To prepare ethene and examine its properties
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Theory
• Ethanol is dehydrated (has a molecule of water removed) using aluminium oxide (Al2O3) to form ethene gas (C2H4)
• Known as an elimination reaction
Equation
C2H5OH —Al2O3→ C2H4 + H2O
Ethanol —Aluminium oxide→ Ethene + water
Procedure
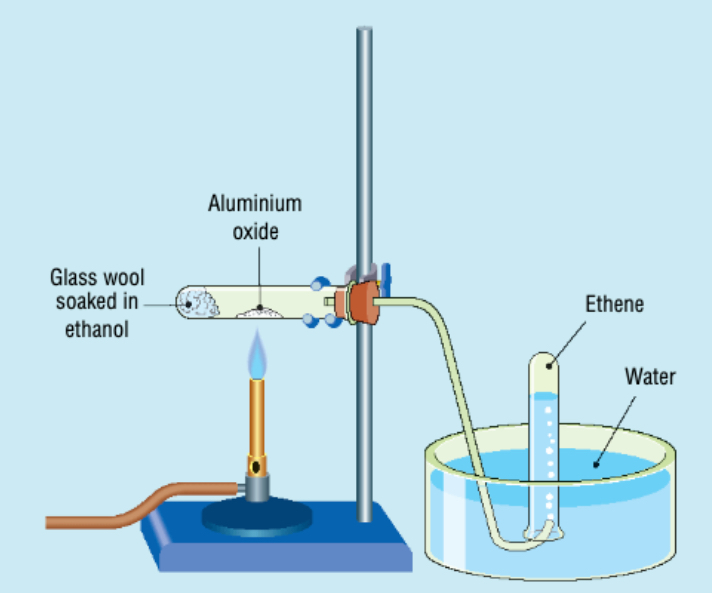
1. Place some glass wool at the bottom of a test tube and put some ethanol onto the glass wool. Clamp test tube horizontal. The glass wool will hold the ethanol in place when horizontal.
2. Add some aluminium oxide to the test tube (centre).
3. Connect the delivery tube to the test tube and inverted test tubes in a water trough.
4. Place a bunsen underneath the aluminium oxide and heat.
5. Fill several test tubes of ethene. The first test tube will contain air and can be disposed of.
6. Lift the apparatus up out of water before turning off the bunsen burner. This prevents ‘suck back’ occurring.
7. Test ethene test tubes to examine properties.
Physical properties
Ethene is a colourless gas with a sweet smell
It is insoluble in water but dissolves in non-polar solvents Example: Cyclohexane
Why do we collect ethene over water
It is insoluble in water but dissolves in non-polar solvents Example: Cyclohexane
test for Combustion
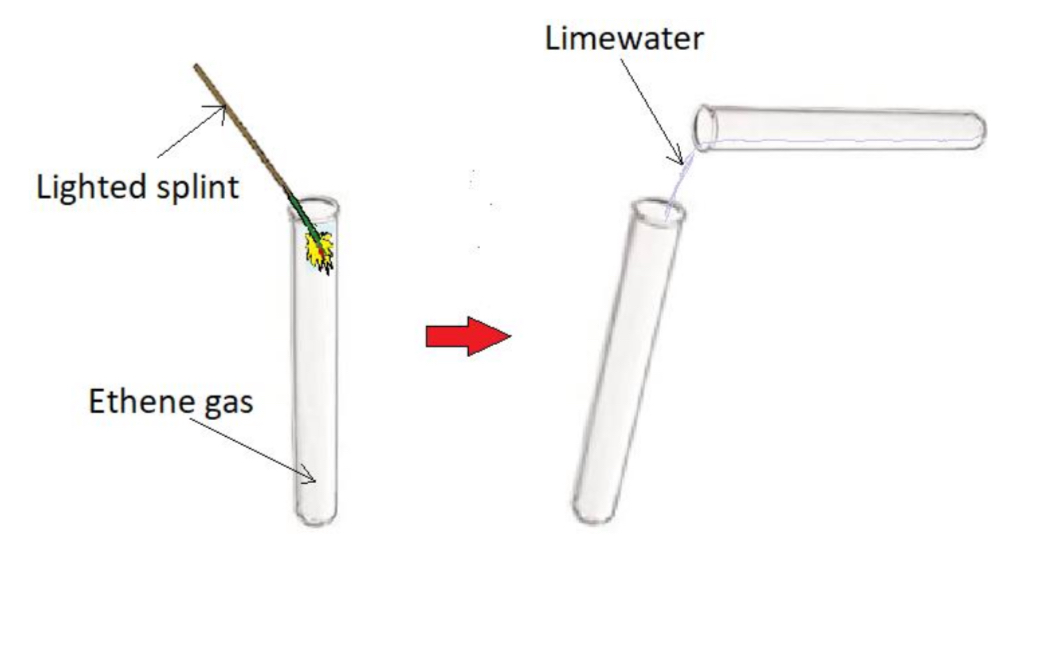
Procedure:
- Place a lighted splint into a test tube of ethene gas
- Add limewater to the test tube, stopper and shake
Result:
- Ethene burns with a bright yellow flame
- Limewater turns milky white proving carbon dioxide was formed in the combustion
Write a balanced chemical equation for the complete combustion of ethene?
C2H4 + 3O2 → 2CO2 + 2H2O
Note: ALL organic compounds, when combusted completely, produce carbon dioxide and water vapour
Write a balanced chemical equation to explain why carbon dioxide turns limewater milky white?
Limewater is calcium hydroxide - when added to carbon dioxide, white calcium carbonate forms
CO2 + Ca(OH)2 → CaCO3 + H2O
Testing for unsaturation (has double/triple bonds between carbons)– Method 1: Using bromine water
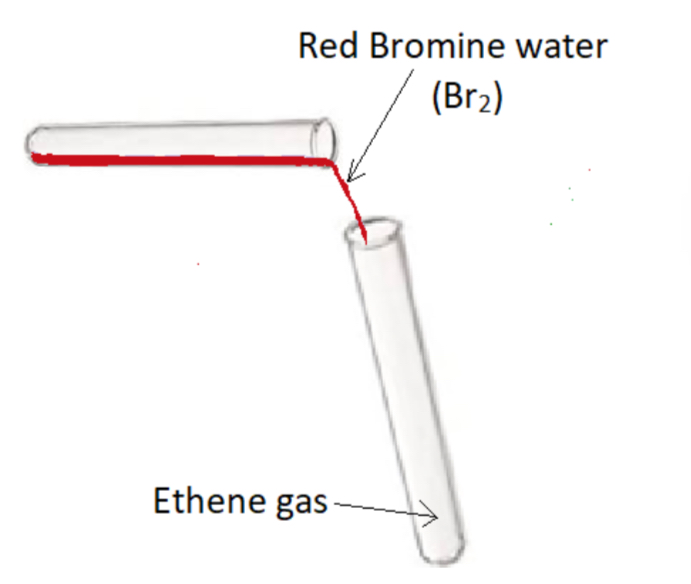
Procedure:
- Add red bromine water to a test tube of ethene gas, stopper and shake
Result: Red bromine water turns colourless – ethene is unsaturated
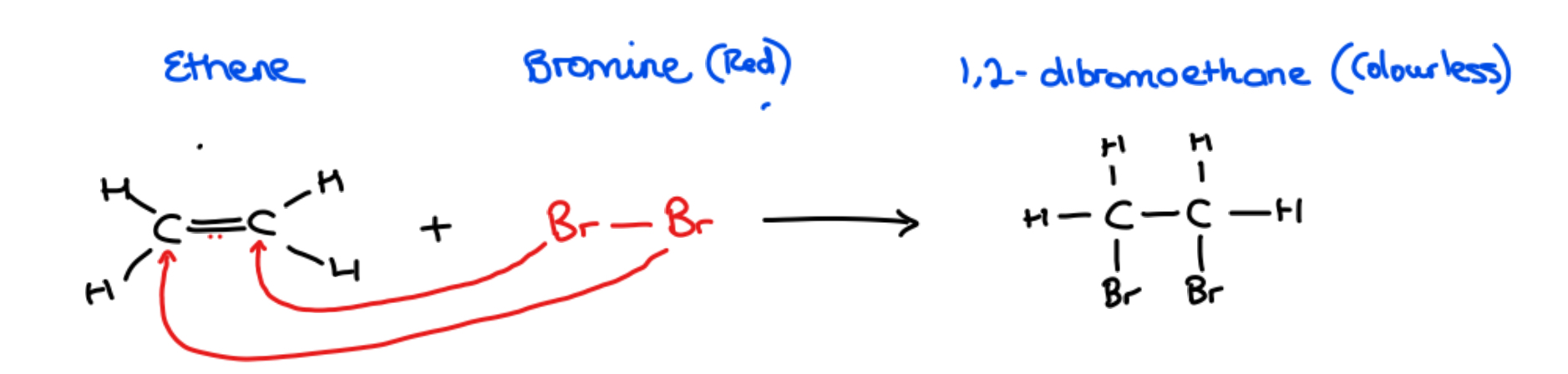
What type of reaction occurs when bromine is added to ethene gas
• Addition reaction – the adding of a bromine molecule across a double bond, breaking the double bond
Equation:
C2H4 + Br2 → CH2BrCH2Br
Ethene + Bromine (red) → 1,2- dibromethane (colourless)
Notice: The product formed 1,2 dibromoethane is colourless - this proves unsaturation
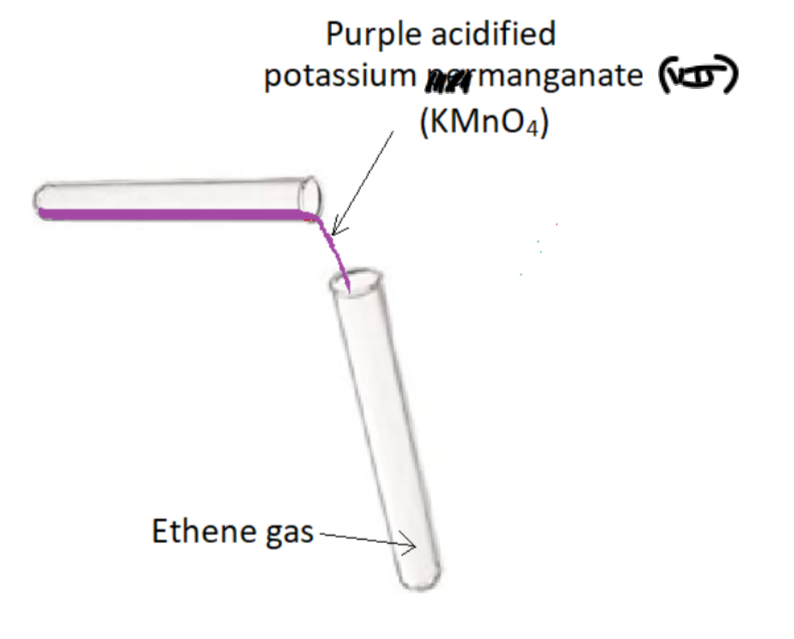
Testing for unsaturation – Method 2: Using acidified potassium manganate (VII) (KMnO4)
Procedure:
- Add purple acidified potassium manganate (VII) to a test tube of ethene gas, stopper and shake
Result:
Purple acidified potassium manganate (VII) turns colourless - ethene is unsaturated
What type of reaction is the preparation of ethene known as?
Elimination reaction or Dehydration reaction (removing a molecule of water)
What is the function of the aluminium oxide?
Aluminium oxide i) acts as a dehydrating agent.
ii) Is a catalyst for the reaction.
What is the appearance of aluminium oxide?
Aluminium oxide is a white solid powder
Why is the aluminium oxide gently heated/why is the ethanol not heated directly?
To avoid the ethanol vaporising too quickly and passing over the aluminium oxide too quickly – little ethanol would be converted into ethene
What is the function of the glass wool in this experiment?
The glass wool soaks up the ethanol and keeps it in one end of the test tube – stops the ethanol being directly heated
State and explain four safety precautions in this experiment
1. Ensure the stopper on the test tube is airtight
Ethene and Ethanol are flammable – essential to keep them away from the bunsen flame preventing a fire
2. Wear gloves and use tongs
Glass wool is an irritant to skin – avoid touching it
3. Raise the apparatus from the water before turning the bunsen burner off
Avoid “suck-back” of water up the delivery tube - cold water being sucked back up into the hot test tube could cause it to shatter
Why are the first few test tubes of gas collected discarded?
The first few test tubes consist mostly of displaced air and not ethene gas
Give an industrial application of ethene
1. Ethene generators are used commercially to speed up the ripening of fruit
2. Polymerisation of ethene is used to produce polyethene (polythene) – used in plastics