Phase 3
0.0(0)
Studied by 1 personCard Sorting
1/22
Earn XP
Description and Tags
Last updated 5:15 AM on 10/27/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
1
New cards
How are chemical reactions represented
By chemical equations
2
New cards
What does the + sign mean in a chemical equation
"Reacts with"
3
New cards
What does the arrow mean in a chemical equation
"Produces"
4
New cards
Reactants
(Starting substances)
The chemical formulas to the left of the arrow
The chemical formulas to the left of the arrow
5
New cards
Products
The chemical formulas to the right of the arrow, represent substances produced in the reaction
6
New cards
Coefficients
The number in front of the formula, indicate the relative numbers of molecules of each kind involved in the reaction
7
New cards
Balancing equations
Determining the coefficients that provide equal numbers of each type of atom of both sides of the equation
8
New cards
Coefficients should be...
The smallest whole-number possible (can also be fractions or decimals)
9
New cards
What does (g) mean
Gas
10
New cards
What does (s) mean
Solid
11
New cards
What does (l) mean
Liquid
12
New cards
What does (aq) mean
Aqueous solution
13
New cards
Types of reaction
- combination reaction
- decomposition reactions
- combustion reaction
- decomposition reactions
- combustion reaction
14
New cards
Combination reactions
Two or more substances react to form one produc. A reaction of this type between a metal and a non metal produces an ionic solid
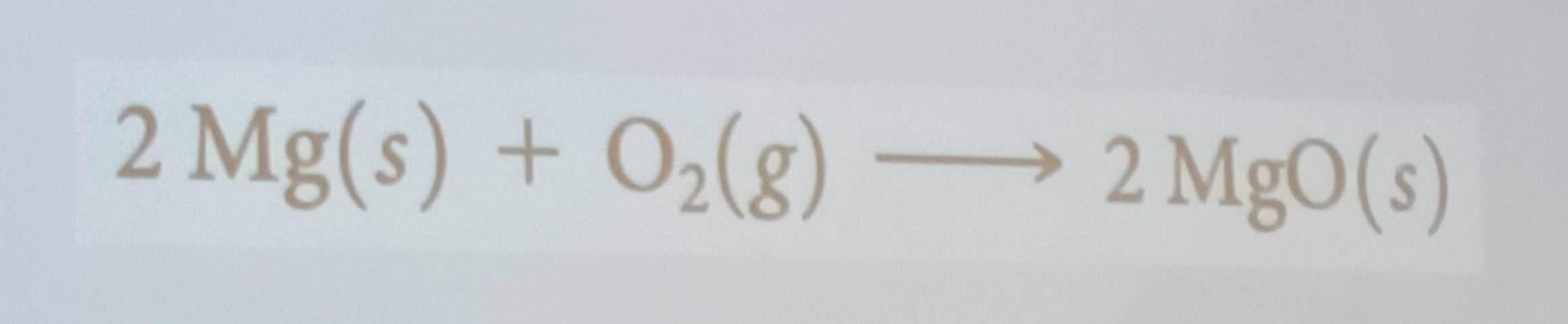
15
New cards
Decomposition reactions
One substances undergoes a reaction to produce two or more substances

16
New cards
Combustion reaction
Rapid reactions that produce a flame (most of them have O₂ from air as a reactant)
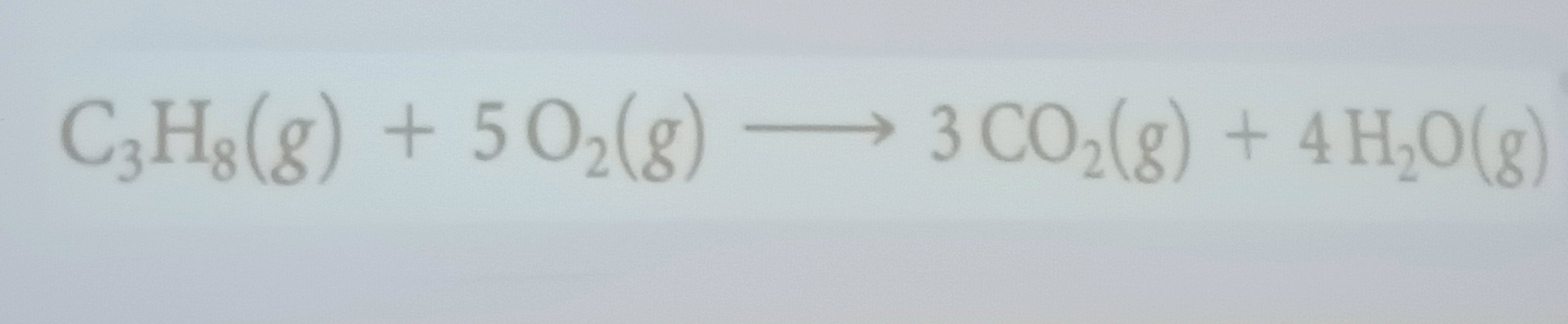
17
New cards
Hydrocarbons combustion in air react with O₂ to form..
CO₂ and H₂O
18
New cards
Single replacement reaction
Is a reaction in which one element replaces a similar element in a compound
-metal replaces metal
-non-metal replaces non-metal
-metal replaces metal
-non-metal replaces non-metal
19
New cards
Double-replacement reactions
Is a reaction in which the positive and negative ions of two atomic compounds exchange places to form to new compounds
-cations replace cations
-anions replace anions
-cations replace cations
-anions replace anions
20
New cards
Neutralization
An acid and a base react to form water and salt, and involves the combination of H⁺ ions and OH⁻ to generate water
21
New cards
Neutralization of a strong acid and strong base...
Has a pH that equals to 7
22
New cards
Neutralization os a strong acid and a weak base...
Has a pH of less than 7
23
New cards
Neutralization of a strong base and weak acid...
Has a pH greater than 7