17. Therapeutic Antibodies I
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
What is Ehrlich’s ‘Magic Bullet’ concept?
Drug that targets origin, rather than symptom, of disease without causing harm to patient.
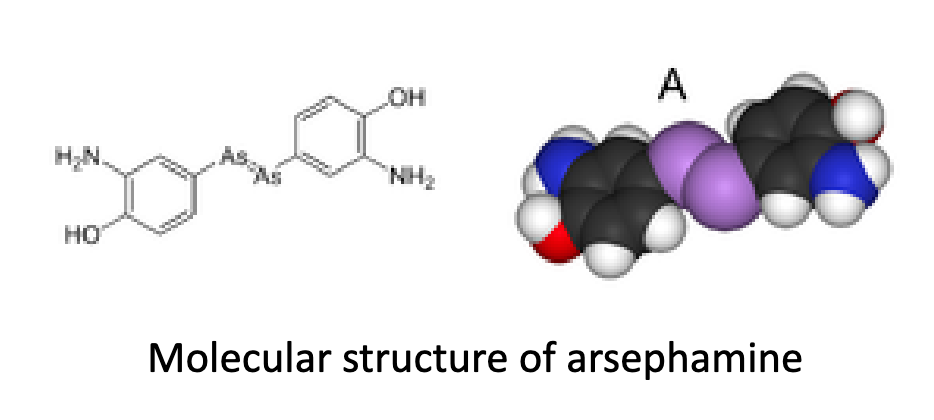
What was the first success of the magic bullet concept?
The development of Salvarsan (arsephamine), an arsenic compound effective against syphilis.
What is the ‘Rule of 5’ in small molecule therapuetics?
Lipinski rules of five (developed at Pfizer) help predict if a biologically active molecule is likely to have the chemical and physical properties to be orally bioavailable.
Mr ≤ 500
5 H bond donors
10 H bond acceptors
clogP <5 (measure of lipophilicity i.e. compound solubility in aqueous buffer)
What are some exception to the rule of 5?
Vancomycin (Mr 1449)
Venetoclax (Mr 868, cLogP 10.4)
Grazopevir (Mr 767, 15 H bond acceptors)
What is the difference between protein and 'small molecule’ therapeutics?
Specificity (discrimination) hinges on non-covalent interactions.
Large-sized molecules provide more contact points (larger SA), granting a greater potential for specificity-conferring interactions
Lipinski molecule: 5 H donors; 10 H acceptors
Protein molecules: numerous H bond acceptors/donors; ionic, vdW, hydrophobic interactions.
What are protein-based therapeutics beyond Abs?
Enzymatic/regulatory activity (Group I)
Specific targeting activity (Group II)
Protein vaccines (Group III)
Protein diagnostics (Group IV)
What are the characteristics of Group I protein-based therapeutics?
Enzymatic or Regulatory Activity
Ia: replacing deficient/abnormal protein
Ib: augmenting the existing pathway
Ic: providing a novel function of activity
Examples: insulin, factor VIII
What are the characteristics of Group II protein-based therapeutics?
Specific Targeting Activity
IIa: interfering with a molecule or organism
IIb: delivering compounds or proteins
Examples: herceptin, infliximab
What are the characteristics of Group III protein-based therapeutics?
Protein Vaccines
IIIa: protecting against deleterious foreign agents
IIIb: treating an autoimmune disease
IIIc: treating cancer
What are therapeutic antibodies?
A subset of protein-based therapeutics that make use of the specificity by which each antibody recognises only one antigen, pinpointed attacks on specific antigens are possible.
Also known as: biological therapeutics, ‘biologics’ (biological medical product), biopharmaceutical.
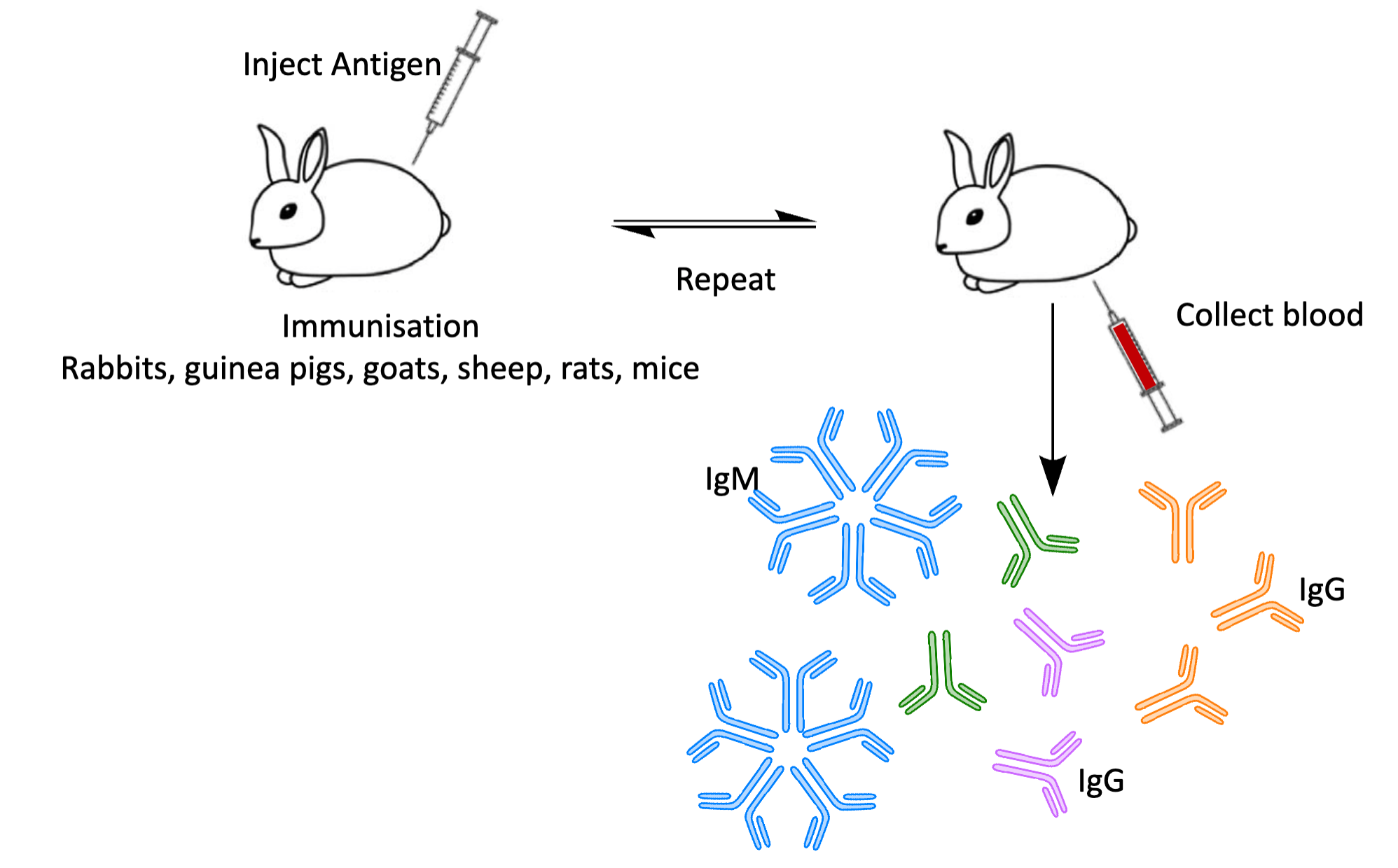
How are polyclonal antibodies made?
An antigen is injected into a mammal (e.g., rabbit, goat) to elicit an immune response.
The animal's immune system generates multiple B-cell clones, each producing antibodies targeting different epitopes on the antigen.
After a series of booster injections, blood is collected.
Serum is separated, containing a heterogeneous mix of antibodies (polyclonal), distinct in sequence and isotope.
These antibodies are purified for use.
Polyclonal antibodies recognise multiple epitopes on the same antigen, providing robust binding but with batch-to-batch variability.
What are polyclonal antibodies?
Antibodies secreted by different B cell clones- a collection of immunoglobulin molecules that react against a specific antigen, each identifying a different epitope.
Describe the proliferation and differentiation phases of polyclonal antibody production.
Proliferation: Following BCR activation by a specific antigen, the B-cell undergoes clonal expansion. All daughter cells express the same BCR as the original B-cell, ensuring antigen specificity is maintained.
Differentiation: Some of these clonal B-cells differentiate into plasma cells, which secrete soluble antibodies. These antibodies are identical in variable regions (especially the CDR loops) to the original BCR, enabling recognition of the same epitope.
In a polyclonal response, multiple B-cells recognising different epitopes of the same antigen undergo this process in parallel.
How do polyclonal and monoclonal antibodies differ?
Polyclonal antibodies represent a population of antibodies that are specific for the immunising agent, but differ in amino acid sequence as they come from different B cell clones.
Monoclonal antibodies result from plasma cells that are descendants from a single B cell clone.
Why are polyclonal antibodies generally not suitable for therapeutic use?
Heterogeneity: They bind to multiple epitopes on the same antigen, which may interfere with therapeutic precision (e.g., if only one epitope is functionally relevant).
Variable affinity: Different antibodies in the mix may have varying affinities, reducing consistency and potency.
Cross-reactivity: Increased risk of binding to off-target antigens, potentially triggering unintended immune responses.
Batch variability: Each production round yields a slightly different antibody profile, complicating quality control.
Higher side-effect risk: Due to off-target effects and immune activation.
Instead, monoclonal antibodies offer consistent, high-specificity binding to a single epitope, critical for safe and effective therapy.
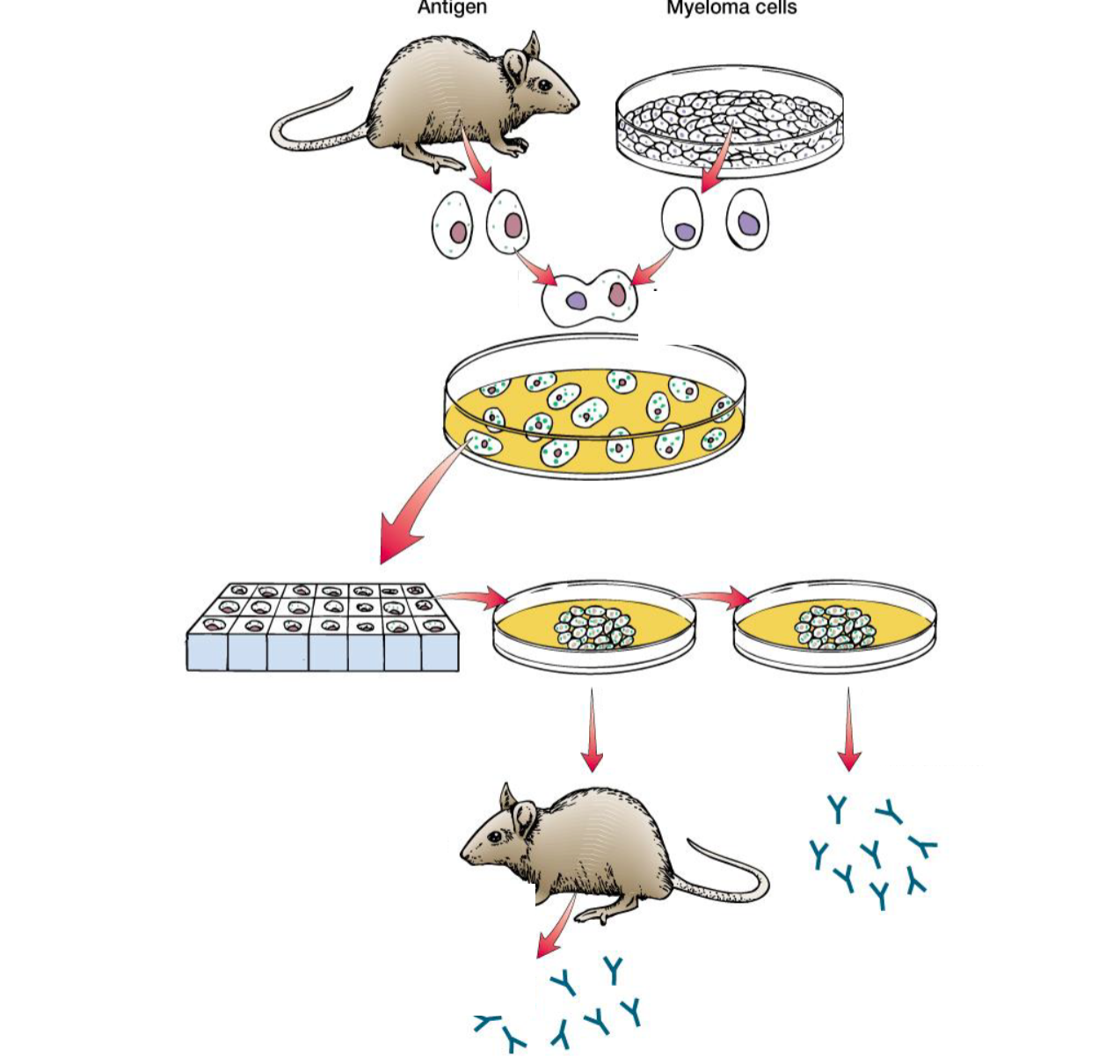
How are monoclonal antibodies made?
Hybridoma Cell Technology. Each monoclonal antibody produced is identical and targets a single epitope, ensuring consistency and specificity.
Immunisation: Animal is immunised with a specific antigen to elicit an immune response.
Lymphocyte harvesting: B-cells producing antigen-specific antibodies are isolated from the animal's spleen.
Cell fusion: These B-cells are fused with immortal myeloma cells (cancerous plasma cells) to form hybridoma cells.
Selection (HAT medium): Cells are cultured in HAT medium. Only fused hybridoma cells survive.
Screening: Hybridomas are screened for those producing the desired antibody.
Cloning: Positive hybridomas are cloned to ensure monoclonality.
Expansion and cryopreservation: Clonal hybridoma lines are expanded in culture and cryopreserved for long-term use.
Antibody production & purification: Monoclonal antibodies are harvested from the culture medium and purified.
What are the problems of hybridoma technology?
Labour-intensive
Low yield
Genetic instability
mAbs of murine origin- human immune system detects mAbs as non-self
How do HGPRT and Tk enzymes enable hybridoma survival in HAT selection?
HAT medium blocks de novo DNA synthesis (via aminopterin), forcing cells to use the salvage pathway.
HGPRT enables use of hypoxanthine; Tk enables use of thymidine.
Myeloma cells used are HGPRT⁻/Tk⁻, so they can't survive.
B-cells are HGPRT⁺/Tk⁺ but die naturally.
Hybridomas inherit both enzymes, allowing DNA synthesis via the salvage pathway → they survive.
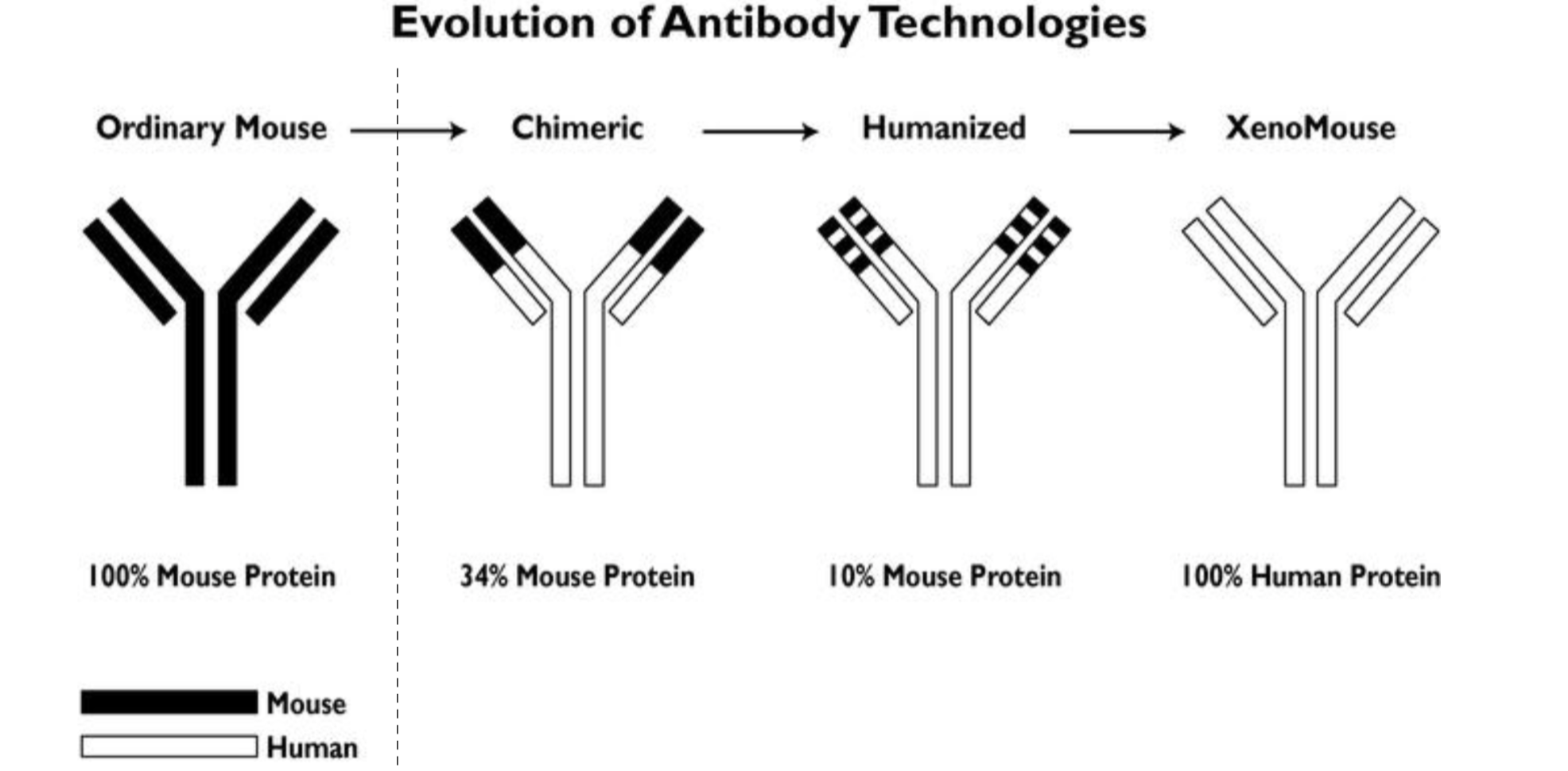
How is immune rejection reduced in therapeutic antibody development?
Therapeutic antibodies are progressively "humanised" to reduce immunogenicity in patients.
Mouse antibody: Fully murine → high risk of immune rejection.
Chimeric antibody (~34% mouse): Mouse variable regions + human constant regions.
Humanised antibody (~10% mouse): Only mouse CDR loops grafted onto human framework.
Fully human antibody (0% mouse): Generated via transgenic mice (e.g. XenoMouse) with human immunoglobulin genes, or phage display or B-cell cloning from human donors.
Reducing murine content lowers immunogenicity and improves clinical tolerance.
How does hybridoma technology differ from traditional antiserum (polyclonal antibody) production?
Aspect | Polyclonal Antiserum Production | Hybridoma (Monoclonal) Technology |
|---|---|---|
Source | Serum from immunised animal | Fused B-cell + myeloma (hybridoma) |
Antibody specificity | Mixed; many B cells = many epitopes | Single specificity (from one B cell clone) |
Antibody type | Polyclonal (heterogeneous) | Monoclonal (homogeneous) |
Production consistency | Variable between animals/batches | Consistent from immortal cell line |
Reproducibility | Limited | High |
Animal use | Any mammal | Mostly mice (or rats) |
Use case | General lab assays, detection | Therapeutics, diagnostics, research |
What are chimeric antibodies?
Link genes for mouse variable regions and human constant regions
~70% human sequence.
What are humanised antibodies?
CDR grafting.
DNA sequences encoding the CDRs from mouse genes are linked to human genes.
Still get human anti-mouse responses.
How are chimeric antibodies made?
Identify a mouse monoclonal antibody (mAb) with the desired antigen specificity.
Isolate the rearranged variable regions (V-J and V<D-J) from the hybridoma’s light and heavy chain genes.
Ligate these variable regions into expression vectors containing human constant regions.
Transfect into non-antibody-secreting myeloma cells.
Result: Cells secrete a chimeric antibody—mouse variable regions + human constant regions (~34% murine).
Chimeric antibodies reduce immunogenicity while preserving antigen specificity.
How are ‘human‘ antibodies obtained from mice?
Delete mouse IgH, IgK genetic loci in germline and replace with corresponding human sequences.
Immunise animal with antigen
Mice produce an α-IL12 (polyclonal antibody) with ‘human’ sequence.
How are fully human antibodies obtained from genetically engineered mice?
Genetic engineering: Mouse immunoglobulin heavy (IgH) and light (Igκ/Igλ) chain loci are knocked out and replaced with human immunoglobulin gene loci in the mouse germline.
Immunisation: Transgenic mice are immunised with the target antigen (e.g., IL-12).
Response: The mice mount a normal immune response, producing human-sequence antibodies (e.g., anti-IL-12).
Isolation: B cells are harvested, and hybridomas or recombinant techniques are used to isolate and clone fully human monoclonal antibodies.
These mice (e.g., XenoMouse) produce functional human antibodies, ideal for therapeutic use with low immunogenicity.
How are human mAbs obtained at a large scale?
Discovery & Cloning:
Isolate DNA encoding human antibody heavy and light chains (from hybridomas or B cells).
Insert into expression vectors and transfect host cells (e.g., CHO cells) via electroporation.
Screen and select high-producing clones.
Cell Line Development:
Expand selected clone to generate a stable, high-yield production cell line.
Large-Scale Production:
Grow cells in bioreactors under controlled conditions.
Secrete monoclonal antibodies into culture medium.
Purification & Formulation:
Harvest supernatant and purify antibodies using techniques like Protein A affinity chromatography.
Formulate for therapeutic use.
What is phage display, and what is it used for?
Phage display is a technique used to identify and select high-affinity antibody fragments against a target antigen.
DNA encoding antibody V domains is inserted into a bacteriophage coat protein gene.
Phages express and display the antibody fragment on their surface.
The phage library is screened against a target antigen (biopanning).
High-affinity binders are retained; low-affinity ones are washed away.
Selected V domains are sequenced and cloned into full-length antibody constructs for production.
Enables rapid, in vitro selection of antibody fragments without immunising animals. domain into the gene encoding the virus coat protein.
What are the different types of therapeutic Ab?
Murine -omab
Chimeric -ximab
Humanised -zumab
Human -umab
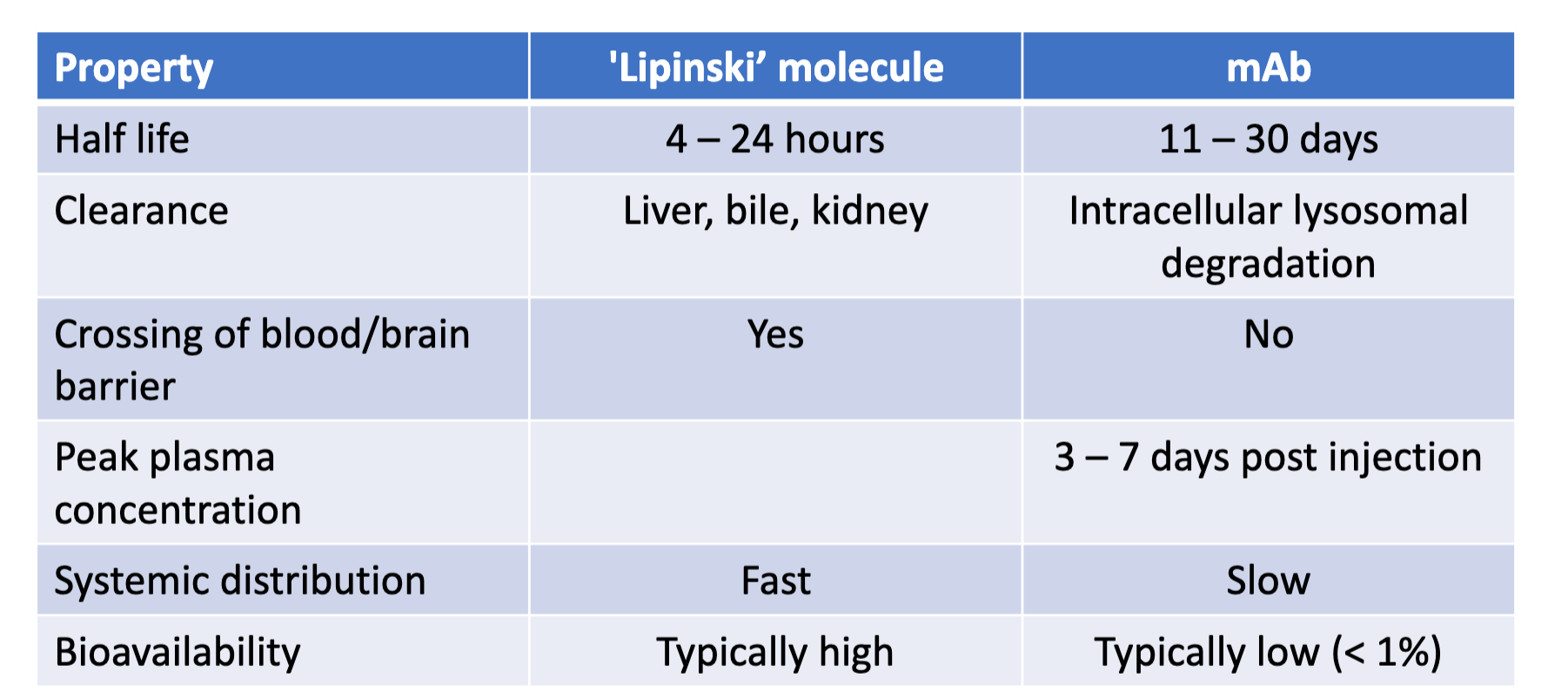
Compare the pharmacology of mAbs to ‘Lipinski’ molecules.
What are the main disease areas using therapeutic Abs?
Cancer
Autoimmune conditions
How is Rituximab used in the treatment of Non-Hodgkin’s lymphoma?
Non-Hodgkin Lymphoma is the cancer of B-cells
Binds to CD20 protein, found on normal and cancerous B-cells in vitro.
Recruits components of the body’s immune system to destroy the B-cells.
Once Rixutan has cleared the body, normal B-cels can begin to grow again.
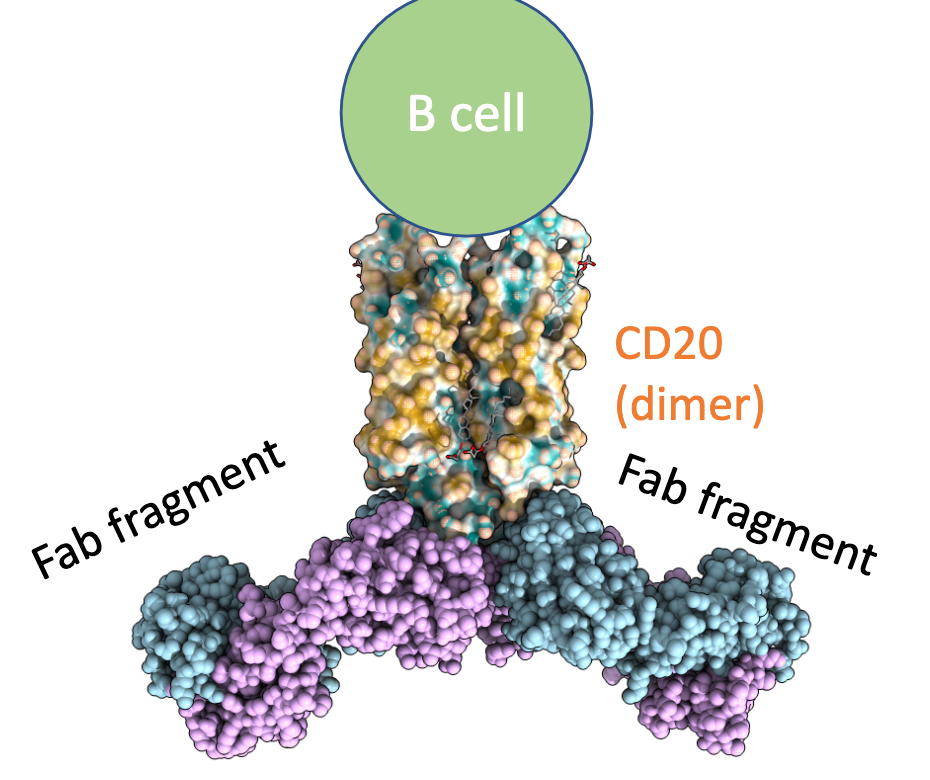
Describe the characteristics of Rituxan (rituximab).
Chimeric antibody, typically used in combination with chemotherapy.
Target: CD20 surface antigen on B cells.
Disease areas: B-cell malignancies, Non-Hodgkin lymphoma, rheumatoid arthritis.
Resistance mechanism: Complement inhibition through CD55, CD59 (Fc receptor-mediated cytotoxicity).
What are mAb-induced effector mechanisms?
Direct cell death
Complement-dependent cytotoxicity
Ab-dependent cellular cytotoxicity
Ab-dependent cellular phagocytosis
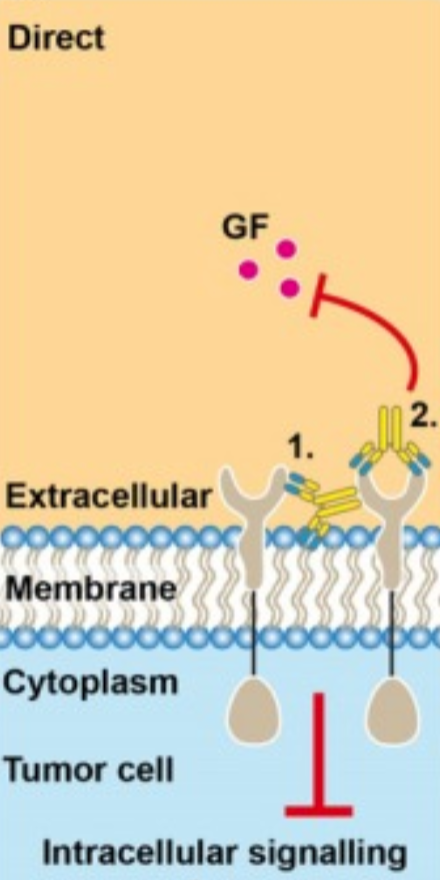
What is direct cell death in monoclonal antibody therapy?
The mAb acts as a direct inducer of programmed cell death in the target cell.
Some mAbs trigger apoptosis or growth arrest directly by binding to their target antigen: receptor cross-linking, signal transduction inhibition, disruption of survival pathways
Does not require immune cell recruitment or complement activation.
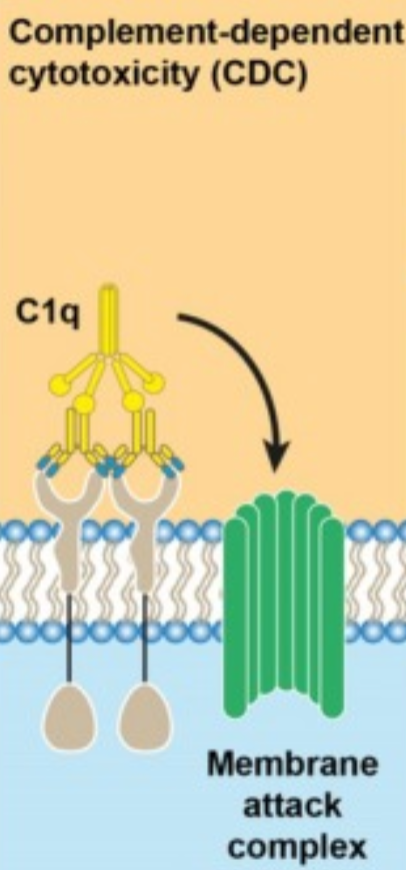
What is complement-dependent cytotoxicity (CDC) in mAb therapy?
CDC is most efficient with IgG1 and IgG3 subclasses due to strong complement activation.
mAb binds to cell-surface antigen.
Fc region of the bound mAb recruits C1q, initiating the classical complement cascade.
Formation of the membrane attack complex (MAC) creates pores in the cell membrane.
Leads to cell lysis.
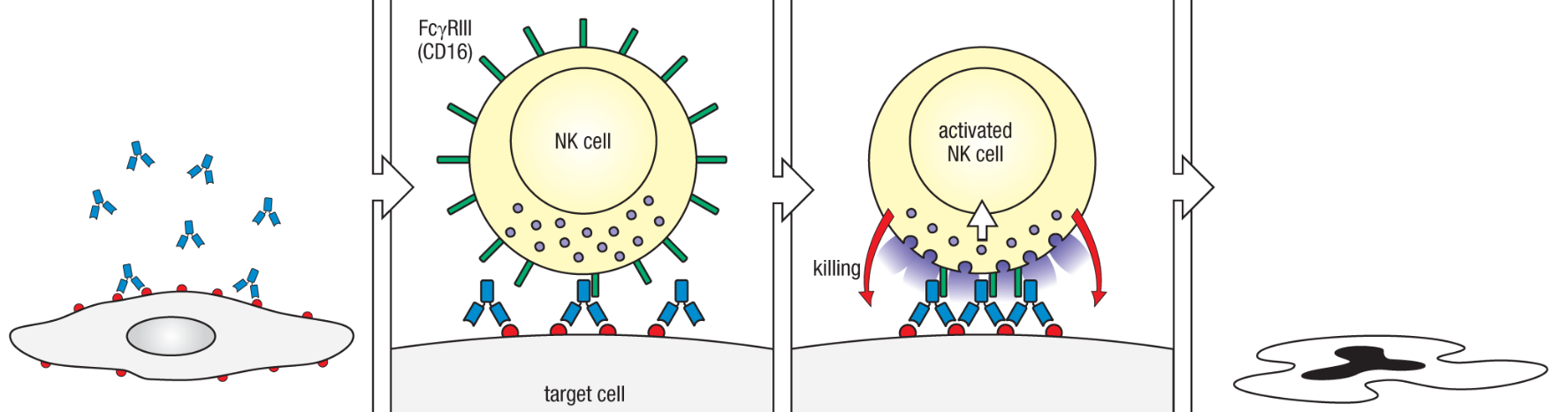
What is antibody-dependent cellular cytotoxicity (ADCC) in monoclonal antibody therapy?
ADCC bridges innate and adaptive immunity, making it a major mechanism for therapeutic mAbs in cancer and infection.
Therapeutic mAb binds to antigen on target (e.g., tumor or infected) cell surface.
NK cells recognize the Fc region of the mAb via FcγRIII (CD16) receptor, specific for IgG1 and IgG3 subclasses.
NK cell is activated and releases cytotoxic granules containing perforin and granzymes.
Target cell undergoes lysis and apoptosis.
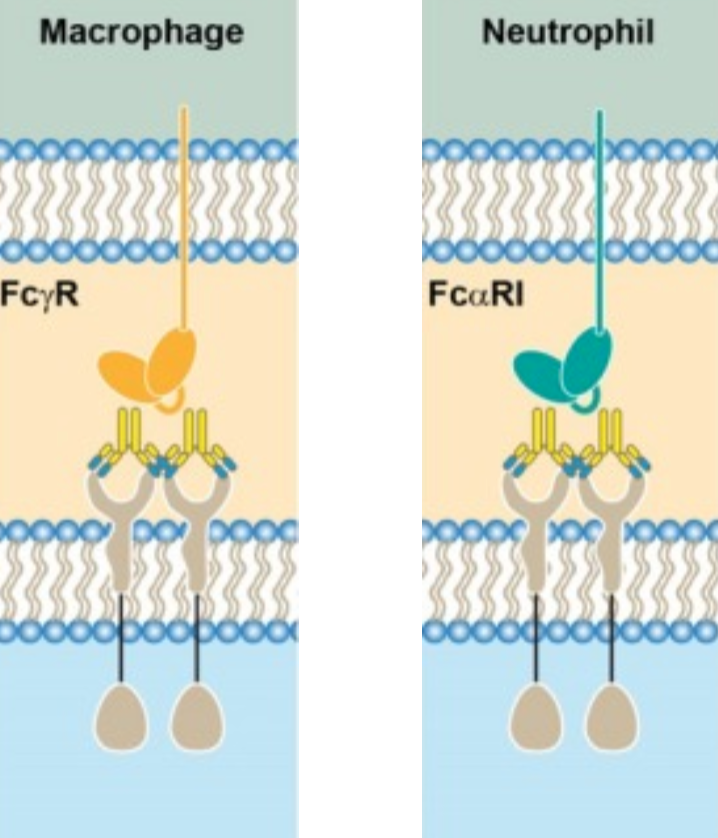
What is antibody-dependent cellular phagocytosis (ADCP)?
ADCP is a key clearance mechanism, especially in cancer immunotherapy.
mAb binds to antigen on target cell.
Fc region engages Fcγ receptors on phagocytes (e.g., macrophages, neutrophils).
Phagocyte internalises the opsonised target cell.
Target is degraded in phagolysosomes.
By looking at the receptor name, how can you determine their affinity?
RI: ‘high affinity’
RII: low/med affinity
RIII: ‘low’ affinity (Kd > 10-6 M)
What does a Fcγ receptor bind?
IgG antibodies
What does a Fcε receptor bind?
IgE antibodies
What does a Fcα receptor bind?
IgA antibodies
Describe the characteristics of Herceptin (trastuzumab).
Humanised antibody, IgG1κ.
Target: EGFR2 (HER2)
Disease areas: HER2 positive breast cancer
Resistance mechanism: Low or no expression of HER2 in tumour cells; activation of alternative signalling pathways
Effects: Cell cycle arrest (Direct Cell Death); ADCC
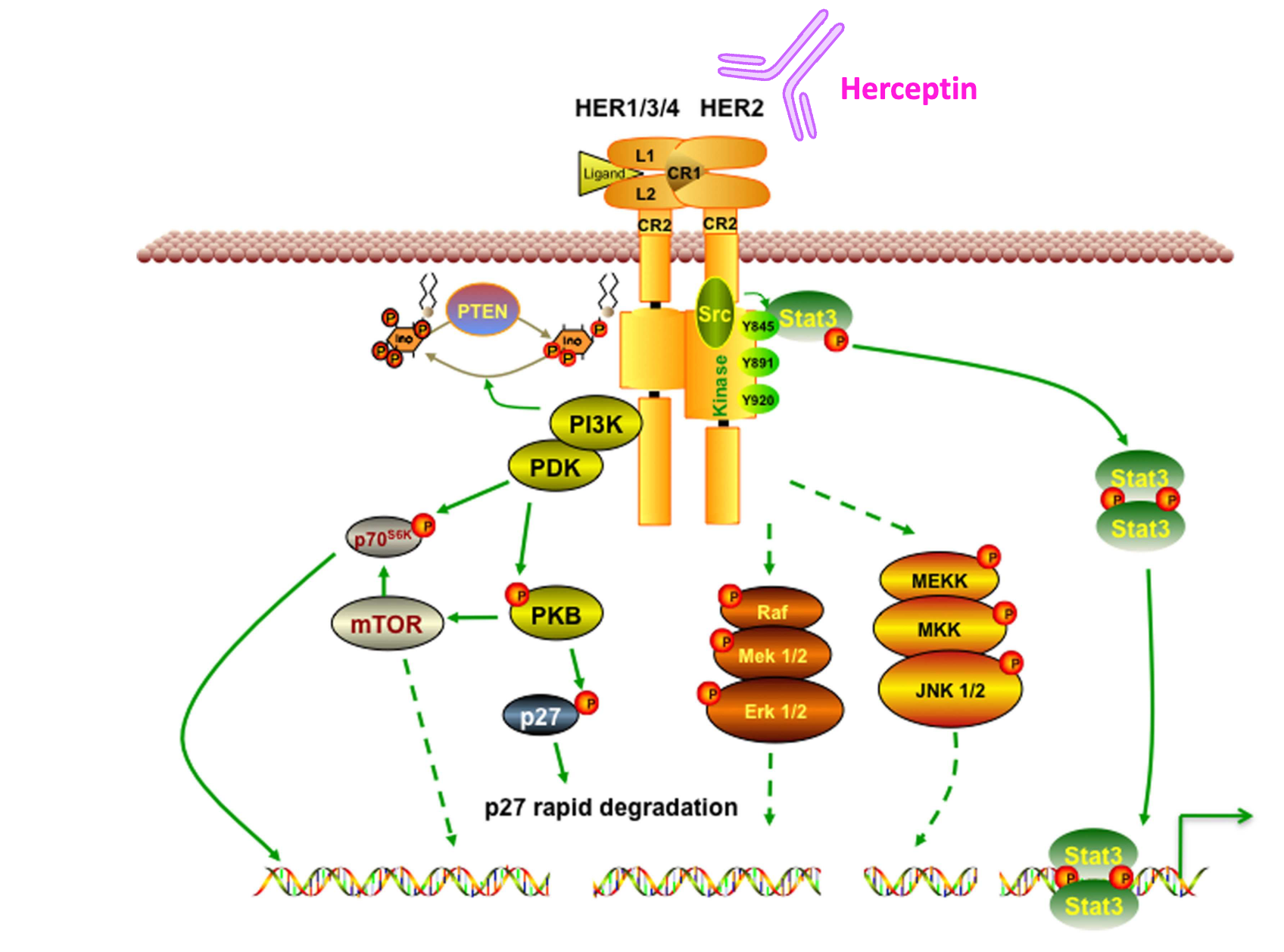
How is Trastuzumab used in the treatment of HER2 positive breast cancer?
Tumour cell proliferation depends on HER2 signalling - blocking HER2 suppresses tumour cell proliferation (direct cell death).
If HER2 is not expressed in tumour cells (HER2-/-), it is useless thus will need personalised medicine therapeutics for treatment.
How is Adalimumab used in the treatment of immune conditions?
IgG1, humanised antibody
Target: TNFα
Mode of action: inhibits TNFα binding to TNF receptors p55, p75; reduces TNFα-induced inflammation
How are mAbs used to treat autoimmune disease?
How do rituximab and obinutuzumab bind to CD20?
Differ in stochiometry
Dimeric RTX binds CD20
Monomeric OTI binds CD20
This illustrates that differing Ab designs to target the same cell surface receptor elicit different effector mechanisms.
What is RTX’s mode of action?
Inter-tetrameric binding to CD20.
Antibody-dependent cellular cytotoxicity
Complement cascade activation → membrane attack complex formation, cell lysis
Very low direct cell death
What is OTI’s mode of action?
Binds CD20, but the de-fucosylated glycosyl group in the Fc region changes killing mechanisms.
Antibody-dependent cellular cytotoxicity, but binds a broader spectrum of Fcγ receptors
More direct cell death
Antibody-dependent phagocytosis
No complement activation
What is CD20?
Transmembrane protein of the tetraspanin family (Tspan4a)
Highly expressed in B cells ( and B cell-derived malignant cells), expression is lost when B cells differentiate into antibody-secreting plasma cells
Function: Ca2+ regulation upon antigen binding to BCR; no physiological ligand
What are the two types of mAbs that target CD20?
Type I: Causes clustering of CD20 in lipid rafts in the membrane, e.g. RTX
Type II: No compartmentalisation in lipid rafts, e.g. OTI
B cell population recovers after anti-CD20 mAbs are withdrawn