Atoms, compounds, molecules
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
What is an electron, proton and neutron relative mass
Electron - 0.005, Proton - 1, Neutron - 1
What is atomic number
The number of proton in the nucleus of an atom
What can we use the atomic number for (3 things)
Work out the number of electrons
Identify the element
Gives position of element in periodic table
What is mass number
The total number of proton + neutron in the nucleus of an atom
What are isotopes
Isotopes are atoms which have the same number of protons but different number of neutrons. They are the same element but they have different masses.
Relative isotopic mass is the mass of an isotope relative to […] of an atom of […].
Relative isotopic mass is the mass of an isotope relative to 1/12th of an atom of carbon-12.
A […] ion (Cation) has […] one or more electrons.
A […] ion (anion) has […] one or more electrons.
A positive ion (Cation) has lost one or more electrons.
A negative ion (anion) has gained one or more electrons.
Why do we use masses and moles
Atoms are weighed by comparing their masses to each other.
Atoms are counted by using the moles - an amount that has a definite mass and contains a fixed number of particles.
Why is carbon-12 used as the standard for relative masses?
Easy to get/abundant
Stable
How does the mass of a magnesium atom compare to the mass of carbon-12 atom?
A magnesium atom is approx twice as heavy as a 12C atom.
For an isotope the relative mass will be approximately […] as its mass number
For an isotope the relative mass will be approximately the same as its mass number
What is the approx relative mass for zinc with mass no. of 65 and Pb with a mass no. of 206
Zn - 65.000
Pb - 206.000
Which group has +1 charge
group 1
Relative Isotopic mass
the mass of one isotope compared to one twelfth of the mass of one atom of carbon-12
Relative molecular mass
the average mass of a molecule compared to one twelfth of the mass of one atom of carbon-12
Why might isotopes have slightly varying physical properties
Because they have different masses
How do you get number of neutrons from the mass and atomic number
Number of neutrons = mass number (A) - atomic number (Z)
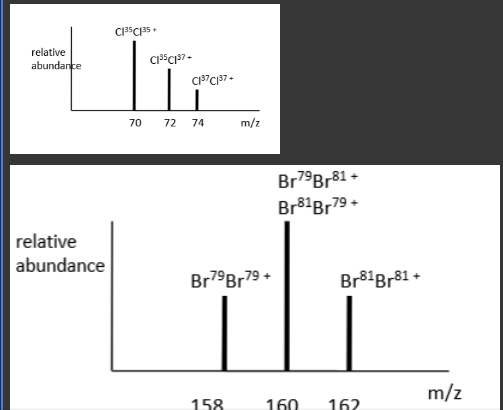
Why does the following spectra appear like this?
Because Cl and Br have two isotopes :
Cl35 (75%) and Cl37 (25%)
Br79 (50%) and Br81 (50%)
What is the atomic number
The atom number, Z, is the number of protons in the nucleus. It is also the number of electron since an element has no overall charge.
Where is a electron, what it is relative mass and relative charge
Orbitals/shells, mass of 1/1800 and charge of -1
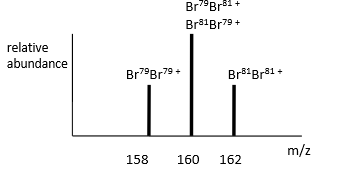
why is 160 m/z so high
The 160 peak has double the abundance of the other two peaks because there is double the probability of 160 Br79-Br81+ as can be Br79-Br81 and Br81-79.
What are Isotopes
Isotopes are atoms of same element with the same number of protons, but different number of neutrons.
what is the mass number
mass number, A, is the total number of protons and neutrons in the atom.
Relative atomic mass
the weighted mean mass of one atom compared to one twelfth of the mass of one atom of carbon-12
-If sodium ion has a plus 1 charge and has a atomic number of 11, Total positive charge of protons will be […] and the total negative charge of electron will be […].
-Just like chlorine with a charge of minus 1 with a atomic number of 17 would have a total positive charge of protons of […] while total negative charge will be […]
-If sodium ion has a plus 1 charge and has a atomic number of 11, Total positive charge of protons will be +11 and the total negative charge of electron will be -10.
-Just like chlorine with a charge of minus 1 with a atomic number of 17 would have a total positive charge of protons of +17 while total negative charge will be -18
Where is a proton, what it is relative mass and relative charge
Nucleus, mass of 1 and a +1 charge
What are the equation for Relative atomic mass of an element
.
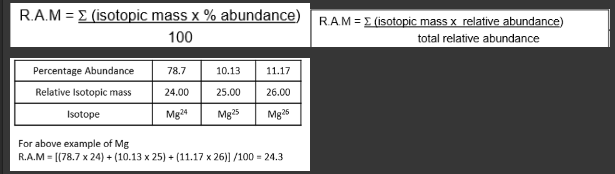
As - for an isotope the relative mass will be approx the same as the mass number.
-Each proton + neutron have a mass of […] atomic mass unit
-we are assuming that the electrons contribute […] mass to the atom mass unit.
As - for an isotope the relative mass will be approx the same as the mass number.
-Each proton + neutron have a mass of 1 atomic mass unit
-we are assuming that the electrons contribute zero mass to the atom mass unit.
The mass of an atom is equal to the number of the […]
The mass of an atom is equal to the number of the nucleons
Where is a neutron, what it is relative mass and relative charge
Nucleus, mass = 1 and charge of 0
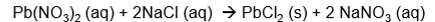
How do you turn full equations to ionic equation using this equation as an example
.

Spectator ions
ions that are not :
Not changing state
Not changing oxidation number
Why do we write ionic equations?
We usually write ionic equations to show the key
changes in a reaction. lonic equations only show the
ions that are reacting and leave out spectator ions.
Find the formula of calcium phosphate
.

How do you work out the formula from the ionic charge use lithium sulfate has an example
.
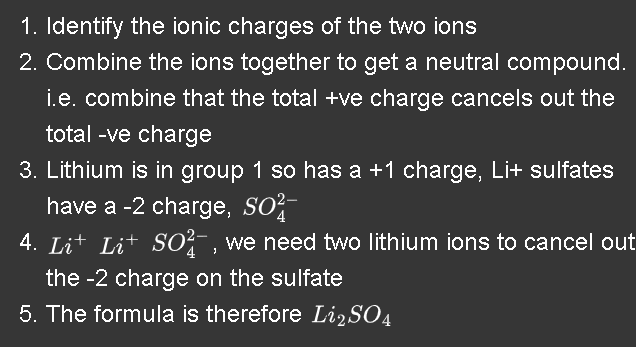
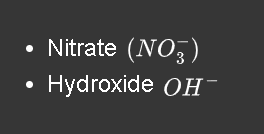
which inorganic compounds have a charge of -1
.
which group have a charge of -1
group 7
which inorganic compounds have a charge of -2
.

what charge does group 6 have
-2
what inorganic chemical has a charge of -3
.

what charge does group 5 have
-3
what transition metal have +3 charge
Iron (III)
What charge do group 3 have
3+
what transition metals have +2 charge
Copper (II)
Iron (II)
What is the charge for group 2
+2
which inorganic compound do you need to know has a +1 charge
.
