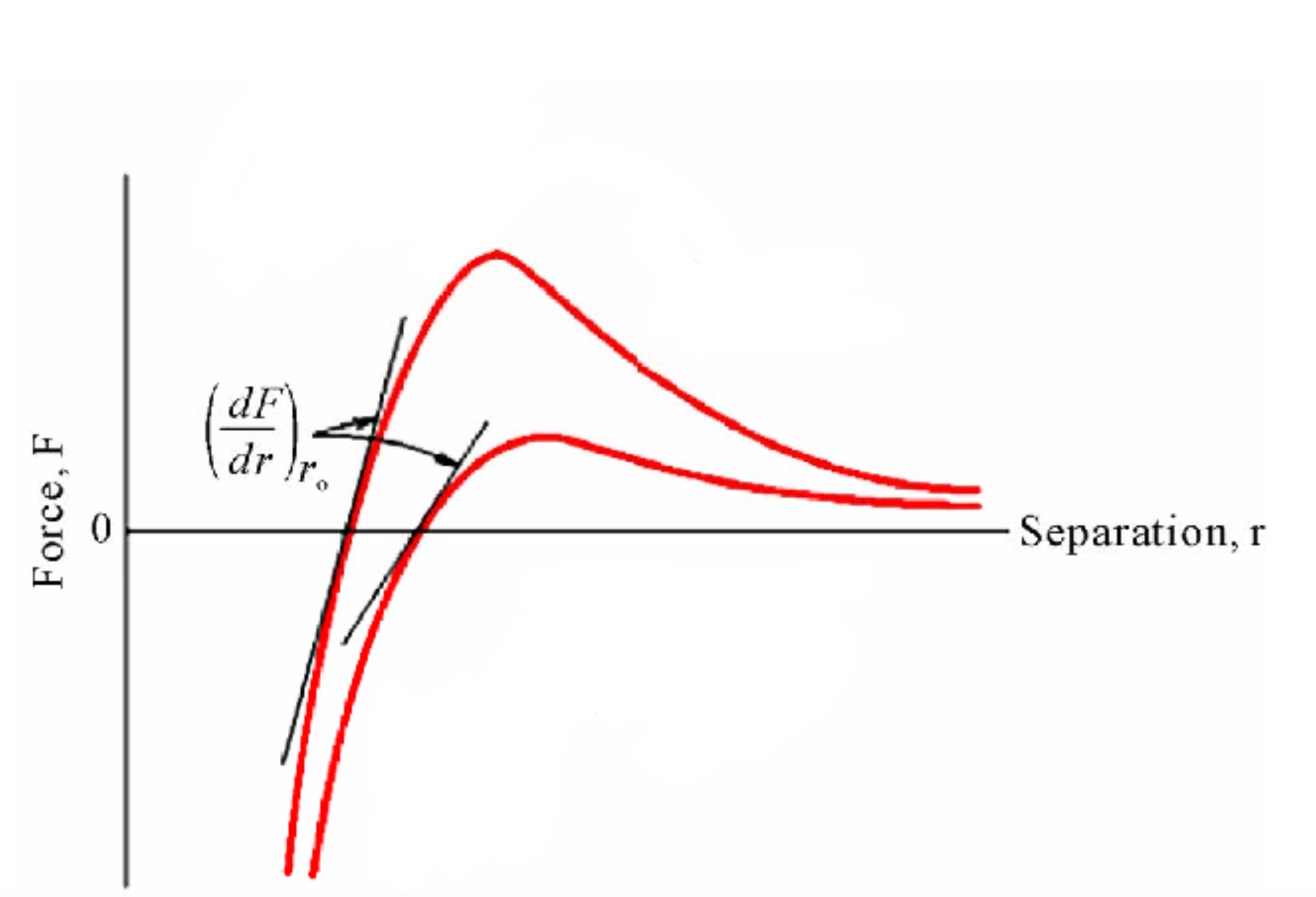
Force-distance graph
30
New cards
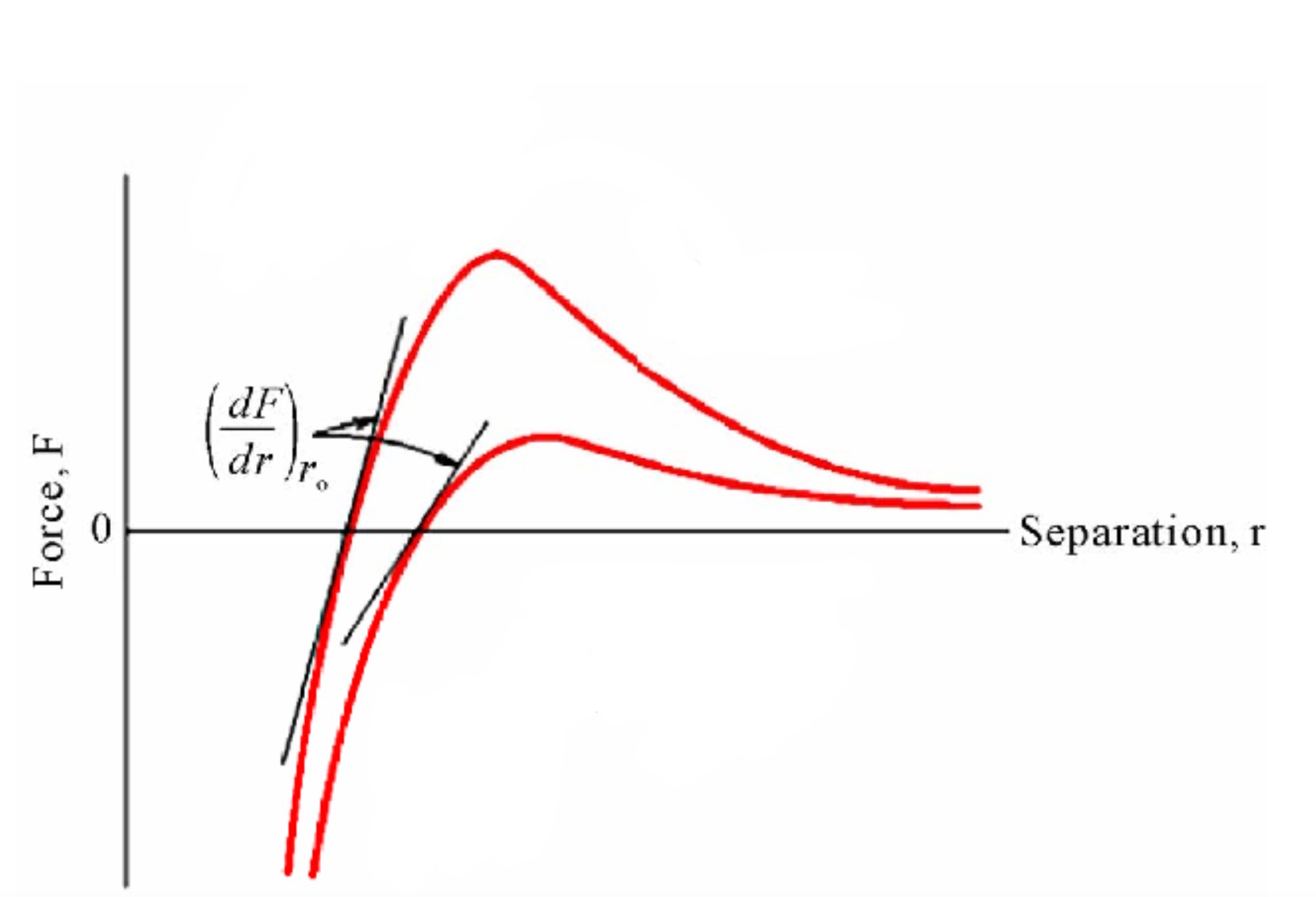
Top has a high modulus of elasticity and is strongly bonded. Bottom is opposite
Which has a higher bond strength and modulus of elasticity
31
New cards
Energy-distance graph
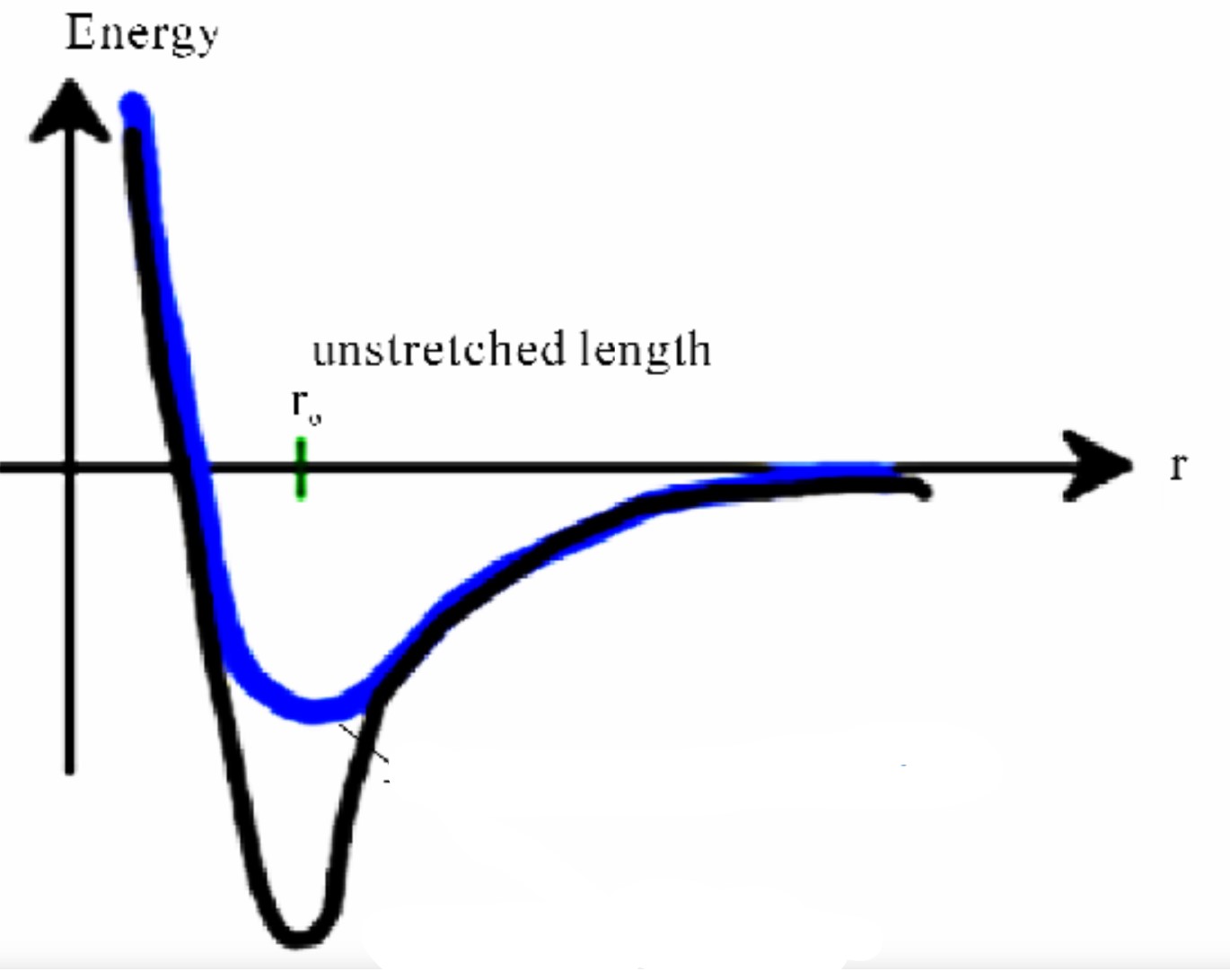
32
New cards
Blue has a lower elastic modulus and bonding strength
Which has a higher bond strength and modulus of elasticity
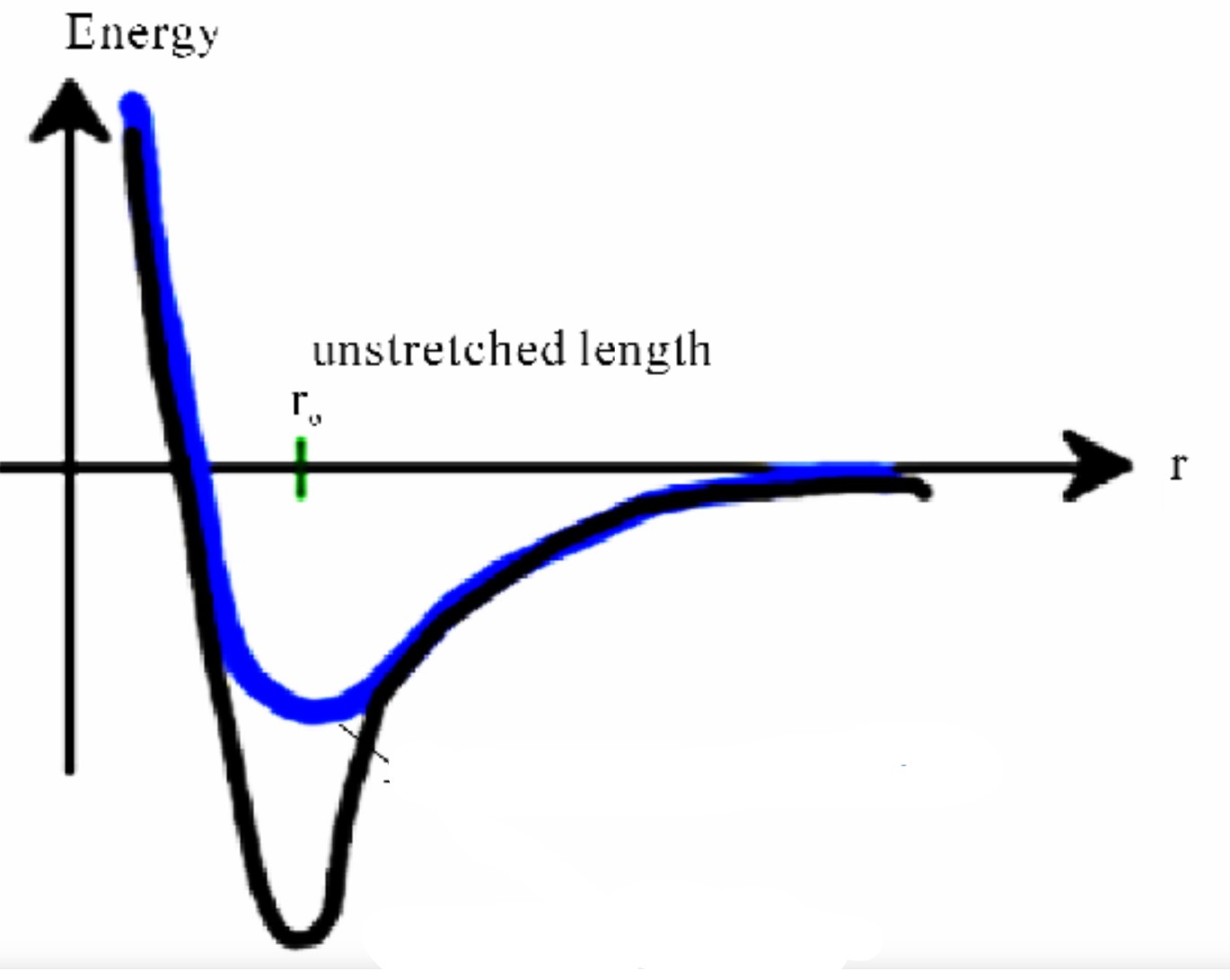
33
New cards
R_0 on energy/force-distance graph
equilibrium separation
where attractive and repulsive forces are balanced
where attractive and repulsive forces are balanced
34
New cards
Bond Energy (E_0) on energy/force-distance graph
energy at r_0
energy required to separate two atoms an infinite distance apart
energy required to separate two atoms an infinite distance apart
35
New cards
Melting temperature
melting is the separation of atoms to an infinite distance
temperature is energy
the larger the E_0 value the more energy must be added to melt
temperature is energy
the larger the E_0 value the more energy must be added to melt
36
New cards
Elastic modulus
amount of elastic deformation
37
New cards
What does a high elastic modulus mean
Little elastic deformation for high stress
38
New cards
What does a low elastic modulus mean
High elastic deformation for low stress
39
New cards
Plastic deformation
Permanent deformation of a material (will not return back to original shape)
40
New cards
Yield stress
stress at which plastic deformation begins
41
New cards
Proportional limit
Idealized transition between elastic behavior and onset of plastic deformation
42
New cards
How to determine Yield stress
used a 0.2% offset parallel to elastic deformation
43
New cards
Ultimate tensile strength (UTS)
Maximum stress on the engineering stress/strain curve
44
New cards
Ductility
The amount of plastic deformation a material undergoes in a tensile test (% elongation)
45
New cards
% reduction in area
Determines how brittle a material is
46
New cards
Brittle material
Low %RA (
47
New cards
Ductile material
High %RA (>5%)
48
New cards
How is yield strength and ductility related
as yield strength increases ductility decreases
49
New cards
Importance of TRIP steels
have alot of plastic deformation and strength
50
New cards
Energy absorbed by a material
Area underneath a stress-strain curve
51
New cards
Distribution of tensile strength in metals
narrow distribution in metals (ductile)
52
New cards
Distribution of tensile strength is ceramics
Wide distribution in ceramics (brittle)
53
New cards
what does a failure probability vs. stress plot show
the higher the slope the lower the variability
54
New cards
Factor of safety
accounts for variations in material quality
55
New cards
Hardness
measure of a material's resistance to localized plastic deformation
not a material property
not a material property
56
New cards
Hardness testing
The small indenter is forced into the material
The indentation shape is converted into a hardness value
need to obtain 3 different readings
variations in microstructure will case changes in hardness
The indentation shape is converted into a hardness value
need to obtain 3 different readings
variations in microstructure will case changes in hardness
57
New cards
Work hardening
creating plastic deformation within a material to increase yield stress and UTS but lowers ductility
58
New cards
Rockwell hardness test
measures depth of indentation
(cone for hard materials)
(ball for softer materials)
(cone for hard materials)
(ball for softer materials)
59
New cards
Vickers/Micro vickers hardness test
dimention of indentation is measured
60
New cards
Brinell hardness test
diameter of indentation is measured
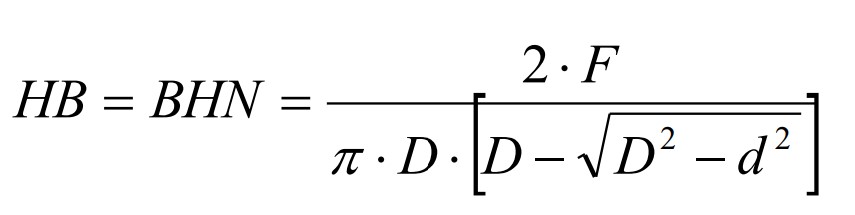
61
New cards
Uses of hardness testing
Estimating UTS
quality control
mechanical properties of small samples
changes in microstructures during processing
wear applications
W=(KFS/H)
quality control
mechanical properties of small samples
changes in microstructures during processing
wear applications
W=(KFS/H)
62
New cards
Thermal expansion and contraction
change in dimension due to temperature
63
New cards
Coefficient of thermal expansion (CTE)
magnitude of expansion
64
New cards
Expansion due to change in termperature
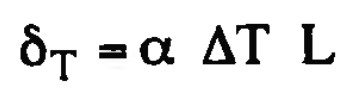
65
New cards
What affects the CTE for a material
As temperature increases bond separation increases
Elastic modulus and bond energies increase
Elastic modulus and bond energies increase
66
New cards
Thermal Stress
Thermal stresses are caused buy a constraint or two materials with different CTE
67
New cards
Constrained system
a metal rod wants to expand as temperature increases
68
New cards
Bimetallic material
The difference in CTE will cause the material to create thermal stress and expand at a different rate
Depending on the properties of each strip the material will deform when heated or cooled
Depending on the properties of each strip the material will deform when heated or cooled
69
New cards
How are CTE and same materials related
Two materials with different CTE values but at the same temperature is equivalent to the same material with different temperatures
70
New cards
Thermal shock resistance
Thermal shocks of ceramics is resistance to cracking at a sudden change in temperature
71
New cards
How can differential thermal expansion be minimized
slow cooling or heating
72
New cards
Prevention of thermal shock
choosing a ceramic with a very low CTE value
73
New cards
Thermal conductivity
rate of heat flow is proportional to the thermal conductivity (k)
74
New cards
Mechanism of heat transportation in solids
by lattice vibrations or waves called phonons
motion of free electrons
motion of free electrons
75
New cards
Metals heat transport
Free electrons are more significant than phonons
76
New cards
How is thermal conductivity related to electrical conductivity in metals
They are proportionally related
77
New cards
How does atoms affect electrical conductivity
The electron arrangement around the atom and the number of atoms that can move across the energy band
78
New cards
How is thermal energy related to kinetic energy in atoms
The thermal energy increases electron kinetic energy
79
New cards
How is heat transfer related to material purity
the more pure a material the more heat that can be transferred
80
New cards
What is the main factor to heat transfer in ceramics
Due to the lack of free electrons, the vibration/phonon mechanisms transport heat
K_l is inversely proportional to the number of scattering sites
wave scattering sites are primarily 2nd phase particles (air or contaminants)
K_l is inversely proportional to the number of scattering sites
wave scattering sites are primarily 2nd phase particles (air or contaminants)
81
New cards
Ceramics with high conductivity
Diamond, Si, BeO, AlN
82
New cards
How do polymers have heat transfer
mainly due to vibration and rotation of polymer chains
free path is very low due to disordered structure
specialized processing can reduce the disorder of the structure and henhase conductivity
free path is very low due to disordered structure
specialized processing can reduce the disorder of the structure and henhase conductivity
83
New cards
Ohms Law
V=IR
84
New cards
Resistivity
rho=RA/l
85
New cards
Electrical conductivity
rho=1/sigma
86
New cards
What is electrical current flow related to in metals
electron motion
87
New cards
what makes the electrical conductivity of metals high
Free electrons
88
New cards
Conductivity
mu is affected by imparities
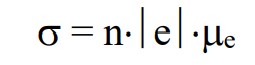
89
New cards
How does scattering centers relate to resuestivity
More scattering centers the higher the resistivity
90
New cards
What are scattering centers
-Impurities
-Vacancies
-Interstitial/substitutional atoms
-dislocations
-Vacancies
-Interstitial/substitutional atoms
-dislocations
91
New cards
what are superconductivity materials
materials with zero electrical resistance
92
New cards
High Temperature SC
materials that behaves as a superconductor above -200 degrees C
93
New cards
Type of Failues
Catastrophic failure, Fatigue, Creep
94
New cards
What causes materials to fail
-Poor material selection
-Poor design
-Environmental conditions
-Physical damage
-Poor design
-Environmental conditions
-Physical damage
95
New cards
What is fracture
-pieces splitting into two
-breaking of bonds between atoms
-breaking of bonds between atoms
96
New cards
Brittle fracture
fracture with little plastic deformation
common in ceramics
occurs suddenly
fracture surface is flat on micro-level
common in ceramics
occurs suddenly
fracture surface is flat on micro-level
97
New cards
Ductile fracture
fracture with large amount of plastic deformation
common in metals and polymers
failure does not occur suddenly
failure surface can be defined as a cup and cone with microvoids
common in metals and polymers
failure does not occur suddenly
failure surface can be defined as a cup and cone with microvoids
98
New cards
Griffith Theory
An existing crack with propagating when energy is available is equal to the energy required to form two new surfaces
99
New cards
what does a Stress concentrations allow for
the nominal stress to overcome the theoretical bonding strength
100
New cards
Effects of plastic deformation absorbs energy
plastic deformation absorbs energy