Grade 12 Biology - Biochemistry TEST #1
1/163
Earn XP
Description and Tags
You will get that 95+
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
164 Terms
Biochemistry
the study of chemical processes in
living organisms.
Elements
A pure chemical substance that cannot be broken
down into simpler substances.
Compounds
Substances that contain two or more elements
chemically combined
Molecules
A type of compound made by covalently
bonding two or more atoms together
Organic Molecules
A carbon-containing molecule in which
carbon atoms usually are bonded to hydrogen
Inorganic Molecules
Molecules that do not contain carbon
Atom
The smallest unit of an
element which retains all of
the properties of the element
Representing Atoms
Element symbol
Atomic Number & Atomic Mass
Atomic Number = # Protons = # Electron
Atomic Mass = Protons + Neutrons
Isotopes
Atoms of the same element that have the same
number of protons
Radioactivity
Radioactive decay involves the spontaneous
transformation of one element into another.
Radioisotopes
Isotopes that have unstable nuclei and undergo radioactive decay.
Half-Life
The time it takes for one half of the
nuclei to decay
Radioactive Dating (for Carbon-14)
Plants absorb a mixture of radioactive (C-14) and non-radioactive (C-12) carbon dioxide from the air and water for photosynthesis and then Scientists can calculate the time that has passed since an organism's death by measuring the ratio of C-12 to C-14 in a dead or fossilized organism
Radioactive Tracers
Radioactive isotopes are used in biological
Nuclear Medicine
Uses radioactive materials either to image a patient's body or to destroy diseased cells
Orbital
Electrons occupy volumes of space (3-D) around
the nucleus called orbitals or energy levels
Valence shell full of electrons = HAPPY =
STABLE ATOM
Electronegativity
A measure of an atom's ability to attract/pull a shared electron pair in a covalent bond
Electronegativity number (EN) - the larger the number
Intramolecular Bonds
Forces that hold atoms together
within a molecule
Non-Polar (Pure) Covalent Bond
Two atoms SHARE electrons (nearly) EQUALLY
Occurs between:
Atoms of the same element (∆EN = 0) *All H O F Br I N Cl elements.
Atoms of of different elements (∆EN < 0.3)
Single
Polar Covalent Bonds
Two atoms SHARE electrons
UNEQUALLY
∆EN = 0.4 - 1.7
One atom is slightly negative (δ -) and the
other is slightly positive (δ +)
Polar molecules have dipoles because of oppositely charged ends
covalent bond
Covalent bonds are bonds formed between two nonmetal elements that share electrons.
Ionic Bond
- Formed by a TRANSFER of electrons from atom to atom (usually metal to non-metal)
- ∆EN= 1.7 - 3.3
- Ions are atoms that have obtained a stable valence shell by losing or gaining electron
Atom losing electrons
→ (+) charge → cation
Atom gaining electrons
→ (-) charge → anion
Electronegativity scale
0-0.3 = Non polar covalent
0.4-1.7 = Polar covalent
1.7-3.3 = Ionic
Intermolecular Bonds
Forces of attraction between molecules
They are weaker than intramolecular bonds
Hydrogen Bonding
A weak association between an atom with
partial negative charge and a hydrogen with
partial positive charge
Hydrophobic Interactions
Polar Molecules = hydrophilic ("water-loving")
Non-Polar molecules = hydrophobic ("water-hating")
Non-polar molecules have a tendency to clump together when mixed with polar molecules
Carbon Chemistry
Compounds that contain carbon (a few exceptions) are
called organic compounds
Carbon
- Carbon can form up to 4 bonds with other atoms (contains 4 valence electrons) giving it covalent compatibility with many different elements
- May form single
Synthetic Organic Chemistry
- The biomaterials industry develops synthetic substances that can
integrate well with living tissues - Biomedical engineers design
Molecular Formula
Shows the number of each atom in an
element or compound
Structural Formula
Shows how the different atoms are bonded together
Skeletal Formula
Shows each carbon atom as an end or bend in the chain of the molecule
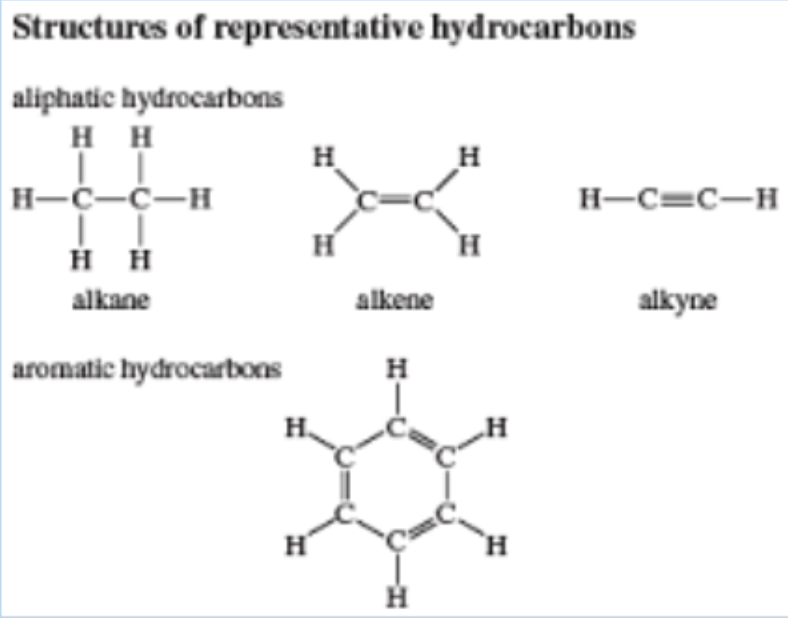
Hydrocarbons
Molecules containing only carbon and hydrogen
atoms
Functional Groups
Parts of molecules involved in chemical
reactionsElements like H, O, S, N, and P (and form reactive clusters)
Often make a molecule polar
R - Group
- "Radical" group
- An abbreviation for any group where a carbon or hydrogen is attached to the rest
- It is NOT a specific functional group
Hydroxyl (-OH)
Polar due to the electronegative oxygen atoms
Water molecules are attracted to hydroxyl group
Names usually end in -ol
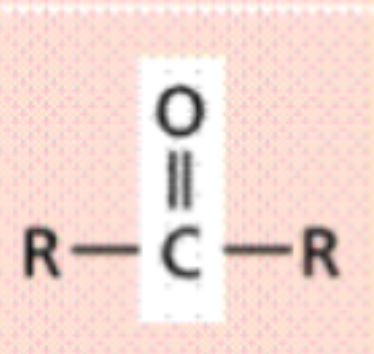
Carbonyl (-CO)
Carbon is joined to the oxygen by a double covalent bond
Polar (hydrophilic)
Aldehyde is formed when this group
is on the end of a carbon skeleton
and the carbon is bonded to a
hydrogen (R-CHO)
Ketone is formed when a
carbonyl group is
not on the end of a (R-CO-R’)
Aldehyde
is formed when this group
is on the end of a carbon skeleton
and the carbon is bonded to a
hydrogen (R-CHO)
Ie. Formadehyde (CH₂O)
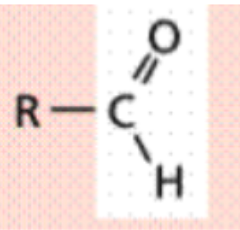
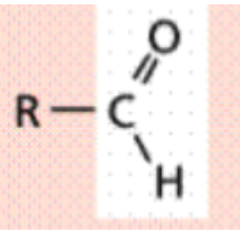
Functional Group?
Carbonyl - Aldehyde
Functional group?
Carbonyl - Ketone
Ketone
is formed when a
carbonyl group is
not on the end of a chain
(R-CO-R’)
Ie. Acetone (CH3COCH3)
Carboxyl (-COOH)
Carbon atom double bonded to an oxygen atom
and single bonded to a hydroxyl group (-OH)
Polar (hydrophilic)
Source of hydrogen ions (H+), which makes a
molecule acidic. These organic acids are called
carboxylic acids.
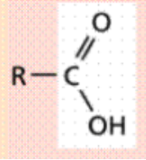
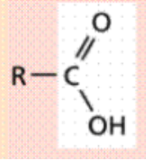
Functional Group?
(-COOH) Carboxyl - acids
carboxylic acids.
Source of hydrogen ions (H+), which makes a molecule acidic.
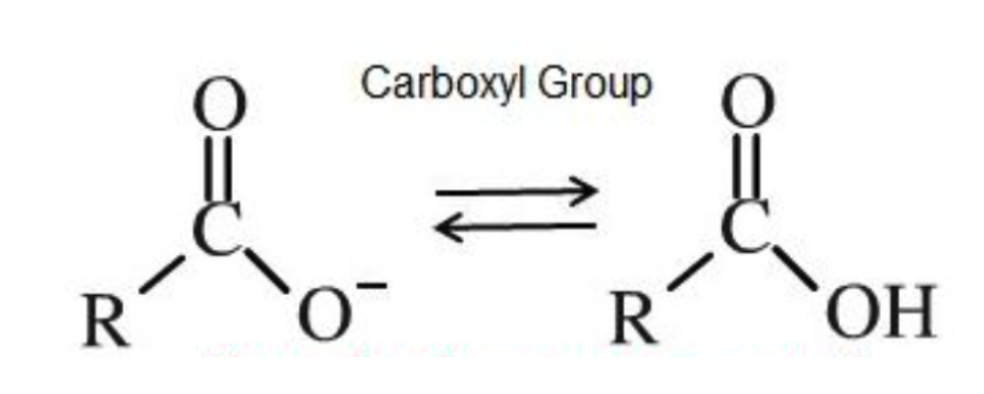
Amino (-NH2)
Nitrogen atom bonded to a hydrocarbon radical and two
hydrogen atoms
Polar (hydrophilic)
Molecule is basic. These molecules are organic bases
called amines.
Act as a base, picking up protons (H+) from the surrounding
solution.
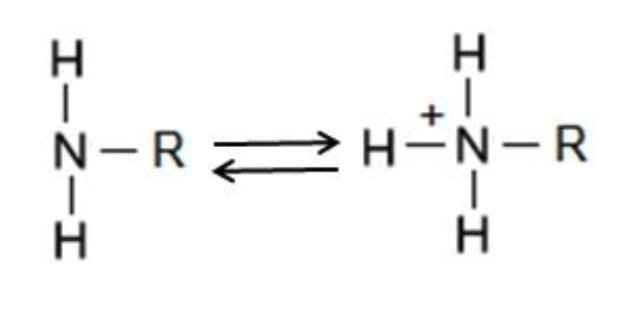
Functional Group?
Amino group = molecule is basic + bases are called amines
Sulfhydryl (-SH)
Sightly polar (hydrophilic)
Help stabilize structures of
proteins
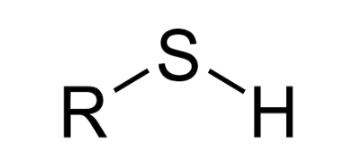
Phosphate (PO₄⁻³)
Polar (hydrophilic)
Negatively charged
Molecule is acidic
Found in molecules that make up the
cell membrane, as well as DNA and
RNA
Transfer of energy between organic
molecules (ATP)
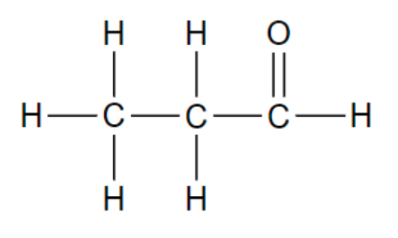
Functional group in these molecules?
Aldehyde
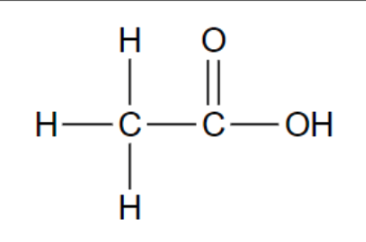
Functional group in these molecules?
Carboxylic acid
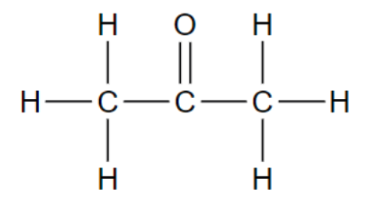
Functional group in these molecules?
Ketone
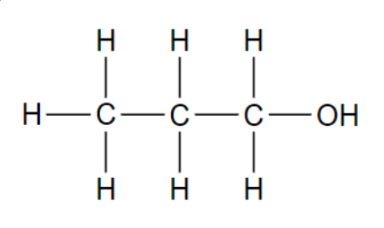
Functional group in these molecules?
Alcohol
Macromolecules
large organic molecule
polymer
large molecule consisting of many identical or similar subunits connected
together
monomer
subunit or building block molecule of a polymer
4 classes of macromolecules
Carbohydrates
Lipids
Proteins
Nucleic acids
Catabolic Reaction
Breakdown of large molecules into smaller ones
Releases energy in the process
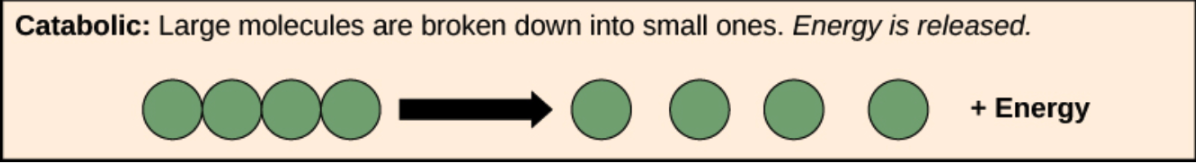
Anabolic Reaction
Buildup of smaller molecules into larger ones
Requires energy input

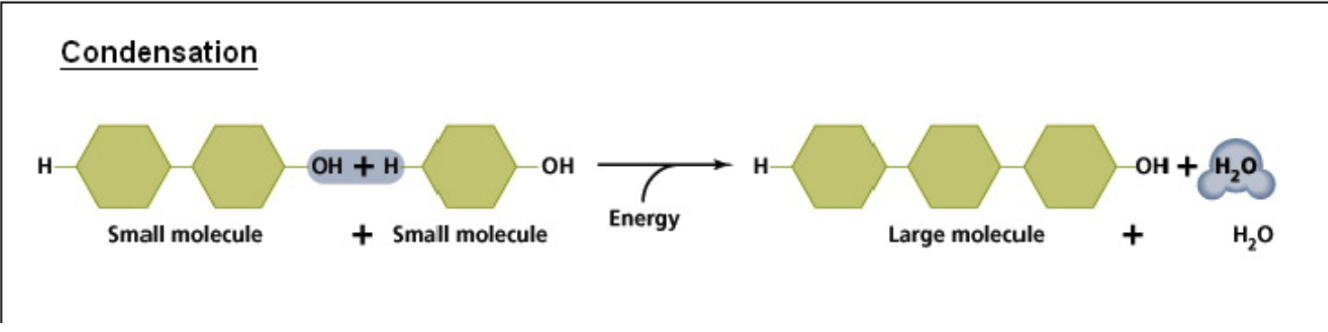
Condensation (Dehydration Synthesis) Reaction
Joining smaller molecules into larger ones with the input of energy (e.g.:
ANABOLISM)
Releases a WATER molecule as a result
An H atom from the functional group of one molecule combines with an OH group from another molecule to form H-OH (i.e.: Water)
The resulting molecules are linked together
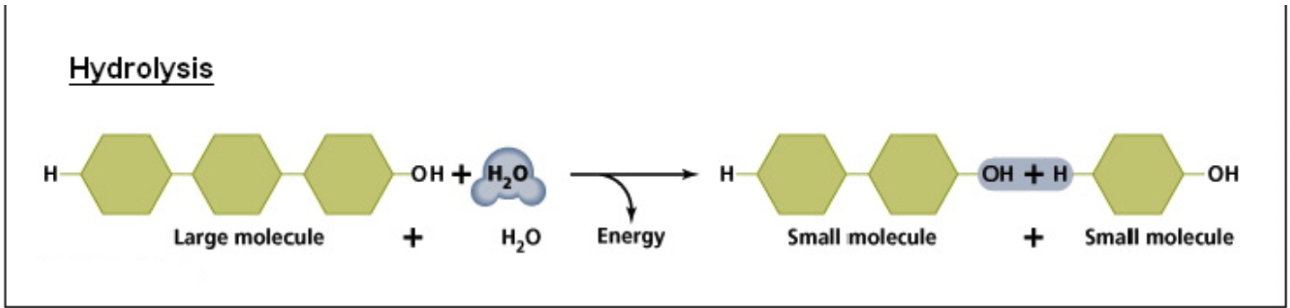
Hydrolysis Reaction
This process involves the ADDITION of water to break the bonds between large molecules to form smaller molecules (i.e.: Catabolism)
Releases energy in the process
The H atom from water is added to one molecule, while the OH group is added to the other
Enzymes are involved as well to help speed up these reactions (catalyst)
Neutralization Reactions
ACIDS
Release H+ ions when dissolved in water
Proton donors
Ex. HCl 🡪 H+ + Cl-
BASES
Release OH- ions when dissolved in water
Proton acceptors
Ex. Ca(OH)2 🡪 Ca2+ + OH-
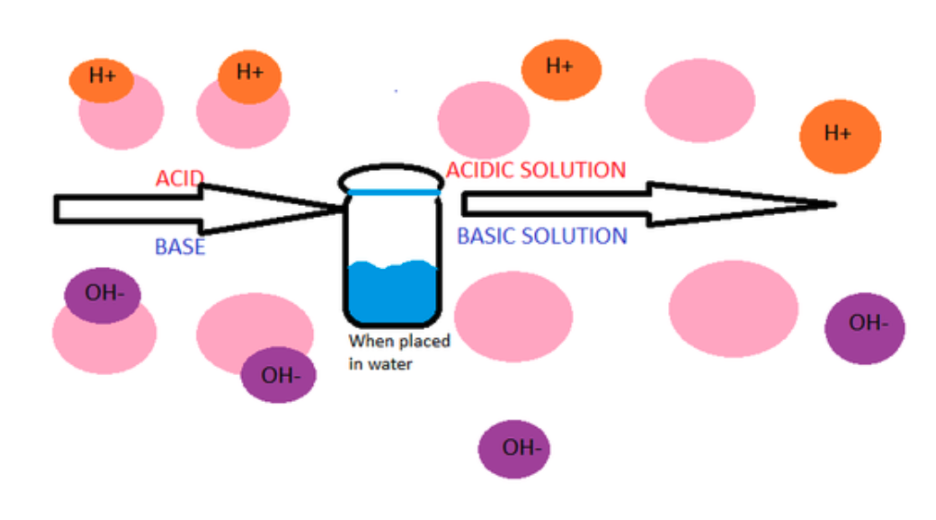
What happens when you mix an acid and a base?
Acid + Base 🡪 Water + Salt
Oxidation Reduction (Redox) Reactions
Transfer of electrons between molecules
OXIDATION = loss of electrons by a molecule
REDUCTION = gain of electrons by another molecule
These reactions occur at the same time - when one molecule undergoes oxidation, the opposite must also occur
In biological systems, redox reactions involve the removal of H
atoms from one molecule and the addition of H atoms to
another molecule
Function of Carbohydrates
Used as sources of ENERGY
Cell surface markers for cell-to-cell communication
Building materials
Carbohydrates
Contain C, H, O in a 1:2:1 ratio
Formula: (CH2O)n (n = # of Carbons)
Sugar names end in –ose
Simple Carbohydrates:
Monosaccharide and Disaccharide
Complex Carbohydrates:
-Polysaccharide and Oligosaccharide
MONOSACCHARIDE
Single sugar molecule (smallest monomer of a carbohydrate)
Formula: C6H12O6
Examples: Glucose, Fructose, Galactose
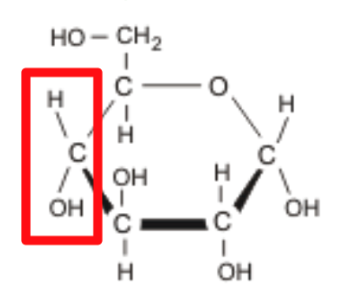
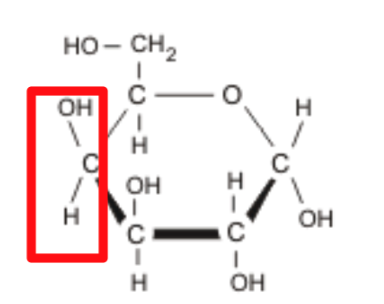
What is this?
Glucose
What is this?
Galactose
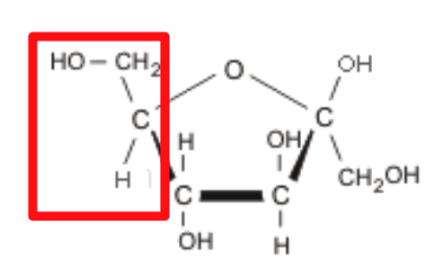
What is it?
Fructose
Carbohydrates are Characterized
1.) Number of carbons
ie. ribose has 5 carbons, glucose has 6
carbon
2.) Functional Groups
An –OH group is attached to each carbon
except one, which is double bonded to a
carbonyl group (=O)
glucose and galactose have an aldehyde
group while fructose has a ketone group
Isomers
are monosaccharides with the same chemical formula but
different arrangement of atoms
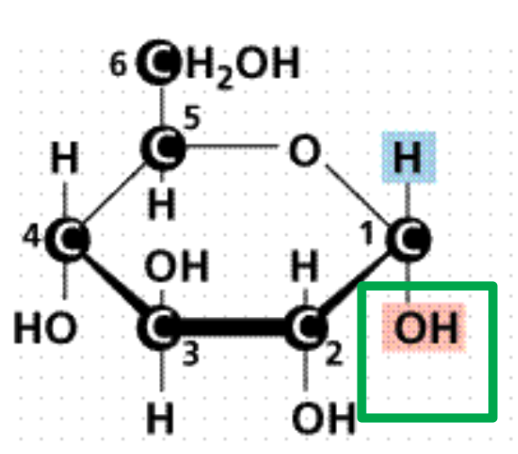
What is it?
Alpha glucose
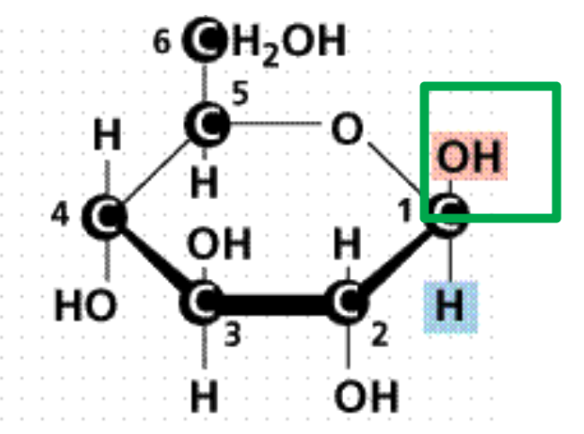
What is it?
Beta Glucose
Disaccharides
composed of two monosaccharides (monomers) joined through a
condensation (dehydration synthesis) reaction, forming a
glycosidic linkage (covalent bonds)
Link between C1 of a glucose molecule and C4 of a second
glucose
Glucose + Glucose
Maltose
Glucose + Fructose
Sucrose
Glucose + Galactose
Lactose
FORMATION OF A DISACCHARIDE:
DEHYDRATION SYNTHESIS: Hydroxyl group (-OH) of 1 sugar is combined with a Hydrogen atom (-H) of another
sugar, forming a water molecule
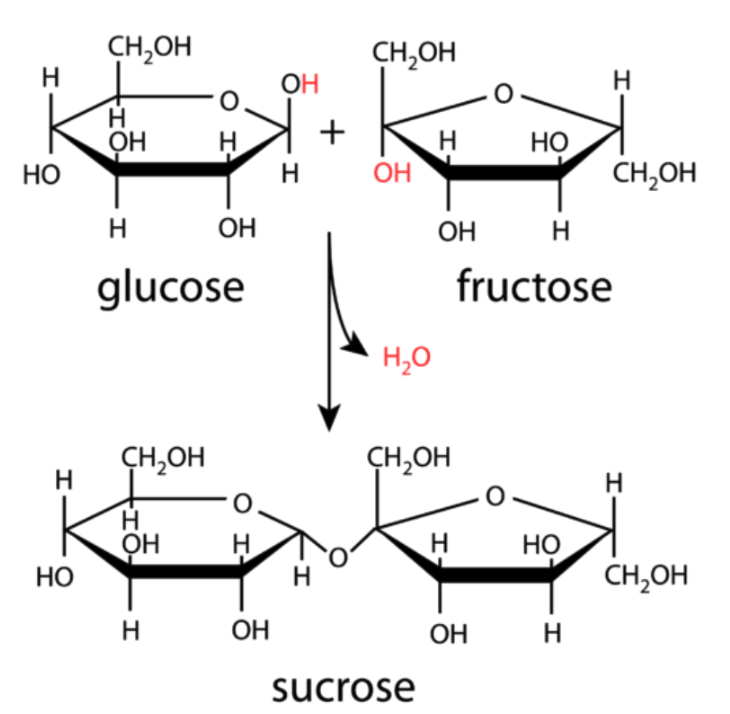
FORMATION OF A DISACCHARIDE (2)
HYDROLYSIS
Opposite of dehydration synthesis
In order to separate disaccharides back into their
component monosaccharides, 2 things are needed:
Water
A specific ENZYME (biological chemical that speeds up reaction
rates)
Maltase (separate maltose into 2 glucose)
Sucrase (separate sucrose into glucose & fructose)
Lactase (separate lactose into glucose & galactose)
Oligosaccharides
3-10 monosaccharides linked
glucose + galactose + fructose = Raffinose
Found in beans, peas, lentils, broccoli,
asparagus
*Humans lack enzymes to digest oligosaccharides (causes bloating,
cramps, gas)
Polysaccharides
More than 10 monosaccharides linked
Most are made up of hundreds of monosaccharides bonded together
- Types:
Starch: glucose storage in plants *long term energy
2. Glycogen: glucose storage in animals *short term energy
3. Dietary Fiber: not used for energy (not digestible by humans)
-Cellulose: structural support in plants
-Chitin: structural support in organisms
Glycosidic bonds in Starch
Starch
A starch molecule contains hundreds of glucose molecules in
either
branched chains: Amylopectin or
unbranched (coiled) chains: Amylose
Straight chain polymer of α glucose 1-4 glycosidic
linkages
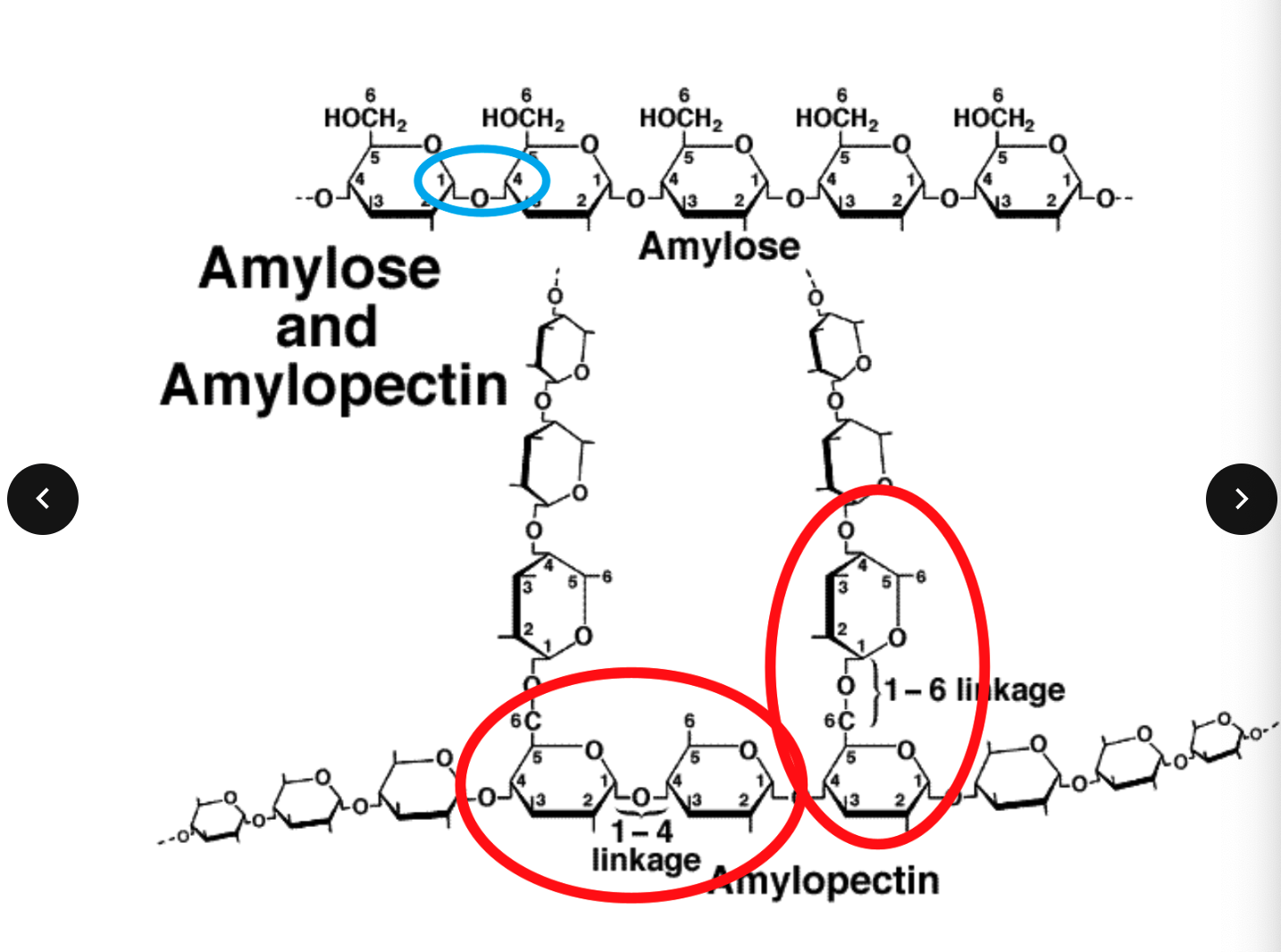
Glycogen
Found in liver and skeletal muscles
Many branch points allows for rapid break down
for glucose to be released and used for energy

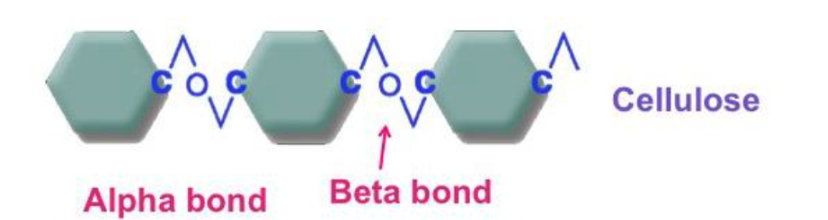
Cellulose
Structural support in plant cell walls
Difficult for humans to digest so used as fibre in diets
Also used by humans in
wood for lumber and paper
cotton and linen for clothing
Straight chain polymer of β glucose 1-4 glycosidic
linkages
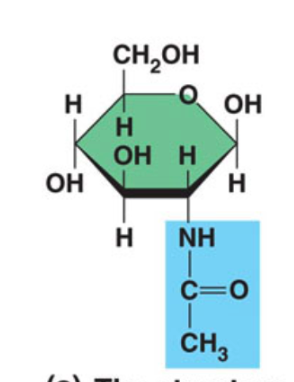
Chitin
Add nitrogen containing group to C2
Structure support - exoskeleton of insects,
crabs, lobsters, fungi cell wall
Also used in medicine: contact lenses,
surgical thread
What is the function of lipids?
composed of carbon, hydrogen, and oxygen atoms
higher proportion of non-polar C-H (high energy) bonds
makes lipids extremely hydrophobic (insoluble in water)
Provides long-term energy storage, cushions and insulates
organs, cell membrane structure, synthesis of hormones
Gram for gram, fats contain more stored energy than
carbohydrates - due to high proportion of C-H bonds
Fat
38 kJ or 9kcal of energy/g
Carbs:
17 kJ or 4kcal of energy/g
Lipids (Types)
1) Triglycerides (fats)
2) Phospholipids
3) Steroids
4) Waxes
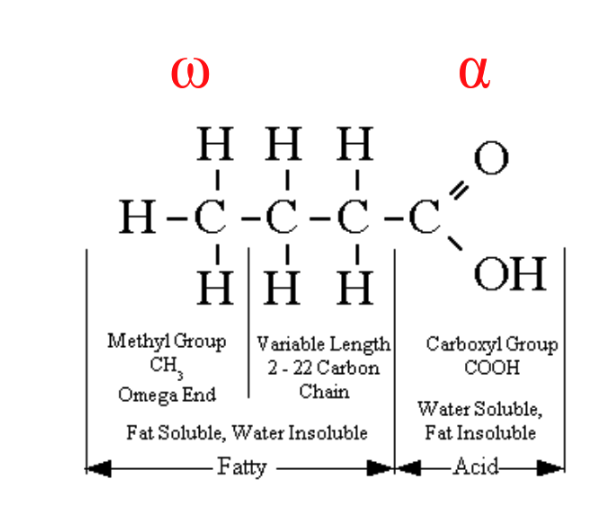
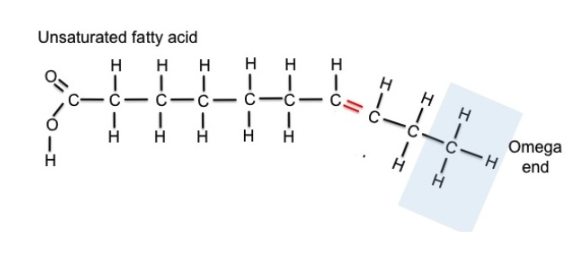
Fatty Acids
The building block
(monomer) of lipids
Chain of carbon atoms
Carboxyl group (-COOH)
at alpha (α-) end
Methyl group (-CH3) at
omega (ω-) end
Lipids yield double the
amount of energy as
carbohydrates per gram
Glycogen is more accessible
to break down; carbs are
used up before lipids are
broken down
How are fatty acids characterized?
Length of carbon chain
Saturation
Degree of Saturation
Location of double bonds
Hydrogenation
Orientation of hydrogen around double bond
Length of Carbon Chain (Lipids Characterization)
-Short-chain fatty acids (<8 carbons)
- Medium chain fatty acids (8-12 carbons)
- Long chain fatty acids (>12 carbons)
Saturation (Lipids Characterization)
Saturated fatty acids:
have only single bonds between C atoms
contain maximum # of H atoms possible
Unsaturated fatty acids:
have one or more C-C double bonds
fewer than maximum # of H atoms possible
formed by removing H atoms from molecule
Double bonds create a “kink” or bend in
shape
Double bonds create oils/liquids because
they prevent molecules form packing
together closely enough to solidify
Saturated fatty acids:
have only single bonds between C atoms
contain maximum # of H atoms possible
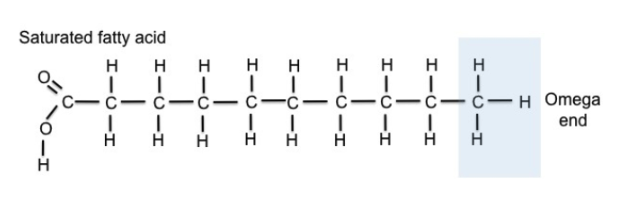
Unsaturated fatty acids:
have one or more C-C double bonds
fewer than maximum # of H atoms possible
formed by removing H atoms from molecule
Double bonds create a “kink” or bend in
shape