bio exam 2
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Atom
smallest unit of matter that
contains the properties of an element
Proton +1, Neutron, Electron -1
Atoms are made up of what 3 characteristic
(subatomic particles)
nucleus
Atoms are arranged with the protons
and neutrons held in the center of the?
electron shells or a cloud
what do electrons form around the outside?
uncharged
In what atom (NOT an ion) does the
number of protons = electrons
zero, two -1 electrons
total charge = what two +1 protons = what
electron shells
electrons exist in rings outside the nucleus called
inside to out
do electrons fill shells inside to out or out to inside
Ionic bond
an electron is fully donated from one atom and accepted by
another
ions (charged atoms)
what do ionic bonds generate
Covalent bond
formed when each atom shares its electrons. Electrons
are NOT transferred and so fully charged ions are NOT created.
Each atom “feels” like it has a full outer ring.
Molecule
joining of 2 or more atoms by a covalent bond
covalent
in what bond are the atoms only “happy” as
long as it continues to share in the bond.
Single covalent bond
only a single pair of
electrons is shared (2 electrons total); H2
Double covalent bond
sharing of two pairs of
electrons (4 electrons total); O 2
Triple covalent bond
sharing of three pairs of
electrons (6 electrons total); N2
Quadruple covalent bond
sharing of four pairs
of electrons (8 electrons total); rarely seen
triple > double > single
bond strength order?
Carbon
what element needs to form four total covalent bonds to fill its outer ring.
Nonpolar covalent
Equal sharing
when electrons stay in the middle;
neither feels slightly positive or
slightly negative
Polar covalent:
an electron “hog”
pulls the electrons closer to it making
itself slightly negative and whatever it
is pulling from slightly positive.
Polar covalent bonds
what bonds create partial
charges
Hydrogen bonds
interactions between slightly positive
hydrogen and a slightly negative oxygen or nitrogen
Capillary action (cohesion), Surface tension, Ice floats (lower density), Absorbs heat, Solvent properties
Water hydrogen bonding gives rise to
several of its properties:
Hydrophobic
water-fearing
Hydrophilic
water-loving
C. 19 protons, 20 neutrons
The atomic number of potassium is 19. The mass number of
potassium is 39. How many protons and neutrons does
potassium have?
A. 19
The atomic number of potassium is 19. The mass number of
potassium is 39. How many electrons does a potassium
atom have?
A. True
In a single covalent bond, two atoms share a
pair of electrons
D. both polar and nonpolar covalent bonds
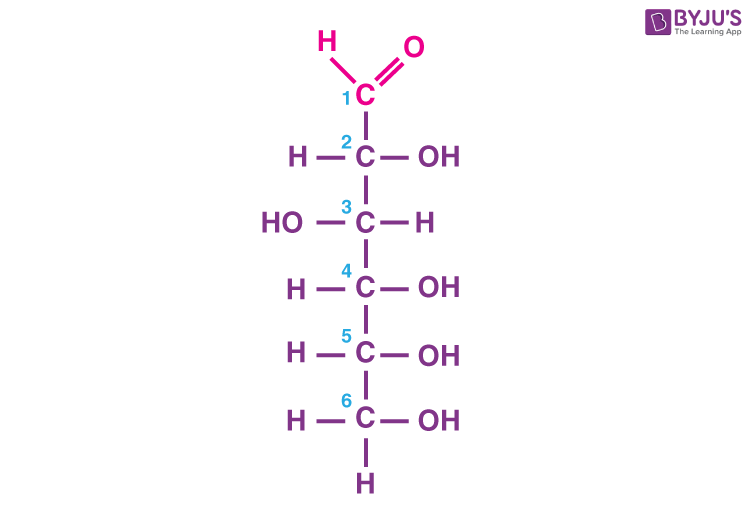
This is the molecule glucose. It has…
A. +
The oxygen atoms in water interact with _____ ions.