Unit 2 Gen Chem 1 Review
1/37
Earn XP
Description and Tags
Taken from the Prof Jones Notes only lol
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Molecules
Matter made up of particles called atoms
Element
Type of matter composed of one kind of atom with each having one idenitial properties
Atoms
Small particles that retain their identity in chemical reactions
Compound
Contains atoms from 1 or 2 elements in fixed porpotions
Law of defitinity porportions
elements in fixed ratio to one another based on mass in a compound.
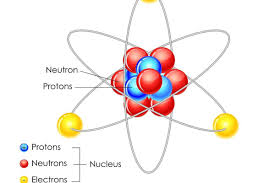
Nucleus
The center of the atom that contains protons and neutrons.
Electrons
The outer sphere of the atomic structure
What’s the charge of the electrons
Negative
Charge of Protons?
Positive
Charge of Neutrons?
Neutural charge
Nuclide
Atom characteized by definite atomic # and mass #
Atomic number
number that identifies specific elemtns and number of protons in nucles of atom
atomic structure
Isotropes
Atoms of some elements but different # of neutrons in nucleus
Atomic atomic mass (amu)
weighed mass of all naturally occuring isotrope
Peroid
Horitontal row, theres 7 of them
Group
Vertical column, theres 18 of them
Allotropes
Forms of the same element that differ in their phyiscal and chemical properties
ionic
metal +non metals
Molecular
2 or more nonmetals
Aqueous
Dissolved in water
Binary
Contains 2 elements
Ternary
Contains 3 or more elements
Acid
Contains hydrogen # one non metal
Oxyacid
Contains hydrogen, a non metal , & oxygen
Cation
Postivitvely charged ion, loses electons
Anion
negatively charged ion
Monoatomic
Charged particle from a single atom
Polyatomic
Charged particle of 2 or more atoms
Chemical formula
notation using atomic symbols w/ subscripts to convey relative porportions of elements
Hydrates
compounds containing a specific amount of water chemically bound
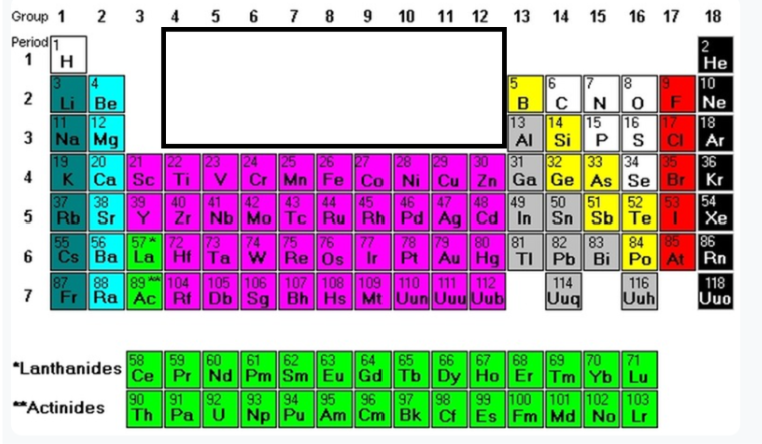
Group1 (minus H)
Alkiali metals
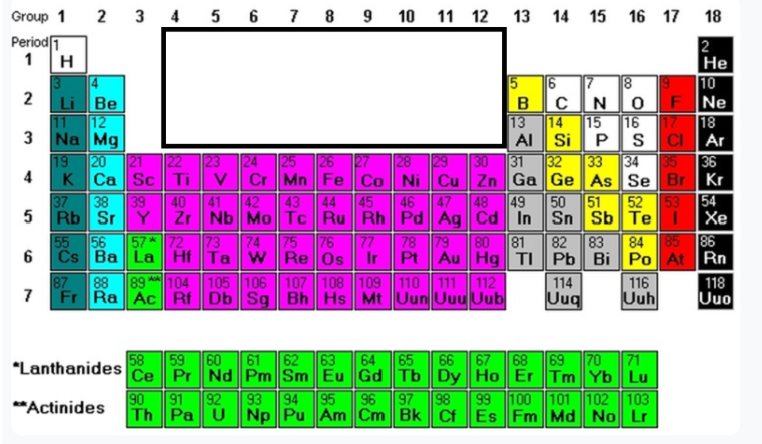
Group 2
Alkiline metals
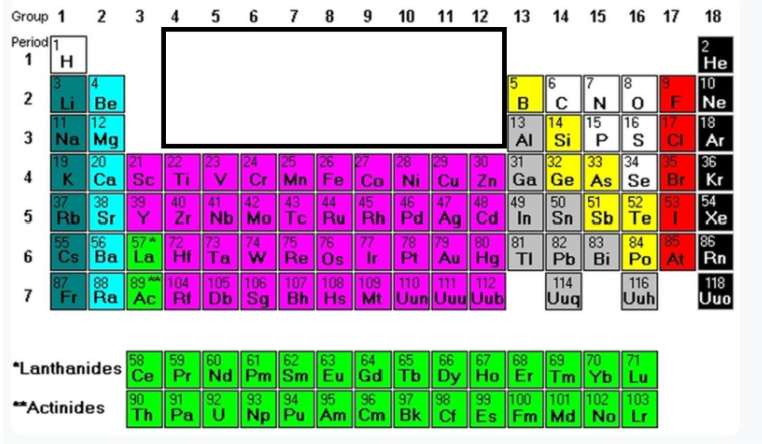
Group 3-12 (minus La & Ac)
Transition metals
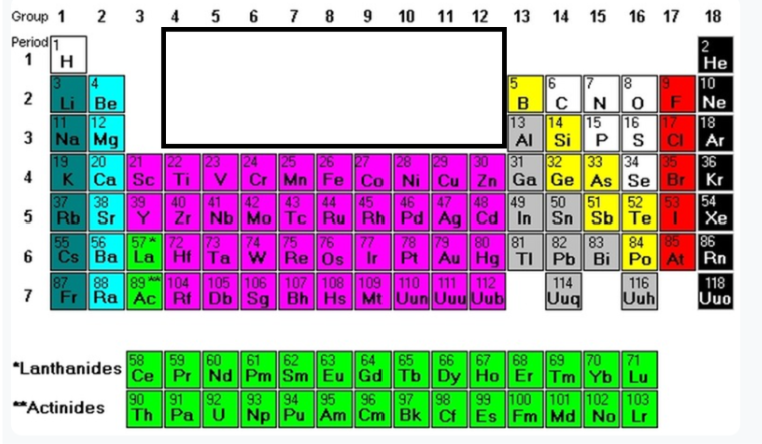
Group 13-16 (Yellow Part)
Metalloids
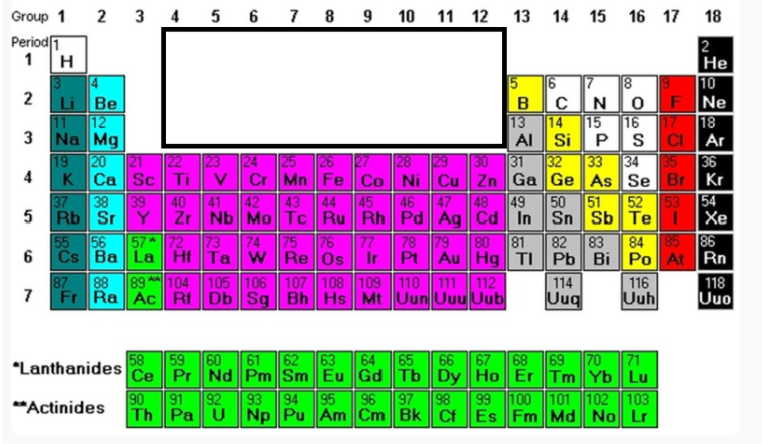
Group 14-16 w/ H (White Part)
Non metals
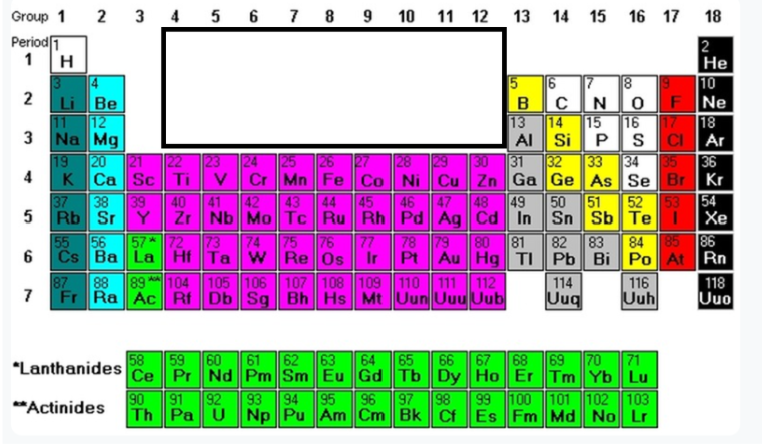
Group 17
Halogens
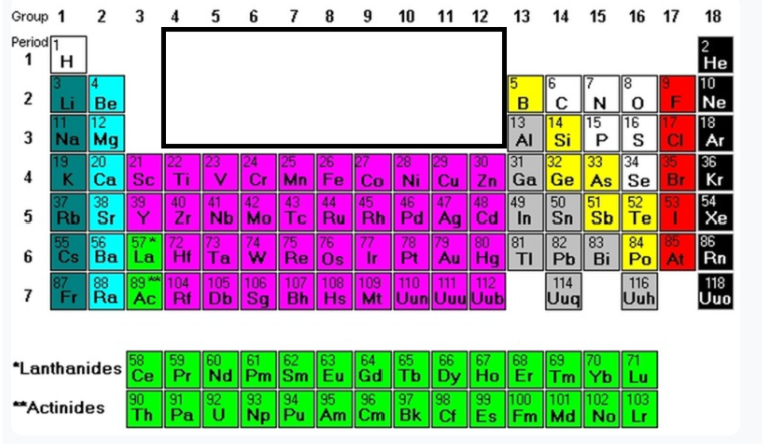
Group 18
Noble Gases