Systems Physiology
1/258
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
259 Terms
Superior (Cranial) and Inferior (Caudal)
Superior = toward the head
Inferior = away from the head
Anterior (Ventral) and Posterior (Dorsal)
Anterior = towards or at the front of the body
Posterior = toward the back
Medial, Intermediate, and Lateral
Medial = toward or at the middle of the body
Lateral = away from the middle
Intermediate = between a more medial and more lateral structure
Proximal and Distal
Proximal = closer to the origin of the body part
Distal = farther from the origin of the body part
Superficial (External) and Deep (Internal)
Superficial = toward or at the body surface
Deep = deep beneath the body’s surface
Midsagittal Plane and Parasagittal Plane
Midsagittal plane = cut was made perfectly on midline from back to front.
Parasaggittal plane = cut was off-centred, not on midline
Frontal (Coronal) Plane
Cuts body vertically from sides, right to left. Produces a frontal or coronal section.
Transverse Plane
Cuts body horizontally, produces a cross section.
4 Principles of Karl Ernst von Baer
General features appear before specialised features
General features progress into specialised features
Embryos from more evolved animals diverge more than embryos from primitive animals
Embryos of higher animals resemble embryos of more primitive animals (but not the adult)
Evolutionary Biology
Compares embryonic development across species.
Teratology
Studies embryonic defects to infer normal development
Modern embryology discovered that…
The amorphous zygote undergoes gradual changes as opposed to being a miniature already formed.
Hans Spemann Transplantation Experiment
Demonstrated that the dorsal blastopore lip of an amphibian embryo acts as an "organizer". By transplanting this tissue to a new location on a host embryo, they induced the formation of a second, complete embryonic body axis, proving that specific tissues dictate cell fate in development.
Alan Turing Reaction-Diffusion Model
Chemical basis of morphogenesis: the initial symmetry in embryos can be broken by the interplay between two diffusible molecules (activator and inhibitor), whose interactions lead to the formation of patterns.
John Gurdon Experiment
Used SCNT (Somatic Cell Nuclear Transfer) to prove that specialized adult cells retain the full genome needed to create an entire organism, fundamentally challenging the belief that cellular differentiation is irreversible. By transferring the nucleus of a mature frog intestinal cell into an enucleated egg, he created cloned, swimming tadpoles.
Shinya Yamanaka Discovery
Found that the Yamanaka factors when put into a somatic cell can reprogram it into a stem cell-like state
What are the Yamanaka factors?
SOX2, KLF4, and OCT4
3 Types of Embryonic Stem Cells
Totipotent, pluripotent, and multipotent.
Totipotent Stem Cells
Can generate all cell types including extra-embryonic tissue (ex. placenta).
Pluripotent Stem Cells
Can generate all cell types and is the derivative of the 3 germ layers but can’t generate extra-embryonic tissue.
Multipotent Stem Cells
Can generate multiple related cell types.
How is pluripotency maintained?
By transcription factors that activate pluripotency genes and repress differentiation programs.
Cellular Tools for Embryology
Fate mapping to determine cell fate: dye and genetics approaches – transplant experiments (chimera) experiments (1960/70’s).
Molecular Tools for Embryology
Forward and reverse genetics (2000’s)
Genome editing (crispr cas9) (2013) - transgenesis (transient vs stable) 2010’s revolution
Gain of function and Loss of Function, Knock in, Fluro Tags cellular and molecular
Steps of Transcription
Initiation, elongation, splicing, 3’ processing, and termination.
Basal Transcriptional Machinery
RNA Polymerase II (synthetize RNA strand) + mediator, integrator and elongation protein complexes = micro machine that produce RNA.
Transcription Factors
Are the switches for the micro machines and are a class of protein that binds (and sometime bend) DNA. They function to induce cell lineage specification and maintain cell type identity. They also to turn on and off gene expression and modulate the process of transcription.
What do transcription factors bind to?
Enhancer and promoter regions
Enhancers
Enhancers are distal, orientation-independent sequences that bind activator proteins to significantly boost transcription rates.
Promoters
Promoters are DNA sequences located immediately upstream of a gene that initiate transcription by binding RNA polymerase, acting as an essential on/off switch.
Process of Transgenic Animal Making
Plasmids are added via electroporation to embryonic stem cells which are then added to embryos before they are implanted into a parent . These animals will then express the desired protein.
How are gain or loss of function animals made?
A mouse is engineered so the target gene is flanked by LoxP sites (“floxed” gene).
A second mouse expresses Cre in a specific tissue (e.g., liver or brain).
The mice are crossed.
In cells where Cre is expressed, the gene between the LoxP sites is deleted → gene is knocked out only in that tissue.
The opposite happens for gain of function where the gene will become activated.
Pregnancy Stages
Duration is 38 to 40 weeks. 1st trimester = embryo and after that = fetus.
Two Types of Germline Cells
Oocyte and Sperm and have 23 chromosomes each.
Oocyte
Female germ (egg) cell, X or X chromosome, 100um, rich in cholesterol (for week 1 nutrients).
Zona Pellucida
A layer of glycoprotein which prevents the egg from attaching to an ectopic site, also provides species-specific sperm binding.
Sperm
Male germ cell, X or Y chromosome, the head is 5.1x3.1um.
Chromosomal Disjunction
Homologous chromosomes in meiosis I or sister chromatids in meiosis II separate ensuring each daughter cell receives a complete set of chromosomes.
Chromosomal Nondisjunction
Failure of homologous chromosomes or sister chromatids to separate properly during cell division.
Spermatogenesis
The process of making sperm cells.
Oogenesis
The process of making an oocyte or egg cell in females.
Arresting of Oocytes
All the eggs (around 1 million) will arrest at prophase I shortly after birth. Each menstrual cycle some oocytes will continue meiosis and arrest at metaphase II until fertilization.
Causes of Female Chromosomal Malformations Due to Meiosis
Maternal age and chromosomal defect: dramatic increase beyond age 35 in trisomy 21 (Down syndrome).
Loss of cohesion complex may be responsible for abnormal meiosis
Main Events of Early Embryonic Development
Week 1: fertilisation to blastocyst formation
Week 2: implantation to bilaminar formation
Week 3: gastrulation to form 3 germ layers
Week 4: neurulation
Week 1 of Early Embryonic Development
Ovulation, fertilization, cleavage, morula, zona hatching to form blastocyst (end of week 1), implantation.
Fertilization
Sperm from a male meets an oocyte from a female to forms a zygote via cell proliferation (termed cleavage).
Blastocyst Formation
Once the zygote loses the zona pellucida there is a sodium and water infiltration which divides the cell type into 2 (inner cell mass and trophoblasts) to from a blastocyst and once this happens it can implant
What do the trophoblasts and inner cell mass form?
Trophoblasts → placenta
Inner cell mass → embryo
Week 2 of Early Embryonic Development
Implantation and Bilaminar Formation
Implantation
The blastocyst travels closely to the uterus wall and L-selectin on the surface of the trophoblasts attaches to the uterine epithelial cells
The trophoblasts secrete enzymes that degrade the epithelial cells and allow the blastocyst to infiltrate the uterine wall to implant
Bilaminar Formation
The inner cell mass becomes 2 layers which are the epiblast and hypoblast. The epiblast becomes the embryo and the hypoblast is a yolk sac rich in cholesterol that functions in embryo patterning, nutrient transport, and are primitive red blood cells.
Primitive Streak
A groove-like structure that appears in the epiblast during gastrulation marking the beginning of the process defining the future anterior-posterior axis of the embryo.
Week 3 of Early Embryonic Development
Gastrulation occurs to form the 3 germ layers including mesoderm differentiation into paraxial and splanchnic components, heart tube formation, and paraxial mesoderm derivative formation.
Gastrulation
Epiblast further divides into the 3 primary germ layers which includes the endoderm (epithelial cells), mesoderm (mesenchymal stem cells), and ectoderm.
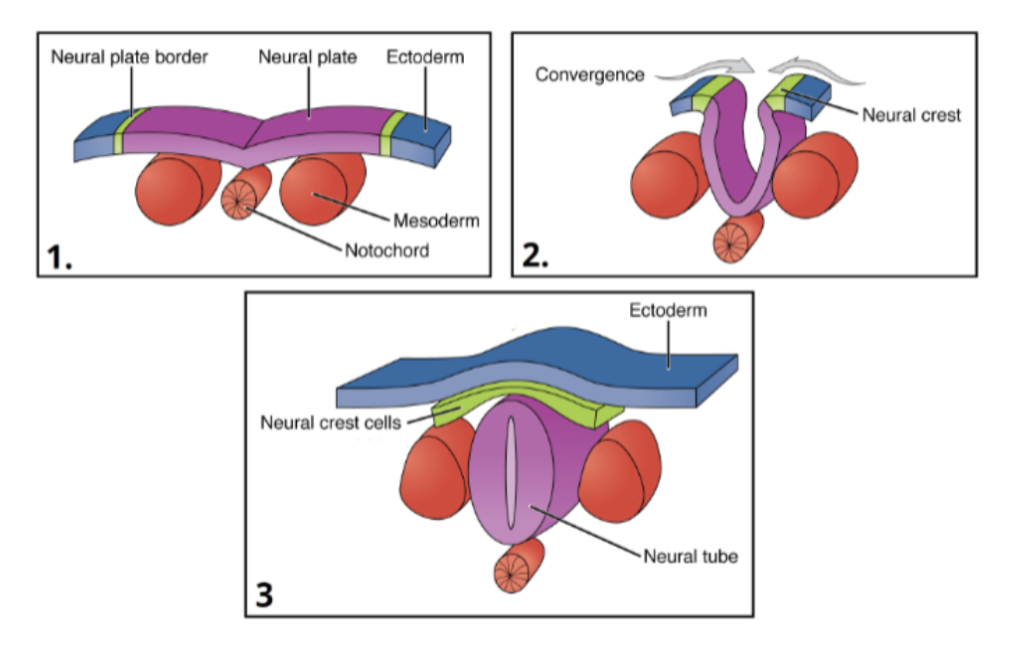
Notochord Formation
Occurs during week 3 and is a rod-like structure made of a cartilaginous substance derived from the mesoderm. Its role is to guide proper development of vertebra and differentiation of neural tube.
Mesoderm
The most abundant germ layer in the human body and gives rise to the musculoskeletal system (bone, cartilage and muscle), cardiovascular system (heart, blood and blood vessels), and connective tissues found throughout our bodies.
Differentiation of Mesoderm
Differentiates into paraxial mesoderm (forms skeletal muscle) and splanchnic mesoderm (forms cardiac muscle and smooth muscles of internal organs).
Molecular Markers on Cardiac and Skeletal Muscle
Skeletal: Myf5 and Pax7
Cardiac: Nkx2-5 and Gata4
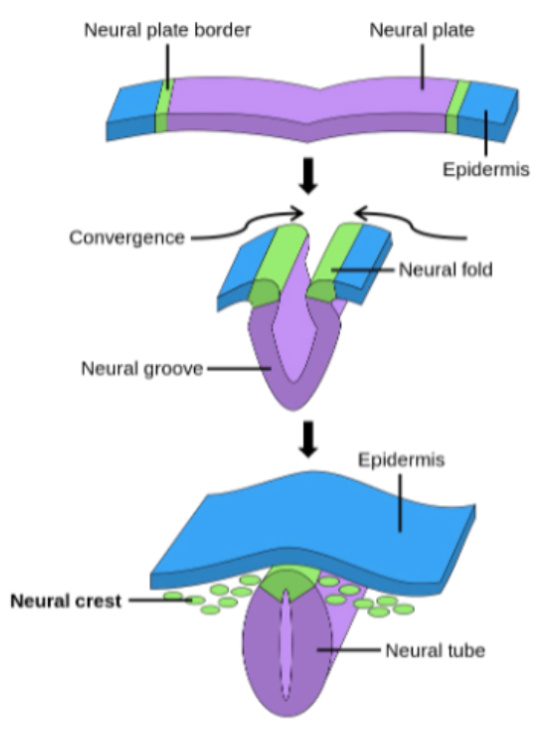
Neurulation
The neural tube will develop into the central nervous system. The cranial neuropore also closes around day 24, and the caudal neuropore closes around day 28.
Neural Crest Cell (Ectomesenchymes) Formation
Occurs during week 4 and develop from neural ectoderm via epithelial to mesenchymal transition and form:
Teeth
Skull bones (anterior skull and facial bones)
Melanocytes
Peripheral nervous system: peripheral neurons, glia cells
Ectoderm
Gives rise to the nervous system (central and cranial), skin and oral mucosa, cranial bone via neural crest cells, and teeth.
Endoderm
Gives rise to the colon, stomach, intestines, liver, pancreas, lungs, thyroid and parathyroid.
Morphogenesis
Cells form the shape and physical structure of the organ. They do this via cell proliferation, differentiation, migration, extracellular matrix deposition, tissue folding and convergence, and apoptosis.
Heart Development
The heart is the first major organ to develop because there is an urgent need for a blood supply to provide oxygen and nutrients to the growing embryo.
Genes Involved in Heart Development
Over 500 genes identified with cardiovascular development and its not fully understood because no single transcription factor/morphogen is responsible for cardiogenic specification or heart development.
What regulates heart development?
Genetic and epigenetic factors, transcription factors, morphogens, apoptosis, cell-cell adhesion, extracellular matic, and mechanical factors including cellular migration and hemodynamics (blood flow).
Days and Thickness of Embryo
Days 0-6 formation of blastocyst
Days 7-12 embryo becomes bilaminar and implanted fully
Days 13-18 embryonic disc becomes trilaminar: gastrulation with three germ layers (ectoderm at the top, mesoderm in the middle, and endoderm at the bottom)
Lateral plate mesoderm (the side bits of the mesoderm) subdivides into somatic, coelom (cavity), splanchnic, and extra-embryonic
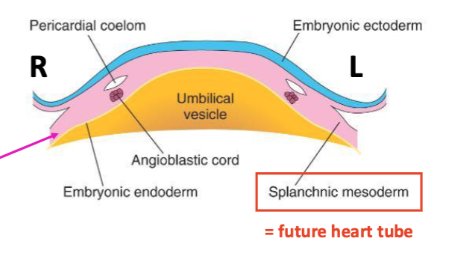
Cardiogenic Specification
Begins on day 18 (end of 3rd week) where the lower region (splanchnic mesoderm) will develop into the heart tube. Includes the specification of distinct regions that will eventually form different parts of the heart via the formation of 2 heart fields.
Morphogen Gradients
A key mechanism in embryonic development that allows for precise spatial and temporal regulation of gene expression. Expressionainhibition zones spatially restrict cardiogenic mesoderm.
What do the morphogens Mesp1 and Mesp2 control?
Migration of the cardiac progenitor cells into the cranial region of the embryo.
What do the morphogens BMP2, BMP4, TGFβ, and Activin control?
They specify cardiogenic progenitors in the lateral mesoderm.
What are BMP2, BMP4, TGFβ, and Activin inhibited by?
The release of chordin and noggin from the notochord and release of Wnt1/3a/8a from the forming neuroectoderm.
What morphogens inhibit Wnt?
Crescent (FRZB2) and Dickkopf (DKK1)
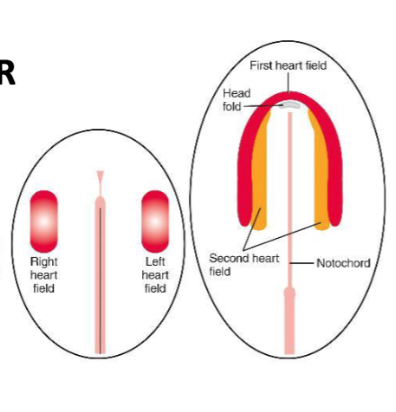
Formation of the 2 Heart Fields
Cardiac progenitor cells form the first heart field and second heart field (pharyngeal mesoderm) located medial to the first heart field.
Genes that specify cardiogenic progenitors in the first heart field include…
Nkx2.5, Gata4, Tbx Family (Tbx5), FGF8, SHH
Genes that specify cardiogenic progenitors in the second heart field include…
Increased Nkx2.5 and Gata4, Hes-1, Islet-1 (Isl1) and additional inhibitors e.g. Notch.
What do the heart fields go on to form?
1st Heart Field: LV, atria, aorta
2nd Heart Field: sinus venosus wall, RV myocardium, atrial septa, outflow tract and the myocardial wall of the RA, LA
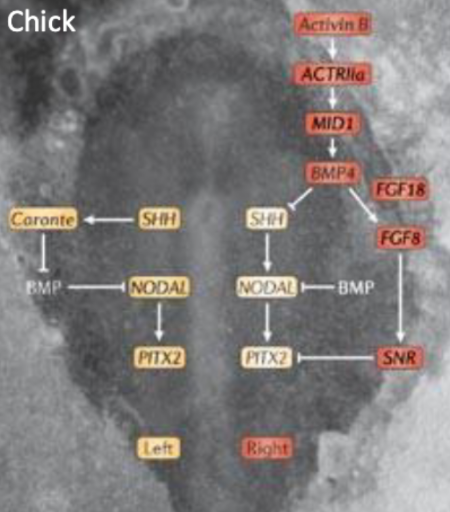
Left to Right Specification of the Heart
The symmetry to specify left and right is initially formed by morphogen gradients. Where there is basically extra transcription factors on the right that inhibit stuff that would be happening on the left on the right.
Right Heart Specification
Activin signal on the right side of Hensen's node
Local repression of sonic hedgehog (SHH) expression
FGF8 expressed on the right side, can inhibit Pitx2 via Snail Related (SnR2)
Left Heart Specification
SHH restricted to the left side of Hensen's node
Allows activation of NODAL expression on the left side of the embryo
NODAL -> Pitx
Pitx2c is a key regulator of left asymmetry
SHH, Nodal and Pitx-2 genes on L
Left sided specification starts prior to heart tube formation
What processes are involved in folding and heart tube formation?
Lateral folding, cranial-caudal (head to tail, starts to curve) folding, and ventral folding.
Lateral Folding
Days 20-21: the sides are coming together
Day 22: the heart tube fuses and the pericardial cavity also fuses to form the epicardium. The myocardium secrete cardiac jelly
Day 24: the heart tube ‘beats’ in a peristaltic motion
Blood Flow Through the Heart Tube
From placenta via the umbilical vein (oxygenated), through heart tube, connected to dorsal aorta. The blood comes in to the heart through the sinus venosus and out through the truncus arteriosus.
What are the layers of the heart tube?
Endocardium (inner), cardiac jelly, myocardium
Cardiac Jelly
Secreted by myoblasts in the myocardium and it assists with cell migration, proliferation, and differentiation.
Cranial-Caudal Folding
Folding of the cranial and caudal parts down and back (for the front). Occurs from day 22-26.
What does the aortic sac of the heart tube become?
The pharyngeal arches/great vessels
What does the truncus arteriosus of the heart tube become?
Ascending aorta and pulmonary artery
What does the bulbus cordis (conus) of the heart tube become?
Outflow track and valves of the right ventricle
What does the primordial/primitive atrium of the heart tube become?
The left and right atria
What does the sinus venosus of the heart tube become?
Large veins and pulmonary veins, visceral pericardium, gestational conduction system, and coronary sinus.
Contribution of the Secondary Heart Field During Tube Formation
While the primitive tube forms primarily from the primary heart field, the secondary heart field continuously adds progenitor cells to the arterial and venous poles. This elongation is what later forces the tube to loop.
What occurs after folding and heart tube fusion?
Looping and chamber specification
What drives heart looping?
Rapid elongation of the heart tube (especially via secondary heart field addition)
Constrained space within the pericardial cavity
Differential growth across regions
3 Stages of Cardiac Looping
C-shape
D-shape
S-shape
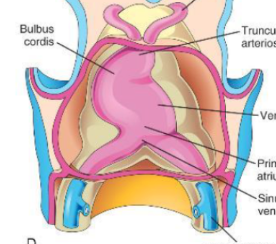
C Looping
The truncus arteriosis and bulbus cordis move to the right and the right ventricle moves down and right.
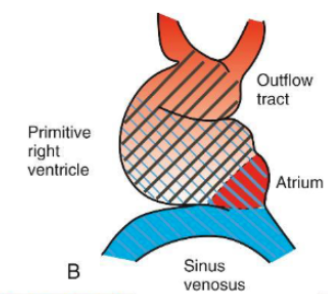
D Looping
Bulbus cordis and primitive ventricle grow rapidly causing the tube to bend ventrally and rightward. This is called D-looping (dextral looping).
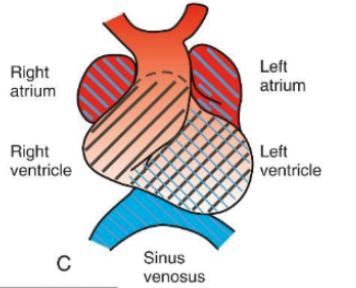
S-Shaped Configuration
The left ventricle moves and left to twist, the pulmonary artery and the sinus venosus move back and up.
What regulates chamber specification?
Morphogen gradients, retinoic acid (contributes to atria specification), and the expression of retinaldehyde dehydrogenenase2 (Raldh2).
Morphogen gradients that regulate chamber specification
Tbx5 specifies atrium and SV
Irx4 specifies ventricles and outflow tracks (OFT: BC, TA and AS)
Combination of both specifies the LV