chem exam 3
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
oxidation-reduction
reactions involving the transfer of electrons from one reactant to the other
aka redox reactions (may involve reaction of a substance with O2)
usually single replacement
for a free element to form an ion, the atom must lose or gain electrons
atoms that lose electrons are oxidized
atoms that gain electrons are reduced
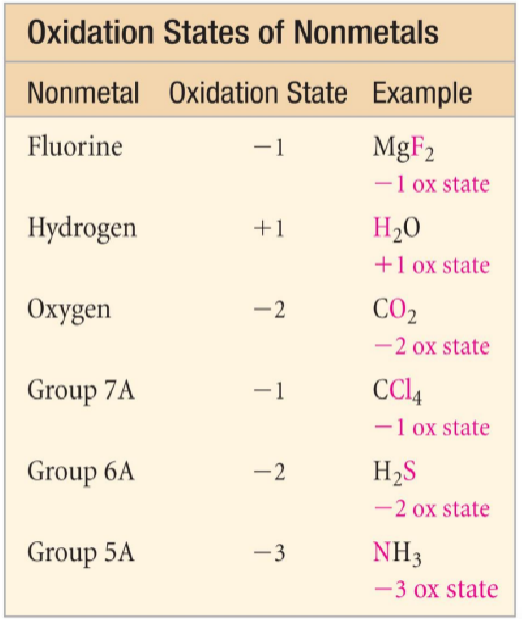
oxidation state rules
imaginary charges assigned based on a set of rules
NOT ionic charges (ionic charges are real, measurable charges)
rules (if two rules conflict, follow the rule that is higher on the list)
oxidation state of an atom in a free element or compound = 0
oxidation state of a monoatomic ion = charge of ion
sum of oxidation states of all atoms in:
a compound = 0
a polyatomic ion = charge of the ion
in a compound, metals have positive oxidation states
group 1A metals = +1
group 2A metals = +2
in compounds, non-mentals have negative oxidation state (table)
redox (OILRIG)
oxidation occurs when an element’s oxidation state increases on the product side (losing electrons, becoming more positive)
reduction occurs when an element’s oxidation state decreases on the product side (gaining electrons, becoming more negative)
oxidation and reduction must occur simultaneously (one atom loses an electron, another must accept the electron)
reactant that reduces an element in the other reactant is the reducing agent (oxidized)
reactant that oxidizes an element in the other reactant is the oxidizing agent (reduced)
waves
light can be described as a combination of oscillating electric and magnetic field that propagate through space as a wave
wavelength = the length of the smallest repeating unit of a wave (ex: node to node, peak to peak, trough or trough)
symbol is λ (lambda)
frequency = a measure of how many cycles of the wave pass by a specific point in a single second
units are cycles per second (s-1, 1/s or Hz)
symbol is f or v (nu)
c = 3.00 × 108 m/s = λv
light
both a wave nature and a particle nature (photon)
Max Plank discovered that photons have an energy
quanta of energy
E = hv
h (Plank’s constant) = 6.626 × 10-34 J x s
higher the frequency (lower wavelength), the more energy the photon has
ex: red visible light has a lower frequency compared to violet light
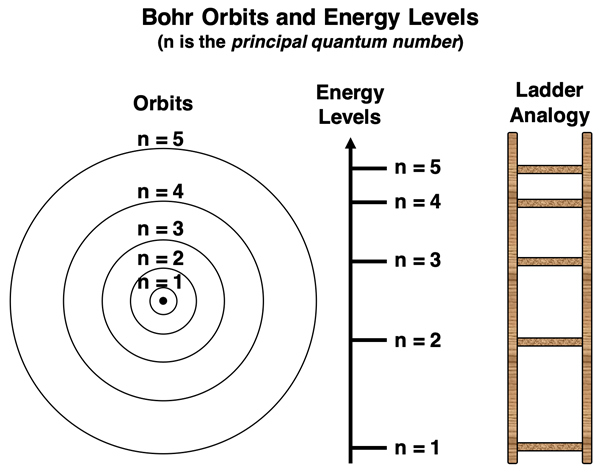
Bohr’s model
electrons can have only specific amounts of energy
fixed amounts - quantized
electrons travel in orbits that are a fixed distance from the nucleus (stationary states)
energy of the electron is proportional to the distance between the orbital and the nucleus
longer distance from nucleus = more potential energy
as electrons move farther from the nucleus, the electrostatic attraction weakens, and they occupy higher energy levels, making them less tightly bound to the atom
electrons emit energy energy when “jump” from an orbital of higher energy to an orbital of lower energy
distance between orbitals determines energy of emitted light
determinacy vs indeterminacy
classical physics = particles move in a path determined by the particle’s velocity, position, and forces acting upon it
determinacy = definite, predictable future
electrons are indeterminant, indefinite (cannot know both position and velocity, cannot predict path it will follow)
wave functions
calculations show that an orbital’s size, shape, and orientation in space are determined by three integer terms in the wave function (added to quantize the energy of e-)
integers are called quantum numbers
principle quantum number, n
angular momentum quantum number, l
magnetic quantum number, ml
principle quantum number (n)
n characterizes energy of an electron in a particular orbital
corresponds to Bohr’s energy level
n can be any integer greater than 1
energies are negative (an electron has E = 0 when it just escapes the atom)
larger the value of n = large the orbital
as n increases, distance between levels decreases
angular momentum quantum number (l)
determines shape of an atomic orbital
has integer value from 0 to (n-1)
each value of l has a letter to designate orbital shape
s orbitals are spherical (l=0)
p orbitals are like two balloons tied at the knots (l=1)
d orbitals are mainly like 4 balloons tied at the knot (l=2)
f orbitals are mainly like 8 balloons tied at the knot (l=3)
s orbital
each principal energy state has one’s orbital (l=0)
lowest energy orbital in the principal energy state
spherical in shape
number of nodes = 0
p orbital
each principal energy state above n=1 has three p orbitals
ml = -1, 0, +1
each p orbital points along a different axis
px, py, pz
second-lowest energy orbitals in an energy state
two-lobed in shape
number of lobe = (n)
node (gap) at the nucleus
d orbital
each principal energy state above n=2 has five d orbitals
ml = -2, -1, 0 +1, +2
four d orbitals aligned in separate plans; 5th aligned with z-axis
dxy, dxz, dyz, dx2-y2, dz2
third-lowest energy orbitals in a principal energy state
most are 8-lobed; some are two-lobed with a toroid (donut shape)
planar nodes; higher principle energy levels also have spherical nodes
f orbitals
each principal energy spate above n=3 has seven f orbitals
ml = -3, -2, -1, 0, +1, +2, +3
fourth-lowest energy orbitals in a principal energy state
more lobes and nodes than d-orbitals
electron spin
electrons spin on an axis and generate a magnetic field
all electrons have the same amount of spin
orientation of spin is quantized
only in one direction
spin up or spin down
spin quantum number
describes how an electron spins on its axis
clockwise or counterclockwise
spin up or spin down
for a given orbital, spins must cancel
paired
values of ms are +1/2 or -1/2
Pauli exclusion principle
electrons in an atom cannot have the same set of 4 quantum numbers
no orbital may have more than 2 electrons
electrons in an orbital must have opposite spins
electron configurations
the ground state of the electron is the lowest energy orbital it can occupy
distribution of an atom’s electrons into orbitals
the number of designates the principal energy level (n)
the letter designates the sublevel and type of orbital (l)
the superscript designates the number of electrons in the sublevel
orbital diagrams
sublevels in each principal energy shell of H have the same energy
orbitals with the same energy are degenerate
for multielectron atoms, energies of the sublevels are split
caused by electron-electron repulsion
lower values of the l quantum number, have lower energy
s < p < d < f
orbital diagrams cont.
sublevels within an energy level are not degenerate cont.
penetration of the fourth and higher energy levels, is so strong their their s sublevels are lower in energy than the d sublevels of the lower energy level
energy difference between levels decreases for higher energy levels (and can cause anomalous electron configurations for certain elements)
orbital diagrams cont. 2
energy shells fill from lowest to highest energy
sublevels fill from lowest energy to highest
s → p → d → f
Aufbau principle
orbitals that are in the same sublevel have the same energy
no more than two electrons in each orbital
Pauli exclusion principle
when filling degenerate orbitals, place one electron in each before pairing
Hund’s rule
valence electrons
the electrons in all the subshells with the highest principal energy shell
electrons in lower energy shells are called core electrons
one of the most important factors in the way an atom behaves, both chemically and physically, is the based on the number of VE
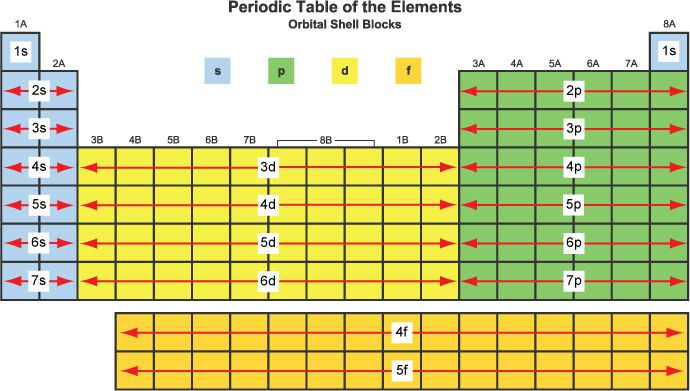
electron configuration, VE, periodic table
for main group/representative elements:
group number (up and down) = number of valence electrons
number of columns in each “block” is the maximum number of electrons that sublevel can hold
period number corresponds to the principal energy level of the VE
electron configuration exceptions
because of sublevel splitting, the 4s sublevel is lower in energy than the 3d, and therefore the 4s fills before the 3d
difference in energy is not large
for some elements, the (n)s only partially fills before the (n-1)d or doesn’t fill at all
therefore, their electron configurations must be found experimentally
ex:
Cr = [Ar]4s23d4 → [Ar]4s13d5
Cu = [Ar]4s23d9 → [Ar] 4s13d10
Mo = [Kr]5s24d4 → [Kr]5s14d5
Pd = [Kr]5s24d8 → [Kr]5s04d10
becod sublevel is one away from being half full (5 electrons) or full (10 electrons), electrons from the s sublevel will move to the d sublevel
becoming more noble-like
alkali metals have one more electron than the previous noble gas
alkali metals tend to lose their extra electron, resulting the same electron configuration as a noble gas
cation with a 1+ charge is formed
halogens have one fewer electron than the next noble gas
halogens tend to gain one electron to attain the electron configuration of the next noble gas
anions with 1- charge is formed
atomic radius
increases down a group
valence shells farther from nucleus (more n levels)
effective nuclear charge fairly similar
decreases across period
adding electrons to same valence shell
effective nuclear charge increases
valence shell held closer
ionic radius (transitional metals)
for transition metals:
atomic radius increases down the group
radii nearly the same within a d-block
valence electrons are ns2 NOT d electrons
effective nuclear charge on ns2 electrons approx. the same
cations form when atom loses VE
for transition metals, d-block electrons may also be lost
Al atom → 1s22s22p63s23p1
Al3+ ion → 1s22s22p6
Fe atom → 1s22s22p63s23p64s23d6
Fe2+ ion → [Ar]3d6
F23+ ion → [Ar]3d5
ionic radius trends
ions in a group have the same charge
ion size increases moving down the group (higher valence shell = larger radius)
cations are smaller than the neutral atom
anions are larger than the neutral atom
cations smaller than anions
except Rb+ and Cs+; comparable to or larger than F- and O2-
for species with the same electron configuration: isoelectronic
larger positive charge = smaller cation
larger negative charge = larger anion
magnetic properties of transition metals
electron configuration with unpaired electrons result in atoms/ions with a net magnetic field
paramagnetic (ex: Mn → [Ar] 4s23d5)
atom/ion attracted to an external magnetic field
electron configuration with all paired electrons result in atoms/ions with no magnetic field
diamagnetic (ex: Zn → [Ar]4s23d10)
slightly repelled by an external magnetic field