Chapter 3 - Mass Relationships in Chemical Reactions | Definition
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Atomic mass
the mass of an atom in atomic mass units (amu)
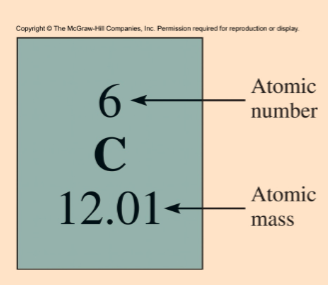
average atomic mass
the weighted average of all of the naturally occurring isotopes of the element
Mole (mol)
unit to count numbers of particles and the amount of a substance that contains as many elementary entities as there are atoms in exactly 12g of C12
Avogadro’s number (NA)
6.02×1023
molecule
an aggregate of two or more atoms in a definite arrangement held together by chemical forces.

Molecular mass (or molecular weight)
the sum of the atomic masses (in amu) in a molecule.
Percent composition of an element in a compound
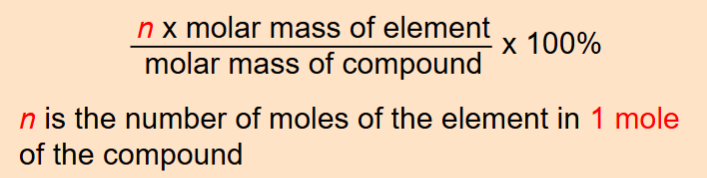
chemical reaction
A process in which one or more substances is changed into one or more new substances
chemical equation
uses chemical symbols to show what happens during a chemical reaction
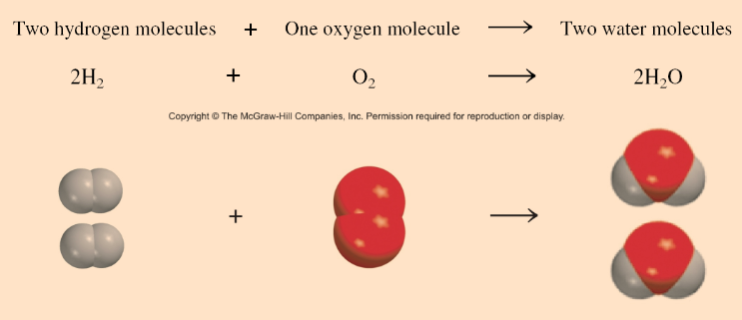
3 ways of representing the reaction of H2 with O2 to form H2O
Theoretical Yield
the amount of product that would result if all the limiting reagent reacted.
Actual Yield
the amount of product actually obtained from a reaction.
Formula of Reaction Yield
