Chem Winter Q
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
solvent
solution component that is the greatest amount
solute
any component in a solution that isnt the solvent
proof is
defined as 2x the precent of alcohol
80proof= 40%alcohol and 60% water
Strong acids
So I Brought No Clean Cloths
H2SO4 (sulfuric acid)
HI (hydroiodic acid)
HBr
HNO3
HCl
HClO4
Strong Bases
Can't Stand Rich Bad Liars Now
(Ca, Sr, Rb, Ba, Li, Na).
Ca(OH)2
Sr(OH)2
Rb(OH)2
Ba(OH)2
LiOH
NaOH
Particles that all acids produce
Hydronium ions
what does a base release when dissolved in water
OH-
What does a acid release in water
H+
Strong electrolytes
nearly 100% dissociated into ions
conduct current efficiently
high electrical conductivity (bc of free moving ions)
high solubility
Salts (ionic bonds)
strong bases
strong acids
How do you find the limiting reactant
which ever element has the least moles
when asked to find mole of concentration(=molarity) use limiting reactant moles
How to identify base
OH
NH3
how to identify acid
starts w H
has carboxyl COOH
O.N for F, H, O, group 7, 6, 5
F= -1
H=+1
O=-2
7= -1
6=-2
5=-3
if its a diprotic acid (pure element) its always going to be 0
OIL RIG
oxidation is loss
reduction is gained
Ammonium
NH4 +
Acetate
CH3COO- OR C2H3O2-
Carbonate
CO3-2
Bicarbonate/Hydrogen carbonate
HCO3-
Hydroxide
OH-
Nitrite
NO2-
Nitrate
NO3-
Chromate
CrO42-
Dichromate
Cr2O72-
Phosphate
PO43-
Hydrogen phosphate
HPO42-
Dihydrogen Phosphate
H2PO4-
Ammonium
NH4+
Hypochlorite
ClO-
Chlorite
ClO2-
Chlorate
ClO3-
perchlorate
ClO4-
permanganate
MnO4-
Sulfite
SO32-
Hydrogen Sulfite/ Bisulfite
HSO3-
Sulfate
SO42-
Hydrogen Sulfate/ bisulfate
HSO4-
Cyanide
CN-
Peroxide
O22-
Properties of Gas
neither definite shape nor volume
fills container
exerts pressurfe on surroiundings
volume changes w temp and pressure
mixes completely with other gases
much less dense than solids and liquids
KMT
Particles in Motion: Gases consist of numerous tiny particles (atoms or molecules) in continuous, random, straight-line motion.
Negligible Volume: The volume of the gas particles themselves is insignificant compared to the total volume of the container.
No Intermolecular Forces Particles exert no attractive or repulsive forces on each other (except during collisions)
Elastic Collisions: Collisions between particles and container walls are perfectly elastic, meaning kinetic energy is conserved.
Temperature & Kinetic Energy: The average kinetic energy of the particles is directly proportional to the absolute temperature (Kelvin).
Average Kinetic energy of Gas molecues
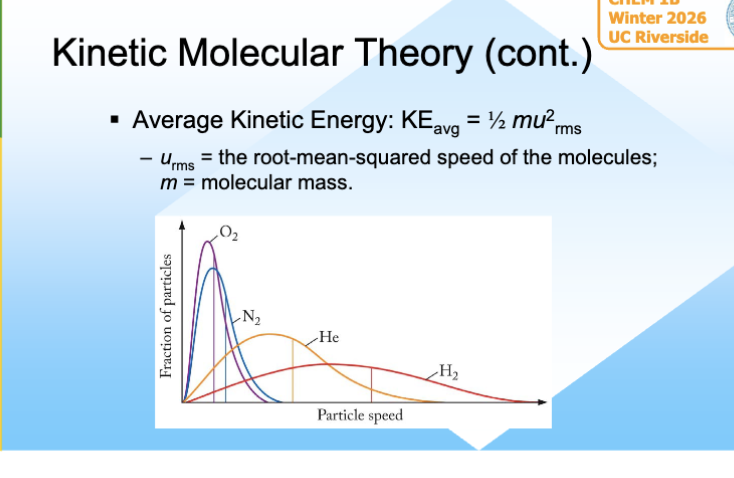
Parameters Affecting Gases
Pressure
volume
temp
number of moles
effusion
process where gas escapes from a tiny hole into a region of lower pressure
Graham’s Law of Effusion
rate of effusion of gas is inversely proportional to square root of its molar mass
if particles of gas A have higher speeds than B then gas A particles collide more frequently w the walls of the container→ inc effusion
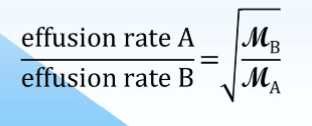
Diffusion
the spread of one substance through another
odors
mean free path
avg distance a particle can travel through air before colliding w another particle
Atmospheric Pressure
Pressure: ratio of force F to surface area A
P=F/A
Barometer
Instrument used to measure pressure
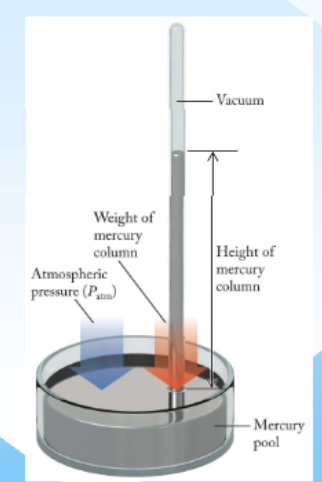
how do barometers work
using mercury to measure pressure
High pressure: when atmospheric pressure inc (heavier air) it pushes harder on the dish, forcing mercury levels up the tube
low pressure: when pressure dec, theres less downward push, and mercury in the tube falls back down into the reservoir
the height in mm indicates current air pressure, higher #=higher pressure

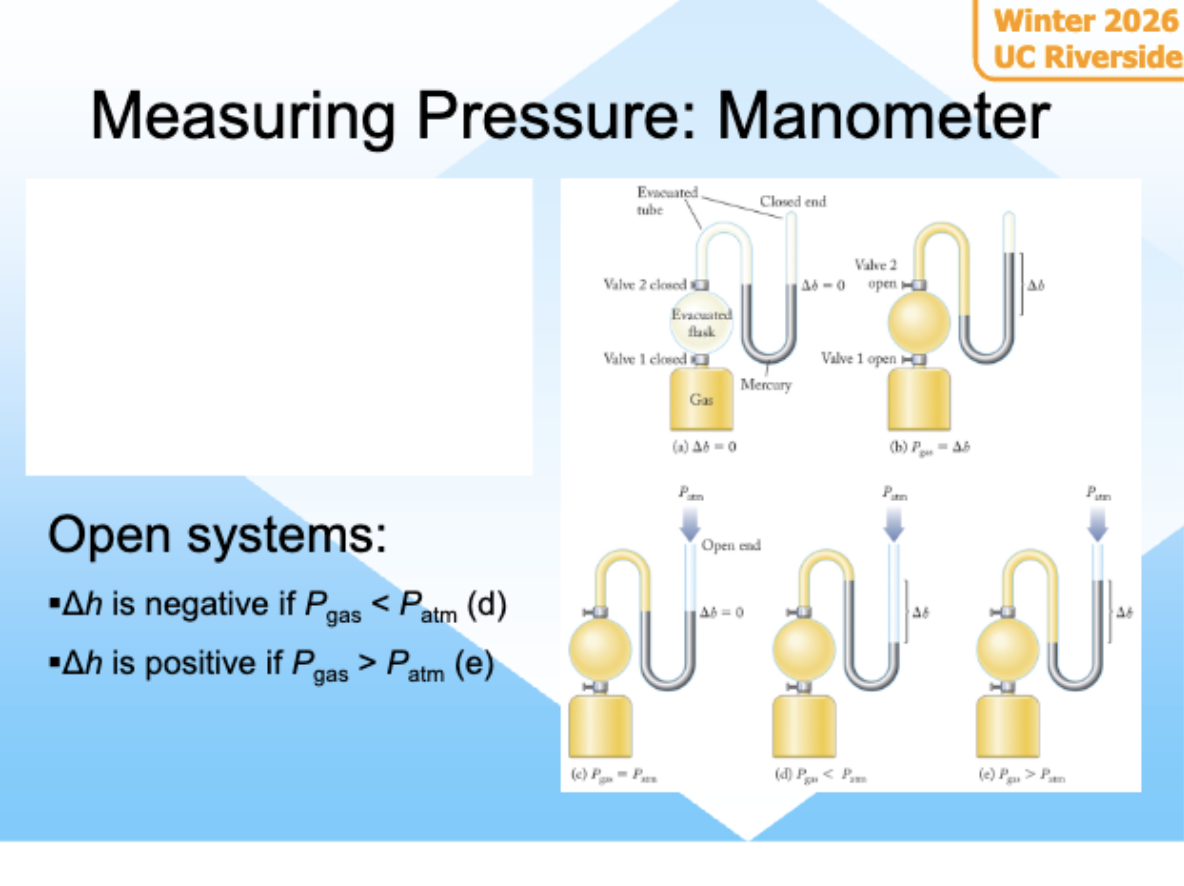
Manometer
Using water to measure pressure or any liquid
u tube contains water, pressure differences pushes liquid and the height differences shows pressure
Precipitation reactions
a reaction where dissolved substances react to form a solid product
double displacement reaction
Method for writing complete and net ionic equations
use solubility rules to deternine physical state
separate all aqueous ionic compounds into individual ions to obtain complete ionic equation
remove sperctator ions from the complete ionic equation for net ionic equation
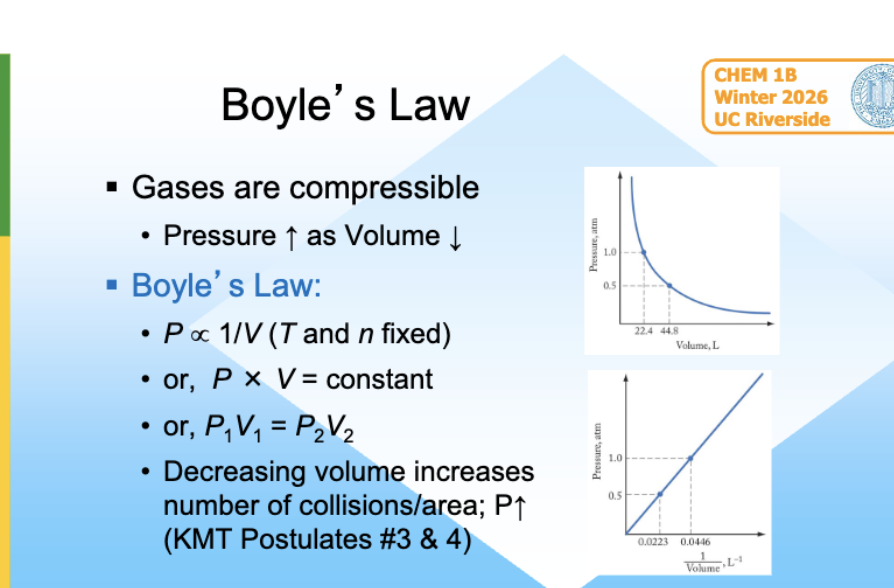
Boyle’s Law
gases are compressible
pressure inc as Volume dec
dec volume inc collesions/area $
Charles’ Law
Avogadro’s Law
Amonton’s Law
combined gas laws

Balancing redox in acidic solution steps
find ON to determine oxidation and reduction
balance other atoms than O & H
balance O by H2O
balance H by H+
balance charges by adding e-
make # of e- = and add half reacdtions
change in oxidation states results from gain/loss if e-
How to determine which gas will effuse the fastest
lowest molar mass
grahams law
Atmospheric pressure
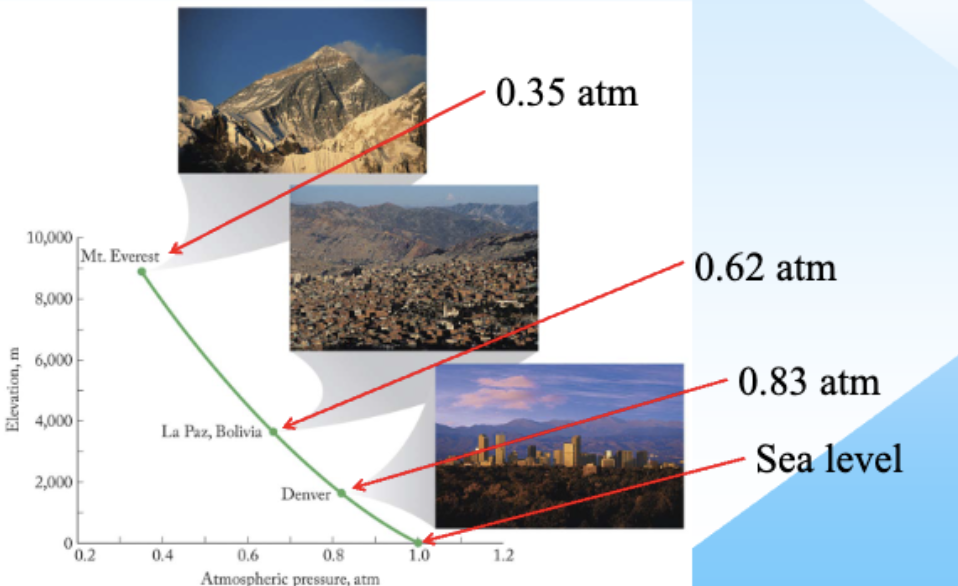
Standard points for Ideal Gases
P= 1 atm
T= 273K
V=22.4L
Precipiation Rules
insoluable

Reducing agent
the species that gets oxidized (loses electrons).
Electrolyte
solute that produces ions in solution, allows solution to conduct electricity
weak electrolytes
only partial disassociation
slightly conductive but poor conductivity
disassociation is reversible
weak bases, weak acids, and insoluble compounds
Non-electrolytes
substance w/no ionization occurs, no conduction of electrical current
dissolve as neutral molecules, not ions
covalent bonds not ionic
sugar, ethanol, urea
Brønsted-Lowry theory
defines an acid as a proton (H+) donor and a base as a proton acceptor
H2O is an acid and base
molecular equation
reactants written as undisassociated molecules (are whole and not separated)
Overall ionic equation
distinguishes between molecular and ionic substances, all elements are seperated
net ionic equation
where spectator ions are removed from ionic equation
Neutralization
reaction where acid reacts w/base profucing salt and H2O
salt
product of neutralization reaction
cation base +anion acid
HCl + NaOH → NaCl + H2O
Titration
analytical method to determine the concentration of a solute in a sample in a sample by reacting it w/ a standard solution
standard solution: solution w/ known concentrations (titrant)
Saturated Solutions & Supersaturation
aqueous solubility of most solids inc w/temp
saturated solution
solution that contains the max amount of solute possible at a given temp
unsaturated solution
solution that contains less than the max quantity of solute
Supersaturated solution
holds more solute than normally possible by heating it and cooling it slowly, making it unstable
ideal gas
in an ideal gas the individual particles of the gas don’t interact
First law of thermodynamics
energy cannot be created/destroyed only change form
total energy of universe is constant
spontaneous
once started occurs w/o outside interaction
non-spontaneous
energy is need for reaction to occur
Second law of Thermodynamics
entropy of the universe inc in any spontaneous process
colligative properties
characteristics of a solution that depend on the ratio of the number of solute of the number of solute particles to solvent particles
NOT IDENTITY OF PARTICLES
changes
freezing point depression
boiling point elevation
osmotic pressure
vapor pressure lowering
van’t Hoff factors
i= how many molecules the element disassociates into
non electrolytes
sugar: glucose, sucrose, fructose
alcohol: ethanol, methanol
urea
protein and polymers
reverse osmosis
Desalination
process that removes most ions from seawater
distillation
reverse osmosis
purification where solvent is forced through semipermeable membranes leaving dissolved impurities behind
Vapor pressure
in a closed container molecules at surface of the liquid are constantly escaping into gas phase (evaporation) & crashing back into the liquid (condensation)
when rate of evaporation = rate of returning gas → equilibrium
pressure exerted by gas molecules at equilibrium is vapor pressure
adding non volatile solute to pure solvent
when adding a nonvolatile solute (sugar/salt) to a pure solvent (water) vapor pressure drops
this is because the entire surface of the water WAS pure water and the molecules were ready to jump in the air
in the solution the surface spots are now taken up by solute particles → less solvent molecules can jump into the air→ lowers VP
factors that affect vapor pressure
temp
suface area
intermolecular forces
stronger forces = higher kinetic E needed to enter gas phase
Volatility
how easy a substance turns into a gas (vapor) at a given temp → bonds hold together
Volatile substances
have HIGH VP bc molecules are weakly attracted to each other and doesn’t take much energy for them to break bonds into a gas
characteristics
evap quickly at room temp
low boiling point
alcohols
liquid gases
sublimation
non volatile substances
LOW VP
molecules/ ion are held together by strong IMF and its difficult for them to become gases at room temp
characteristics
doesn’t noticeably evap at room temp
usually odorless
high boiling point
table salt, sugar, oils
ideal solutions
used for raoult’s law
attraction between different molecules solvent-solute is exactly the same as the attraction between the molecules of solvent-solvent
BUT if molecules repel/attract eachother you get deviations
positive deviation
molecules repel each other →escaoes easier→ higher pressure than predicted
solute-solvent interactions are weaker than solvent-solvent/solute-solute bonds
negative deviation
molecules stick together strongly → escape less easily→ lower pressure than predicted
solute solvent interactions are STRONGER than solvent-solvent/ solute solute interactions
solubility of gases
depends on T&P
solubility inc as P inc
solubility dec as temp inc
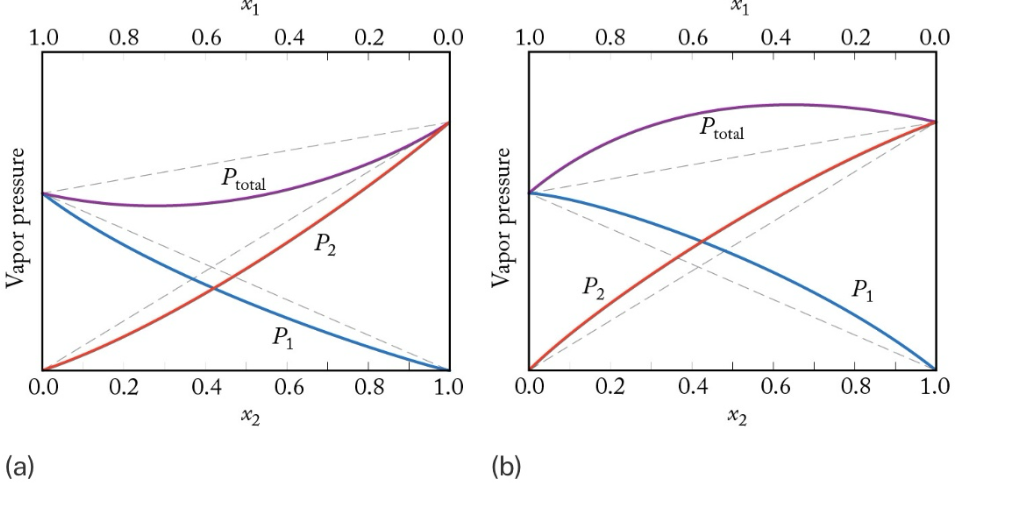
which is negative/positive deviation
a= negative
b= positive
determining a rate law

Fundamentals of Rate Laws
Rate law exponents must be determined through experiments; they cannot be figured out just by looking at the balanced chemical equation's
3 types of Reaction Rates
Average Rate: The change in concentration of a reactant or product over a specific, measurable time interval (Δ[A]/Δt)
Instantaneous Rate: The rate at a specific single point in time, determined graphically as the tangential slope of a concentration vs. time plot
Initial Rate: The instantaneous rate at the very start of the reaction (t = 0), immediately after reactants are mixed
Rate Law
equation that defines the relationship between the reactant concentrations and the reaction rate.
Method of determining Initial Rates
practical technique to determine the rate law by comparing how the initial rate changes when the starting concentration of one reactant is varied while others are held constant
If doubling a concentration doubles the rate, the order is 1st order.
If doubling a concentration quadruples the rate, the order is 2nd order.
If changing the concentration has no effect on the rate, the order is 0th order.
Factors Affecting Reaction Rates
temp: high temp inc kinetic energy of molecules leading to more frequent and energetic collisions
Activation energy: min energy required for a collision to result in a reaction
catalyst
temperature
change in concentration of a reactant
physical state of the reactants
pesudo first order
occurs when all reactants except one are in such high concentrations that they stay virtually constant leading the limiting reactant to control the rate