Topic 2 - Bonding
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What is an ionic bond
An ionic bond happens when a metal looses electrons and non - metals gain electrons. It is a transfer of electrons
The metal atom loses electrons and becomes a positive ion. The non-metal atom gains electrons and becomes a negative ion. The oppositely charged ions attract = ionic bond
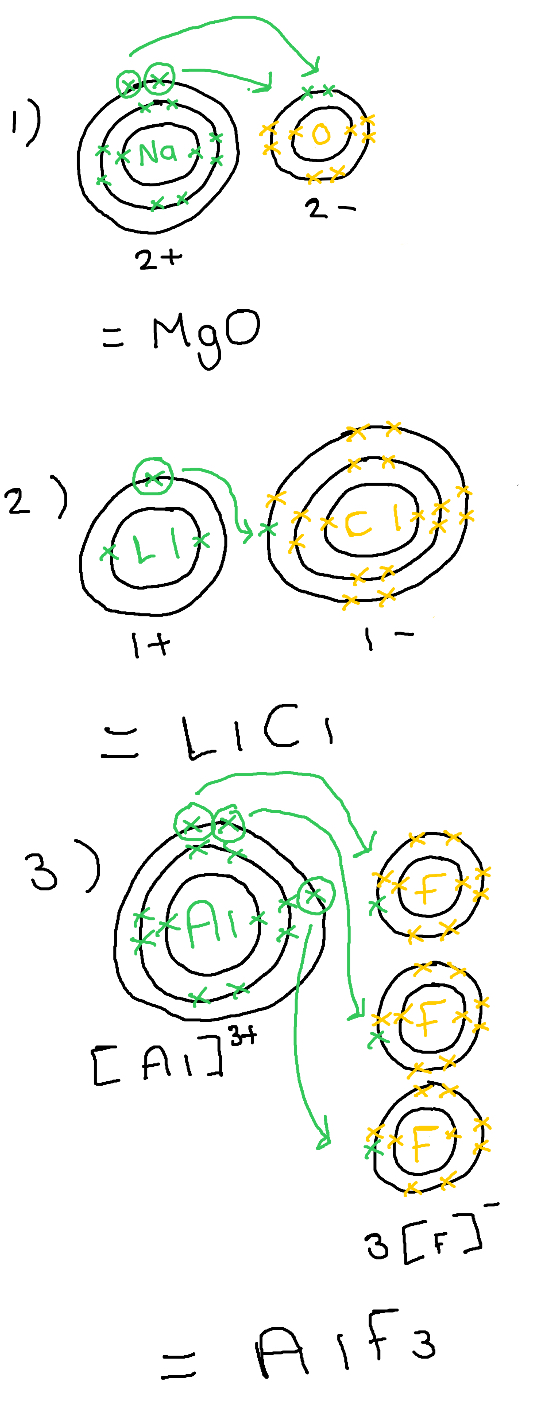
Draw electron arrangement for:
1) Mg12 + O8
2) Li3 + Cl17
3) Al13 + F9
Ions for each group
Group 1 = +
Group 2 = 2+
Group 3 = 3+
lose electrons → becomes positive ion
Group 7 = -
Group 6 = 2-
Group 5 = 3-
gain electrons → becomes negative ion
Complex/Molecular ions
Molecular ions are made of 2 or more atoms and stay together in a formula:

Work out ionic formula for:
1) Sodium (g1) Chloride (g2)
2) Sodium (g1) Oxide (g6)
3) Aluminium (g3) Oxide (g6)
4) Magnesium (g2) Hydroxide
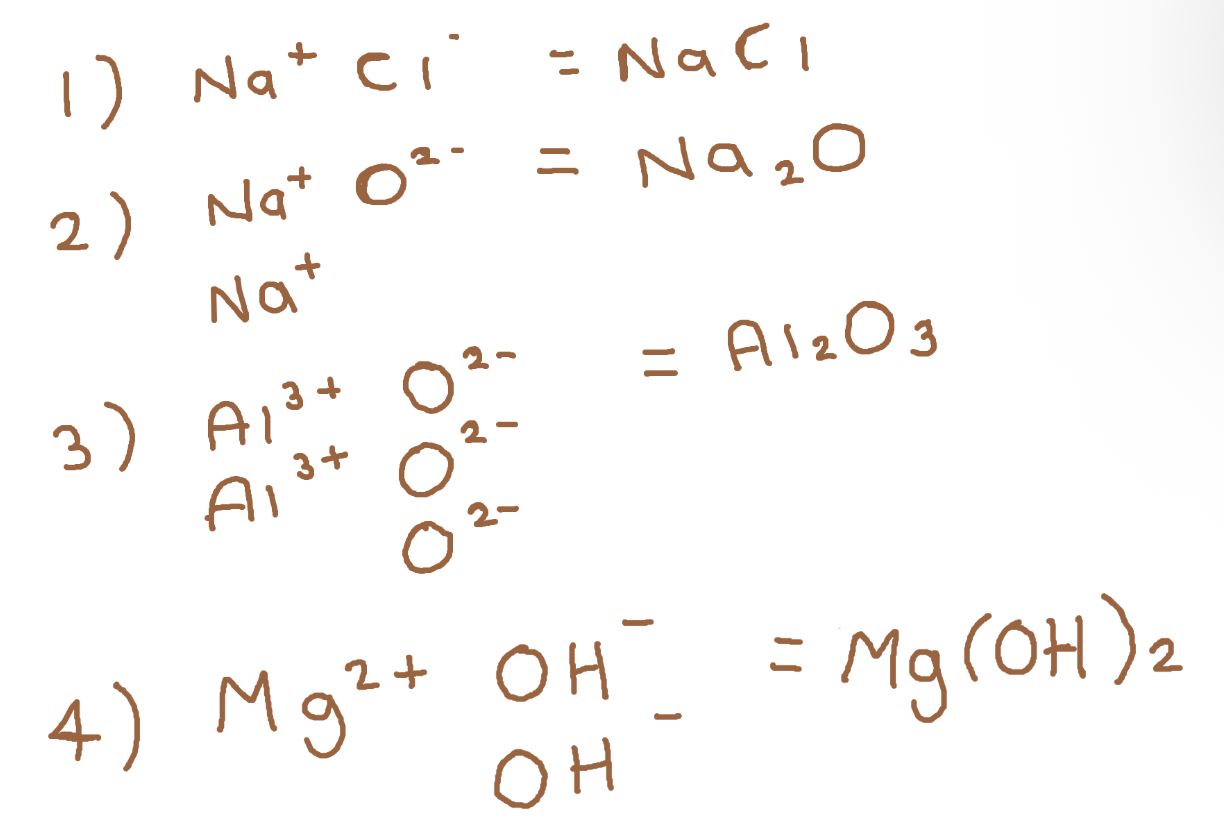
Ionic properties
1) High melting points
→ very strong (electrostatic) force of attraction between oppositely charged ions
2) Form giant lattice structure
→ crystals
3) Conduct electricity when molten or dissolved
→ ions can move + carry a charge
What do all ionic compounds form
All ionic compounds form giant lattice structures
Regular giant lattice structure
regular structure of alternating charged ions
Increases number of folles
Causes very high melting points
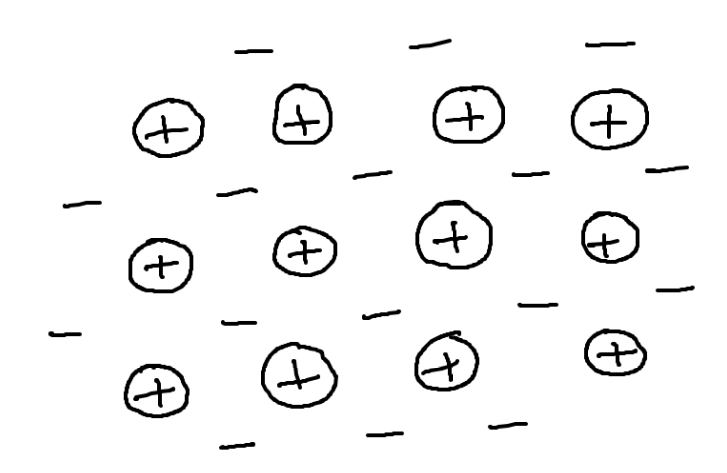
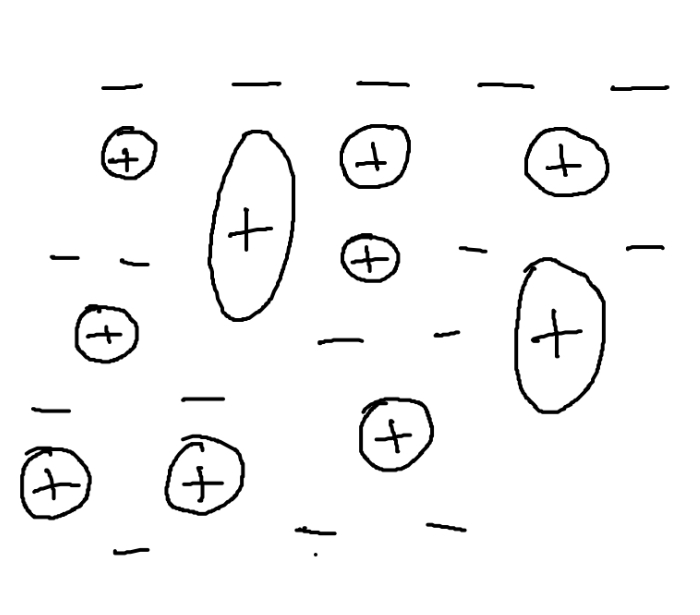
Pure metals
rows of positively charged metal ions
Surrounded by a sea of delocalised electrons
The metallic bond is caused by strong forces of attraction between + ions and - electrons
What are alloys
Is a mixture of metals
Change properties
Alloys example
Stainless steal (iron)
Brass
Bronze
Gold
Structure:
2 or more sizes of atoms
Disrupts the regular lattice structure
Metallic Bonding key properties
1) Conduct electricity
→ delocalised electrons can move and carry a charge through the structure
2) Conduct heat
→ delocalised electrons can move and transfer energy through the structure
3) Malleable/ductile
→ layers can slide/be combined
4) High mp/bp
→ opposite charges of electrons + ions
→ high (electrostatic) force of attractions
→ lots of energy required to break
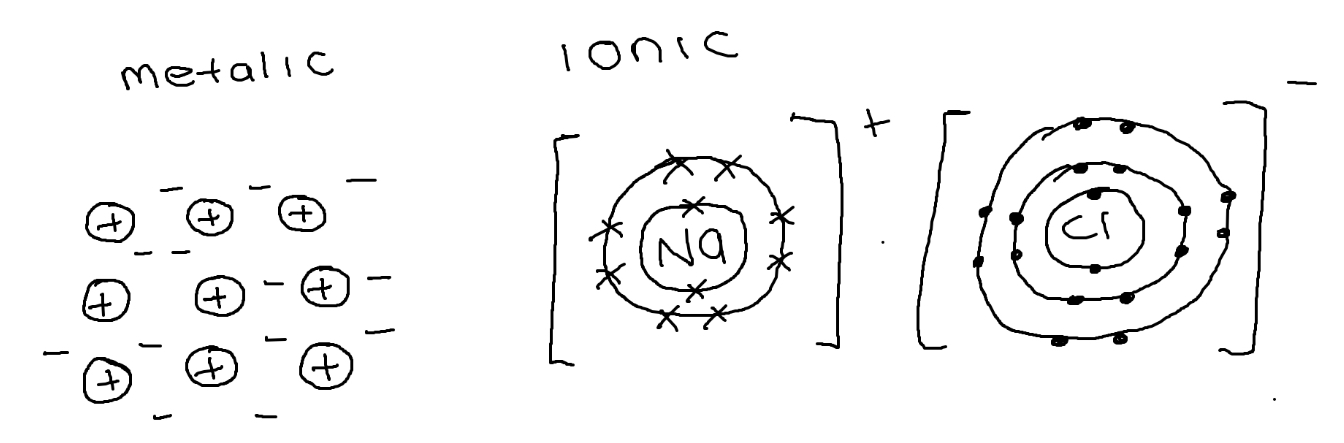
Compare the bonding of sodium (metalic) + sodium chloride (ionic)
metallic:
Rows of + ions
Sea of delocalised electrons
Strong force of attraction
Ionic:
Transfer of e- from metal to non-metal
Opposite charged ions attract
Strong force of attraction

Describe, in terms of transfer of electrons, what is happening in the reaction between sodium & oxygen
Sodium looses 2 electrons and becomes + ion, oxygen gains 2 electrons and becomes 2- ion therefore 2 sodium required per oxygen
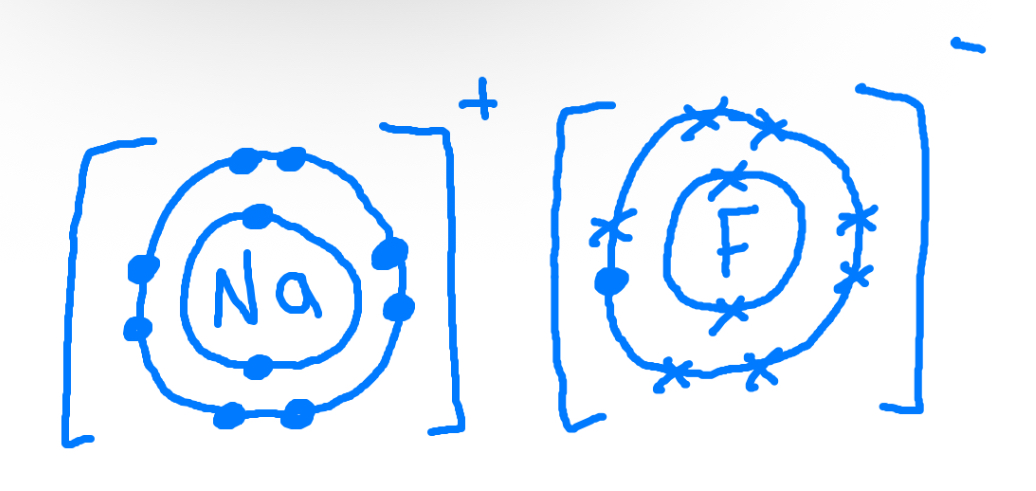
Describe, in terms of transfer of electrons, what happens when a magnesium atom reacts with a fluorine atom to form magnesium fluoride
Magnesium loses 2 electrons to become Mg2+ and fluorine gains 2 electrons to become f2- to become MgF2
Suggest why pure magnesium is a better conductor than pure lithium
Magnesium has more outer shell electrons than lithium (2 vs 1)
Therefore more electrons available to be delocalised and move/carry charge through structure
How do metals conduct electricity
Metals have delocalised electrons that are free to move throughout the structure and carry electrical charge when a potential difference is applied
Describe how sodium conducts thermal energy
sodium conducts thermal energy through the movement of delocalised electrons and the vibration of ions
Explain why steel is harder than iron
Steel I harder than iron because the carbon atoms disrupt the regular arrangement of iron atoms, preventing the layers from sliding over each other easily
How does an atom of Fe forming an Fe3+ ion occur
An atom of Fe looses three electrons from its outer shell to form a positive ion with a 3+ charge
Dot and cross diagram
Eg Sodium Fluoride
Covalent bonds
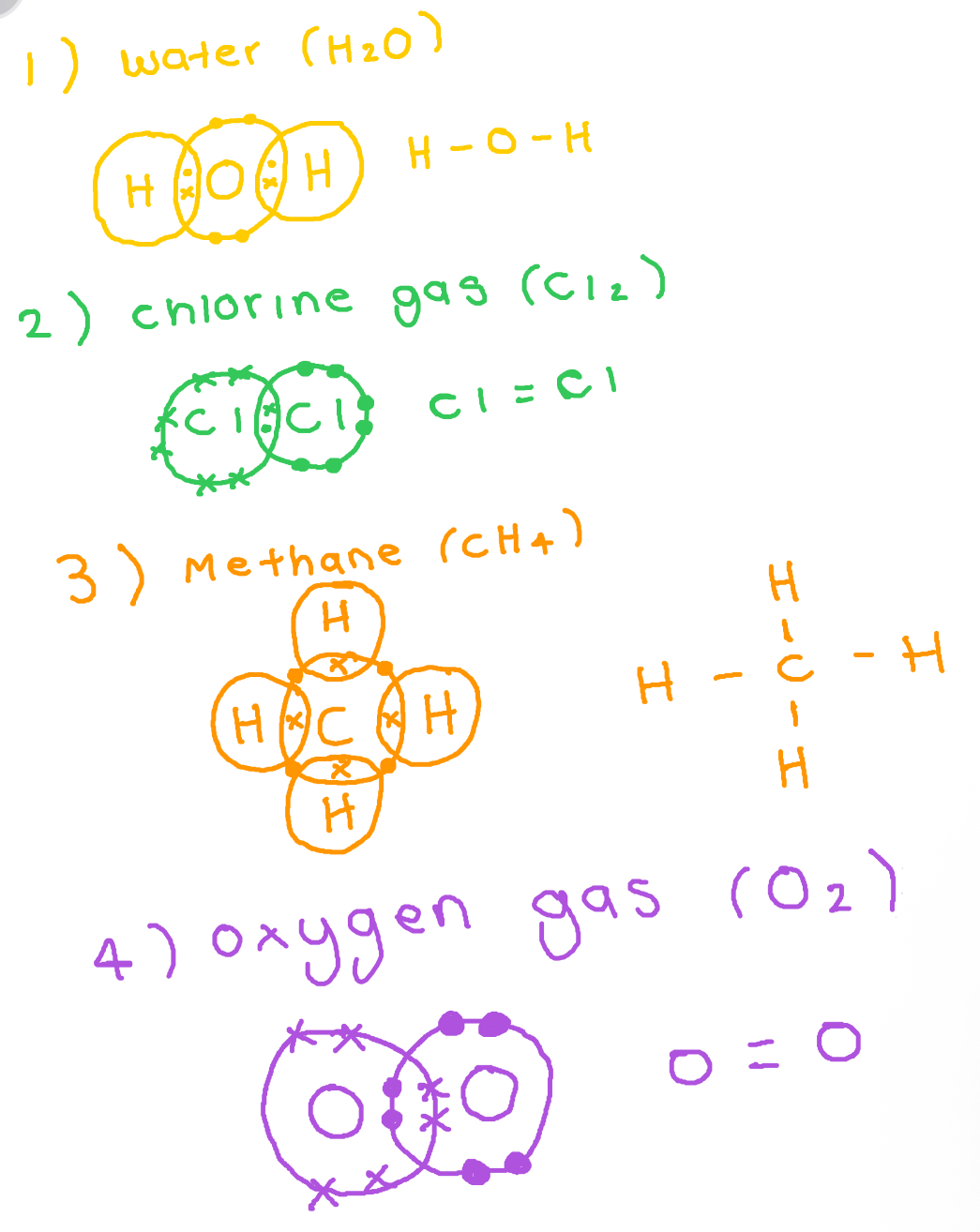
All non metal atoms want to gain electrons. How can 2 do this at the same time
By sharing electrons
Properties of covalent bonding
1) Do not conduct electricity
2) Have low mp/bp → weak intermolecular forces between molecules (imfs) we break the imfs not the strong cov bond
H-H /// H-H
Weak imf
Strong bond
Giant covalent molecules
Most covalent molecules are small/simple however, carbon can form a variety of giant structures called allotropes
The two most common are diamond and graphite
Diamond and Graphite
Diamond:
Each carbon bonded to 4 other carbons
Incredibly strong as carbuns cannot move
High mp
Shiny/lustrous
Does not conduct electricity
Use it for jewellery + tools
Graphite:
Each carbon bonded to 3 others
Forms layers with week imfs holding them together
Layers can slide
Has delocalised electrons → conducts electricity
Use it for pencils
Compare the bonding and structure of diamond and graphite
Diamond is bonded to 4 other carbons where as graphite is bonded to 3 others
Diamond has a crystalline structure so each carbon is held in place where as graphite has layers for structure
Diamond has high mp, does not conduct and is strong where as graphite has high mp (lower than diamond), does conduct because it has free electrons and is soft
Other carbon allotropes
Graphene = a single layer of graphite
→ 1 atom thick (can see through it)
→ each carbon bonded to 3 others
→ very strong
→ conducts electricity due to delocalised electricity
Nanotube = rolled up graphene
Buckyball (C60)
(Buckyminster Fullerene)
→ alternating pentagon/hexagons
→ football
→ personalised medicine
What is nanoscience
The study of particles between 1-100 nm
How small is 1nm

Why use nano particles
Very large SA:V
Can use more for less cost
Can cost something without being visible → suncreme
Limitations of ionic bonding models - 2D & 3D space filling models:
2D & 3D space filling models:
Both only show a few ions & not the huge number that are in a giant lattice
3D only shows the outer layer of ions

Limitations of ionic bonding models - Ball and stick giant lattice model:
Ball and stick giant lattice model:
The ions are shown for apart when they are actually closely packed
The sticks/lines could be confused with covalent bonds

Limitations of ionic bonding models - Dot & cross diagram:
Dot & cross diagram:
Seem to show that there are only pairs of ions instead of the huge numbers found in a giant lattice
Do not show relative sizes of the ions

Limitations of covalent bonding models for molecules - Molecule 2D diagrams:
Molecule 2D diagrams:
Do not show the shape of the molecule in 3D
Do not show which atoms the electrons in the bond have come from

Limitations of covalent bonding models for molecules - Molecule dot & cross diagrams:
Molecule dot & cross diagrams:
Do not show relative sizes of the atoms
Do not show the shape of the molecule
