Thermodynamics A2 Anki
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Define second electron affinity.
the enthalpy change when one mole of gaseous 1- ions gains one electron per ion to produce gaseous 2- ions
Define second ionisation energy.
the enthalpy change to remove 1 mole of electrons from one mole of gaseous 1+ ions to produce one mole of gaseous 2+ ions
Define standard enthalpy of atomisation.
The enthalpy change when 1 mole of gaseous atoms is formed from the element in its standard state under standard conditions
Draw the Born Haber cycle for determining the lattice enthalpy of dissociation of CaO.

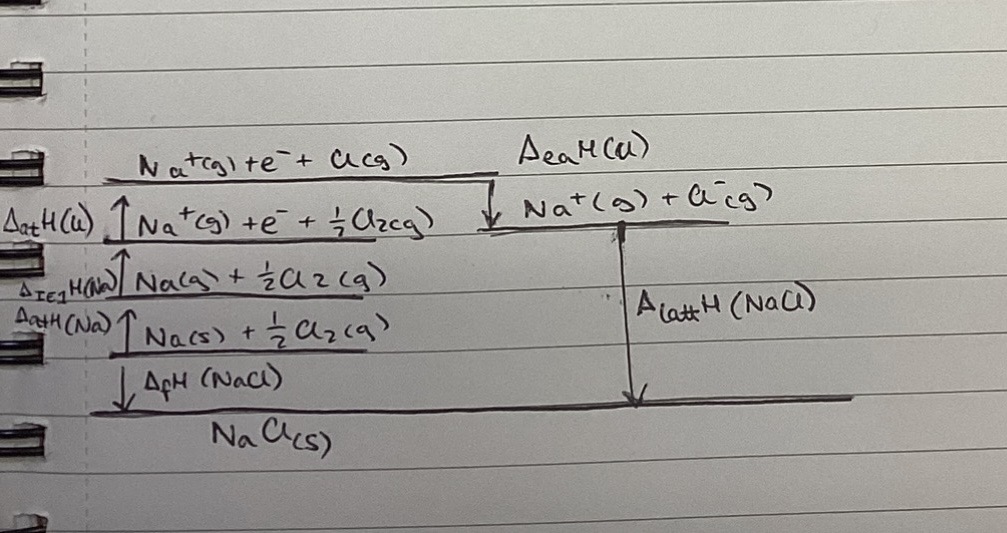
Draw the Born-Haber cycle for determining the lattice enthalpy of formation of sodium chloride.
Equation for entropy change
Standard entropy change of reaction = (Sum of standard entropies of products) − (Sum of standard entropies of reactants)
Unit of entropy is JK-1mol-1
How are theoretical lattice enthalpies calculated?
How are experimental lattice enthalpies calculated?
Theoretical lattice enthalpies are calculated using computer modelling assuming a perfect ionic model.
Experimental lattice enthalpies are found using a Born-Haber cycle (with values from experimental data)
How can you calculate at what temperature a reaction becomes feasible?
Reaction becomes feasible at ΔG = 0 so
ΔG = ΔH - TΔS
ΔH - TΔS = 0
ΔH = TΔS
T = ΔH/ΔS
How can you predict a change in entropy qualitatively?
In general, a significant increase in entropy will occur if:
there is a change of state from solid or liquid to gas
there is a significant increase in the number of molecules between products and reactants
Number of molecules is less important than change of state.
Is bond dissociation enthalpy endothermic or exothermic?
endothermic (+ve)
Is the enthalpy of hydration endothermic or exothermic?
Why?
exothermic (-ve)
because bonds are formed between the ions and the water molecules
Is the enthalpy of lattice dissociation endothermic or exothermic?
endothermic (+ve)
Is the enthalpy of lattice formation endothermic or exothermic?
exothermic (-ve)
Is the first electron affinity endothermic or exothermic?
exothermic (-ve) for atoms that normally form negative ions
Is the first ionisation energy endothermic or exothermic?
endothermic (+ve)
Is the second electron affinity endothermic or exothermic?
endothermic (+ve)
Is the second ionisation energy endothermic or exothermic?
endothermic (+ve)
Is the standard enthalpy of atomisation endothermic or exothermic?
endothermic (+ve)
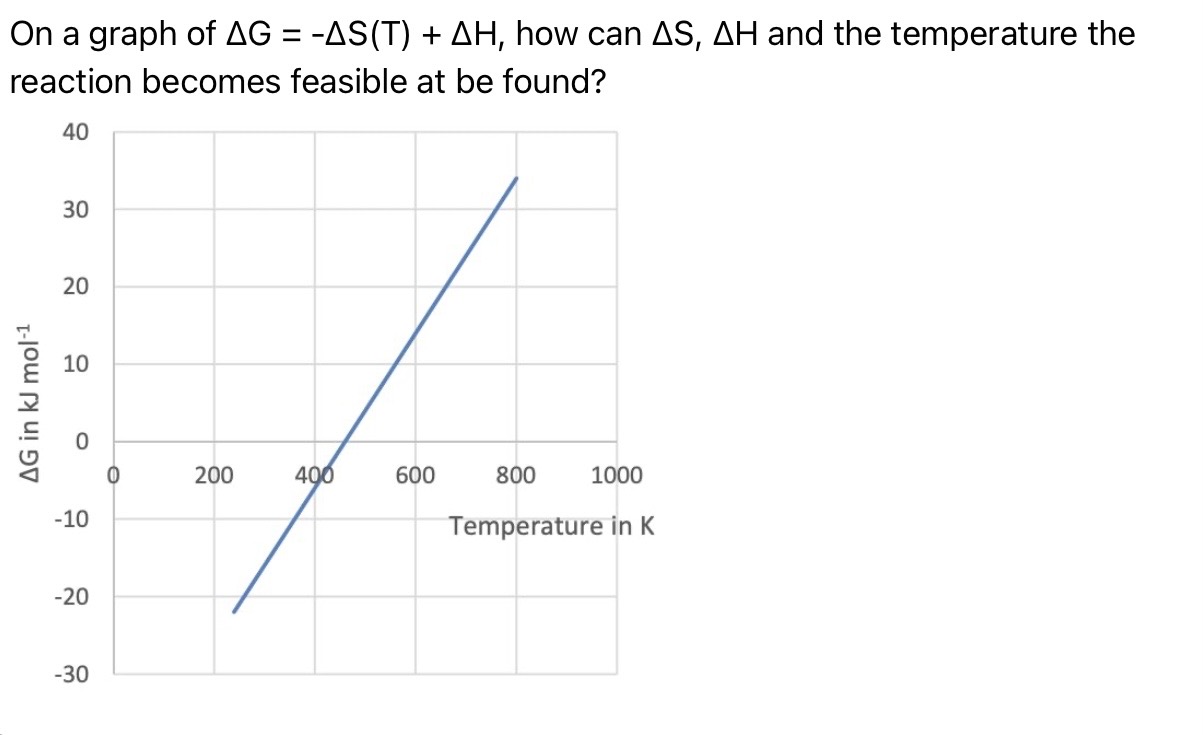
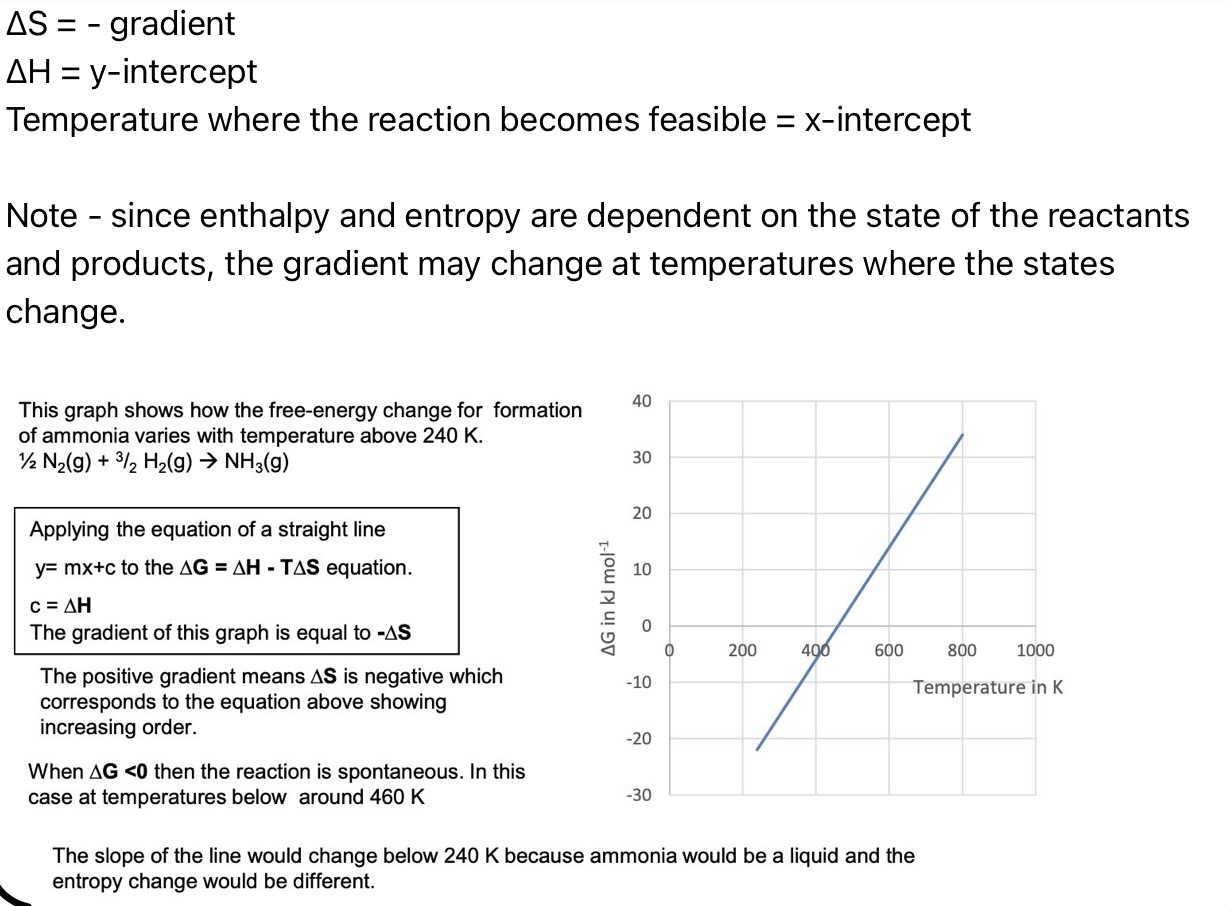
On a graph of ΔG = -ΔS(T) + ΔH, how can ΔS, ΔH and the temperature the reaction becomes feasible at be found?
ΔS = - gradient
ΔH = y-intercept
Temperature where the reaction becomes feasible = x-intercept
Note - since enthalpy and entropy are dependent on the state of the reactants and products, the gradient may change at temperatures where the states change.
The lattice enthalpies become less _______ down any group.
negative
Theoretical lattice enthalpies assumes a perfect ionic model.
What does this mean?
100% ionic bonding (attraction is purely electrostatic)
ions that are perfect spheres
no polarisation of ions
What 4 things could cause a change in entropy?
Change in temperature
increase in temperature increases particle movement so more entropy
Change of state
there is a bigger entopy increase in boiling than melting
Dissolving solids
particles are more spread out so move more so more entropy
Reactions
need to consider the state and number of reactants and products
What are Born-Haber cycles used for and why?
used to calculate lattice enthalpy (usually of formation)
because lattice enthalpy cannot be determined directly from experiment
What does similar experimental and theoretical lattice enthalpies mean?
there is an almost perfect ionic model (100% ionic bonding, spherical ions, no polarisation)
What does the magnitude of the enthalpy of lattice formation depend upon?
It is a reflection of the strength of attraction between the positive and negative ions so depends upon:
The charge on the ions - the greater the charge of the ion, the greater the attraction between the ions so the larger the lattice enthalpy is (more negative).
The size of the ions - the smaller the ions, the larger the attraction is between them
Both of these increase the charge density.
What does very different experimental and theoretical lattice enthalpies mean?
There is some covalent character.
The greater the difference, the more covalent character there is.
When a compound has some covalent character- it tends towards giant covalent so the lattice is stronger than if it was 100% ionic. Therefore the Born-Haber value would be larger than the theoretical value.
What is enthalpy of hydration affected by?
Ion size - smaller ions will attract water molecules more readily so will be more exothermic
Ionic charge - higher charged ions will attract water molecules more readily so will be more exothermic
What is the equation for change in gibbs free energy?
Note - entropy change needs to be converted to kJK-1mol-1 from JK-1mol-1

What is the equation for enthalpy of solution (Hess' law is used)?
Enthalpy of solution = Lattice dissociation enthalpy + (Sum of hydration enthalpies of all ions)
What is the relationship between enthalpy of lattice formation and enthalpy of lattice dissociation?
Enthalpy of lattice formation = - Enthalpy of lattice dissociation
What makes a reaction feasible/spontanteous?
Change in gibbs free energy is negative
However, the reaction may still not take place if there is a high activation energy
When does entopy = 0?
at 0K
When is covalent character more likely?
Positive ion has a high charge density (smaller ion)
Negative ion is easily polarisable
larger - electrons are further away from the nucleus
higher (ionic) charge - more electron density to be attracted towards the positive ion
Why does calcium chloride have the formula CaCl2 and not CaCl or CaCl3?
CaCl2 has the most exothermic enthalpy of formation so it will be the most thermodynamically stable
Why does covalent character occur?
The positive ion attracts the electron density of the negative ion towards itself.
If there is a great enough attraction then overlap (of the electron densities) occurs (covalent character).
Why is it difficult to predict whether the enthalpy of solution is endothermic or exothermic?
The same factors affect the size of the lattice enthalpy
and the enthalpies of hydration (higher charge density
increases both).
Enthalpy of solution = Lattice enthalpy of dissociation (+ve) + _ Enthalpies of hydration (-ve)
Why is second electron affinity endothermic?
it takes energy to overcome the repulsive force between the negative ion and the electron
Why is the first electon affinity exothermic for atoms that normally form negative ions?
the ion is more stable than the atom and there is an attraction between the nucleus and the electron
ΔH is negative.
ΔS is negative.
At what temperatures is this reaction feasible?
at low temperatures
ΔH is negative.
ΔS is positive.
At what temperatures is this reaction feasible?
Reaction is always feasible.
ΔH is positive.
ΔS is negative.
At what temperatures is this reaction feasible?
Reaction is never feasible.
ΔH is positive.
ΔS is positive.
At what temperatures is this reaction feasible?
at high temperatures