7.2 ionisation energies
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
what is ionisation energy?
the measure of how easily an atom loses electrons to form positive ions
define first ionisation energy
the energy required to remove one electron from each atom in one mole of gaseous atoms of an element to form one mole of gaseous 1+ ions
what are the 3 factors that affect ionisation energy?
atomic radius, nuclear charge, electron shielding
what is the affect of atomic radius on ionisation energy?
the greater the distance between the nucleus and outer electrons (atomic radius), the less the nuclear attraction.
what is the relative strength of the effect of atomic radius?
large effect, the force of attraction falls off sharply with increasing distance
what is the effect of nuclear charge on ionisation energy?
the more protons there are in the nucleus of an atom, the greater the attraction between the nucleus and the outer electrons.
what is the effect of electron shielding on ionisation energy?
the greater the repulsion (shielding effect), the less the attraction between the nucleus and outer electrons. - inner-shell electrons repel outer-shell electrons
define second ionisation energy
the energy required to remove one electron from each ion in one mole of gaseous 1+ ions of an element to form one mole of gaseous 2+ ions
why is second ionisation energy always greater than first ionisation energy?
after the first electron is lost, the attraction on the remaining electron increases and more ionisation energy will be needed to remove this second electron
what does a large increase between ionisation energies suggest?
this is when an electron is removed from a different shell, the ionisation number with the greater ionisation energy is closer to the nucleus with less shielding
what is the trend in first ionisation energy across each period?
a general increase across each period
what is the trend in first ionisation energy between the end of one period and the start of the next period?
a sharp decrease (e.g H → Li , Ne → Na)
why does first ionisation energy increase across a period?
nuclear charge increases
same shell: similar shielding
nuclear attraction increases
atomic radius decreases
what is the trend in first ionisation energy down a group?
first ionisation energies decrease down a group
why does first ionisation energy decrease down a group?
atomic radius increases
more inner shells so electron shielding increases
nuclear attraction on outer electrons decreases
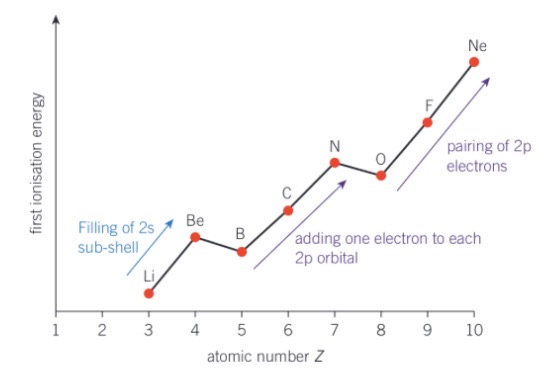
what causes the fall in first ionisation energy between Be → B
and N → O
filling of 2s sub-shell and adding an electron to each 2p orbital