Organ Transplant Exam #3 notes
1/86
Earn XP
Description and Tags
Pray for the best and hope you get ovr a 95
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
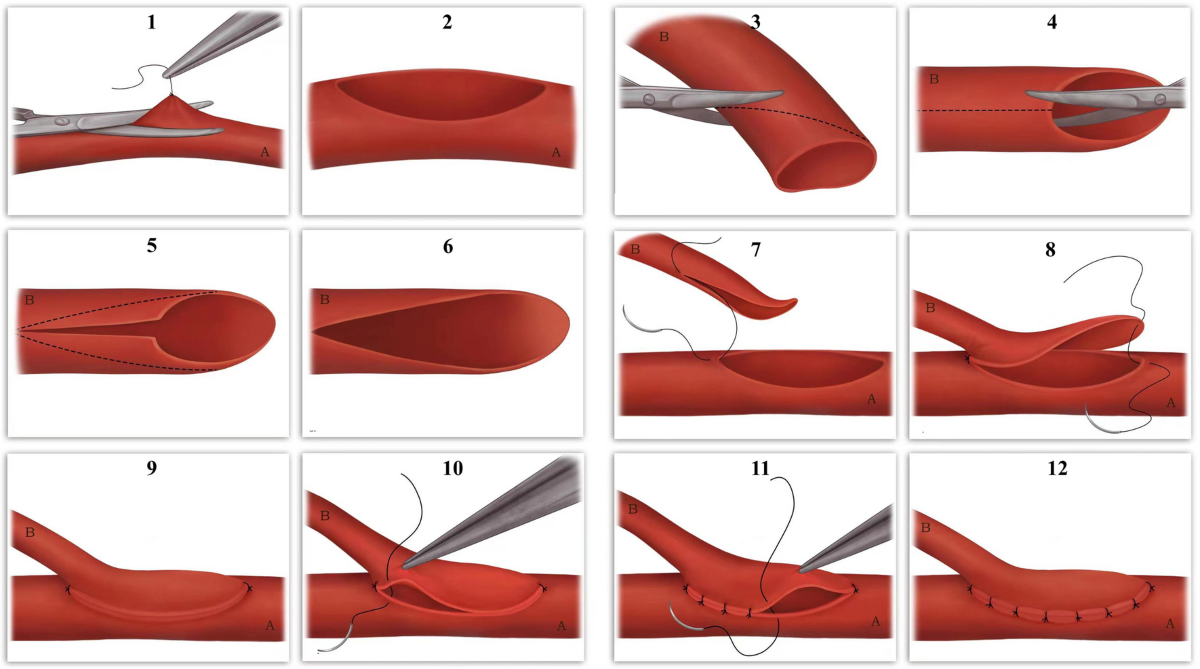
Q? What are anastomosis and what are some of the challenges that come with it?
anastomosis: Surgical procedure for connecting two tubes (blood vessles)
Challengs: Preventing leakage, infection, aneurysm, fibrosis, narrowing, and clotting
at the time when they started no antigoagulatns were avaliable
what methods are used: finer silk and finer needles
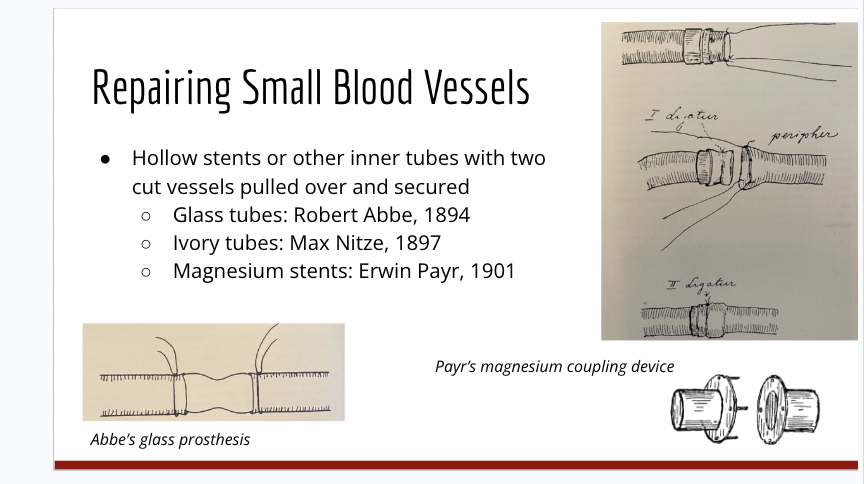
What are some of the ways small blood vessels are repaired. What are teh different types of mechaniss and know what they look like
Hollow stents or other inner tubes with 2 cut vessels pulled over and secured
used glass tubes
ivory tubes
and magnesium stents (couling device
In what ways are large blood vessels. What are the advatntages , what are lateral anatomosis method
generally more successful than reparing small vessels
uss lateral anastomosis
over and over silk stretches to join two larege abdominal vessels
advantages
more successful than repairing small vessels
Large blood vessels repair methods: invagnation? what is it, what makes it useful.
invagination technique: placing part of one vessel inside of another
femoral artery damaged by a gunshot wound

Who is alexis carel and what is the triangulation method of reparing large bloodvessles
alexis carrel
considred father of modern vascular and transplantation surgery
used better needles with much finer sutoring material
Triangulation
hold vessel open
make three sutures and pull to create 3 flat lines = 3 separate sutures
do not penetrate the wall

What are some of Alexis carrels contributions to transplantation history. What is actual life and latent life and how does it play into transplantation? What developments can be attributed to him
Standardizd the use of strict aseptic conditions of surgery
used fine straight silk
explored methods of prolonging tissue survival outside the body
“Actual life” - culture at normal body temperature allows for normal cellular functions
“latent life” - culture at colder temperatures = less risk of infection and reduced metabolisms
Demonstrates the organs die at different rates
colder temperatures
he standardized the use of strict aseptic conditions of surgery
used fine straight silk
flusing with saline regulary
lubricaiton of threads
full penetration of vascular wall
and holding of smal vessels with clamps
Developments
the triangulation methods
the carrel patch
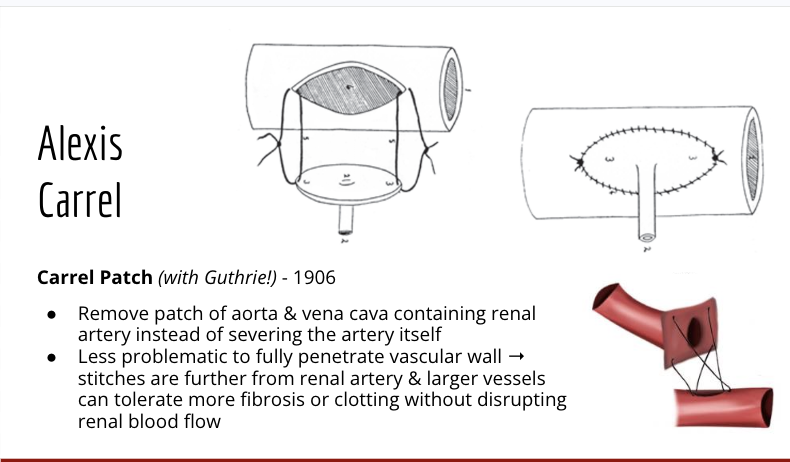
What is the carrel patch and what does it do? what makes it so effective
methods
removal of a patch of the aorta and vena cava containing renal arteries
used to prevent severying of the full artery
it is less problamatic to fully penetrate the vascular wall
stiches are futher from the renal artery and larger vessels = can toelrate more fibrosis or clotting without disrupting renal blood flow
What are some of Alexis Carre’s faliures and what did it result in(hint hint pumping)
he attempted to ake perfusion pumps but it often resulted in infection
helped developed a closed sterile pumping system
Why were kidneys the first major solid organ transplatnt choice
they were easy to access and least challenging surgically
patients did not depend on only on
live donors
chronic diseases were common
What are the major advantages of Corneal (the eyes) grafts and what do they do
Corneal grafts
no blood circulation = no lymphocite infiltration or graph rejeection
since it is avascular, no major blood vessles to the cornea
allows for suffiecient donor tissue
major resistance by the public bc corneal grafts come from passed people but it became routine early on
What are some of the early attepts at immunosuppression
Radiation
total body radiation reduced antibody responses
radiation prolonged life of rat tumonr grafts = reduced innate and lymphocite infiltraiton
Benzol
benzene used as an early immuno s uppresiont but stopped bc it was dangerous
exposure lower blood coutns and damaged bone marrow
affects white cells (suppressed antibody function)
casued lukemia
suppressed antibody formation
Nitorgen mustard
used during the ward, used to depress antibody formation
used as a poision in world war 1
resuts in low white blood cell counts, significant bone marrow changes, very weak immune system, reduced antibody formation
What is histocompadibility and how does it play into organt ransplation
histocompatability = siilarity fo antigens between donor and recipient cells
ex. tumor from strain a mouse bearing antigen 2 would thrive in other mise also carying antigen 2 but is rejected by th rest
What casued the reuptake of transplantation in the us
war (again), burns, surgery ect
expecially areial combat they suffered suvere burns
What is and what is the significance of the second set experiment
female pateint given a skin pinch from her brother
second set of skin pinch was applied two weeks later
both skin grafts were rejected at the sae time but the second set was rejected faster = example of adaptive immune response streingth
allowed for the assumption that immue responses were antibody diriven
reduced the idea of homografts and started a new field in tissue rejection
What are some of peter medawar’s contributions to cell rejection methods? what is the freemartin experiments
What did peter medawar discover
all tissue could immunize against future skin grafts
responsible antigens present in most cell sof the body
supported that lymphmocites are required for graft rejection
Fremartin experiments (used twin cows lol)
realized all tisssues could immunize against future skin grafts —> responsible antiges are present in most cells of the body
results = all twins accepted skin grafts from eachohter = shoed immunoligcal tolerance can be induced
later experiments showed that you can induce immunoligal tolerance to an allogenic graft
what is mosaicism and does it relate to graft rejection
it was found that shared blood types = suggested that fetuses have an abiltity to recognize all tissue presnt during prenatle development as self
he used blood. type expeiiments for early induction efforts = induction of immunological toerance to an allogenic graft
What is chimerism and what is adult to;erance
Typically when you insert biological dna or smt from one thing to another
ex donor bone marrow from different people to incorperate into a recipient body
Adult tolerance: only skin grafts from strain of donor marrow could surivive (when donor bone marrow exists)
a state where the recipient’s immune system accepts a donor organ without requiring lifelong immunosuppression, while maintaining the ability to fight infections.

Describe some of the early methos of histocobalitbility testing
normal lymphocyrte transfer set
irridated hampster test
third man test
normal lymphocyrte transfer set
inject lymphocytes from donor into skin of recipent and monitor for minor hos vs graft response
irridated hampster test
injectino of lymphocites and apcs from one bone doro and recipent into the skin of irridated hampsters and cehkf for an immune response
third man test (dangerou)
graft heathly vounteer with recipient skin graft, next you take recipient family skin (donors) and graft it to the same volunteer, see if there is a second set style accelerated recetion = indication of similar antigens between the recipient and donor.
Tissue (usually skin) from Donor A is grafted onto a Third Person (Person C).
Later, tissue from Recipient B is also grafted onto Person C.
If the second graft is rejected very quickly, it suggests Donor A and Recipient B share similar histocompatibility antigens (HLA).
risky bc could make immune repsonse stronger or srpead disease
sensitizes the imune sytem = exposure to frogien HLA antigens
causes the production of memory t cells and antibodies against those antigens
increase risk of future graft rejection
accelerated hyperacute rejeciton
unnecesary imune risk
the third individual does not ned a transplantation so they are just at risk for nothing
What are some of the disadvantages of early appraoches to histocompatability
Skin graft can sensitize patients to kidney transplant & accelerate rejection
Potential to transfer disease (increasing transmission of hepatitis B in dialysis units at this time…)
Tests were too slow relative to urgency of transplantation
Couldn’t scale to large number of patients
What is the plastic revolution and how does it play into translant history
cell isolation, cell culture, and ability to stuyd cells in vitro = allowed for this
allowed ofor the developent of techniques for motering cell killing and mgiration throughtout the body
alllowed for the ability to frieeze, store , and revive cells
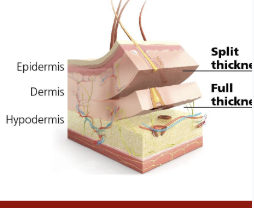
What are the split thickness skin grafts and what were the nature of the first two skin grafts
developed by james barrett brown
it is a skin graft invoving the epidermis and portion of the dermous
thicker than previous grafts only including the pideris
first successfull skin graft between identical twins (syngenic)
What is te bonnoer vs oran place and how did it pursh for mrore patoient rights and protections around surgery
boy was convinced to undergo procedure without his mothers concent and surgeyr ws a failerur
this estaglished legal precedences that infromed rconcent was required for surgyer and parental consent is required for childre
Damages for assault and battery
Boy was convinced to undergo procedure by his and his cousin’s aunt
Boy’s mother was home ill and never consulted → established legal precedent that informed consent is required for surgery & parental consent is required in the case of children
Outline the main accomplishments of the Boston Group and identify one or more unethical practices they engaged in.
team of 5 surgeions who undergo extensive underground transplant research at brigham and women’s hospital
Achievements
sucessfully transplanted allogenic skin grafts to ww1 piolets
First attepts of immunesppressive methods using
ACTH, cortisone, and testosterone
used blood type compatability too
early immunosuppresive irridation and radiaition methods used
breakthroughts in early experimental kidney transplantation in humans
demonstrated temporary kideny graft was possible
developed the used of pre-treatment dialisis to keep patients alive
first successful kideny transplant between idientical twins = transplantation when imune rejection was avoided
helped advanve bone marrow/hemapoetic stem cell transplantation and early immune tolearnce induction (human chimerism)
Unethical practices
did experimental transplatation in secret without proper oersight or supervisors
delayed reporting negative results due to fear of criticis
used organs from cadaves or vulneraable children = no infomed consent
high risk experimental procedures with many casulties
they lacekd transparencey, oversigth, and ehtical safeguards
What allowed for the first succesfful kidney allograft
fraternal twin brothers
recived toatl body irridation but no other imunosuppresison
allowed for full recovery = more reserch into immunosuppressors
What do you nee dto know for allografts to succed, how do twins relate to transplants
surgical techniques were succesful but imune rejection was a hard hurdle to pass
transplants by twins were generaly the only succsffull ones without immuno suppression
you need ot know the follwoign things to considre imuno suppresssion
mechanims of rejction
how to match patients and donors
and effective imuno suppression efforts
Tolerance mechanisms: Central tolerance vs peripheral mechanis. What is invovled? what are th
Central tolerance: negative selection “Clonal deletino” of self reactive lymphocites in central lyphoid organs
T cells: thalamus
B cels: bone marrow
Peripheral toelrance; Various mechanism for deleting or inacivating self-reactive lmphocites outside of central lymphoid organs
How do you break Central Tolerance mechanisms?\
T cell negative selection is a sepctru = not all alutorective t cells are deleted
developing B cells do not se all self antigesn (bc in the bone marow you are not exposed to evrything)
not all self reactive B cells are deleted
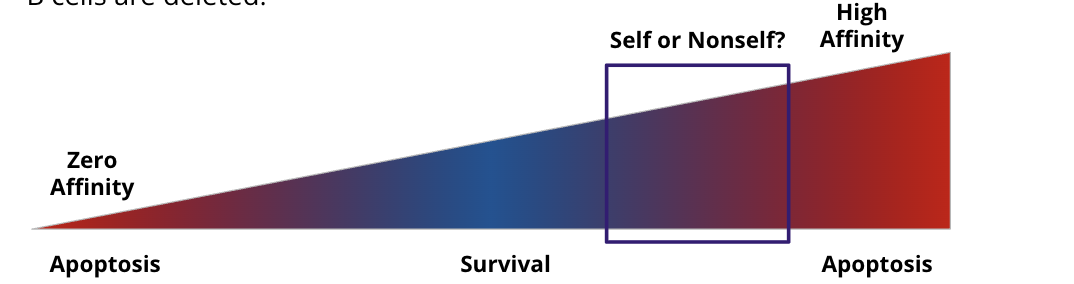
What are some of the tolerance mechanisms? how are t cells used (special type)? What mechanims and signals are needed? What are the results if this happens or doesnt happen
T cells
self reactive T cells become regulatory t cells in developent
iTregs = t cells specifc to self or healthy antigens differentiate into regulatory t cells thorughout the lifespan
Tregs: bystandur suppresion, infections tolerance, and IL-2 consumption
Ntregs = natural t regs
develop in the thalamus
Itregs = indcued t regs
derrived from native CD4+tcells in the periphery
Three signals are need for tolerance
antigen
costimulation
without this = anergy = inactivation
cytokines
innate immue system determines self/nonself and healthy and dangerous cells
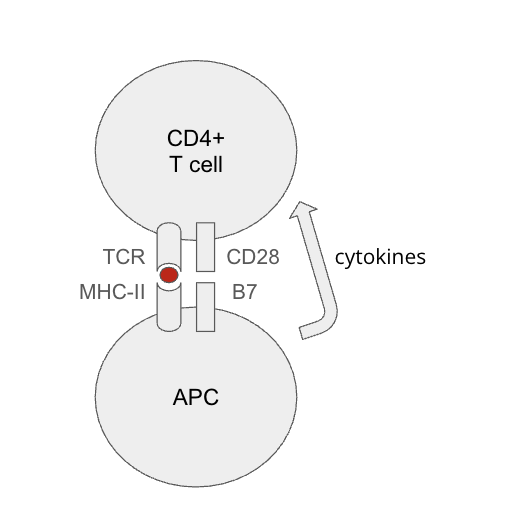
What are tthe checkpoint molecuels for tolerance mechanisms
Activated T cells express inhibitory molecules
CTLA-4: agonist of B7 molecules (blocks CD28 signalling)
PD-1: binds to PD-L1 and PD-L2
Pd stands for program death ligand
two distinct ligands that bind to PD-1, transmitting a signal that inhibits T-cell activity, allowing tumor cells to evade immune destruction
good for maintaining immune homeostasis by preventing autoimmunity, but bad in the context of cancer and chronic infections
PD-L1: expressed by wide variety of cells
used by tumor cells to inhibit T cell responses
PD-L2: expressed by antigen presenting cells during inflammation
Involved in immune reg
Negative feedback to prevent overactivation of T cells
Exploited by pathogens & cancer cells → checkpoint inhibitors are a common and effective cancer treatment!
Stimulate expression to prevent organ transplant rejection?
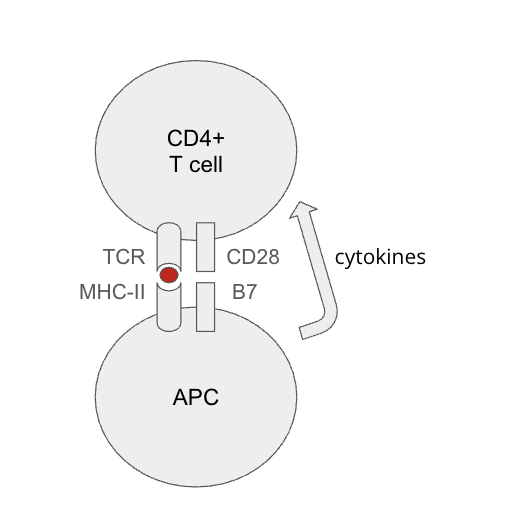
What are other ways tolerance is broken? molecular mimicaray? antigen presentation? Damps
Molecular Mimicry (cross reactivity): dangerous antigen looks like self antigen
Antigen presentation of healthy self and dangerous non-self (viral envelope) → response against healthy self
DAMPs can come from sterile tissue damage → activate innate response against self
What are the 3 stages of graft rejection
Hyperacute Rejection (Minutes to Hours)
Acute Rejection (Weeks to Months)
Chronic Rejection (Months to Years)
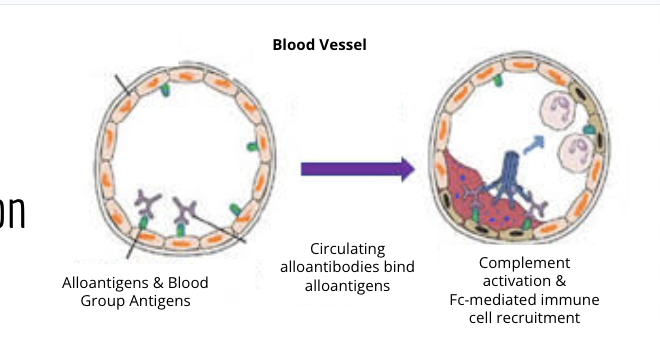
Describe hyper accute rejeciton and use the blood vesssel as an example. What happens? what are the mechanims involved
occurs when blood does not match the host body
allontigens and blood group antigens cause mismatch = rejection by natural antibodies (MHC)
antibodies bind to the endotheilum and triegger complent, clotting cascade and immune cell recruitment
causes plaque build up and artherosculrosis in the blood vessle
mostly problematic folliwng other blood trasfusions, transplants, or xenografs
How are damps related to accute graft rejection? what happens? what is the overall mechanism
during surgery blood and lymphatic vessels are severed = very traumatic
tissue is breifly starved of oxyggen = decrease in ph
sudden reprofusion = additionalinjry
results in the relase of DAMPs = prr activation = inate activation = adaptive immune response to damage
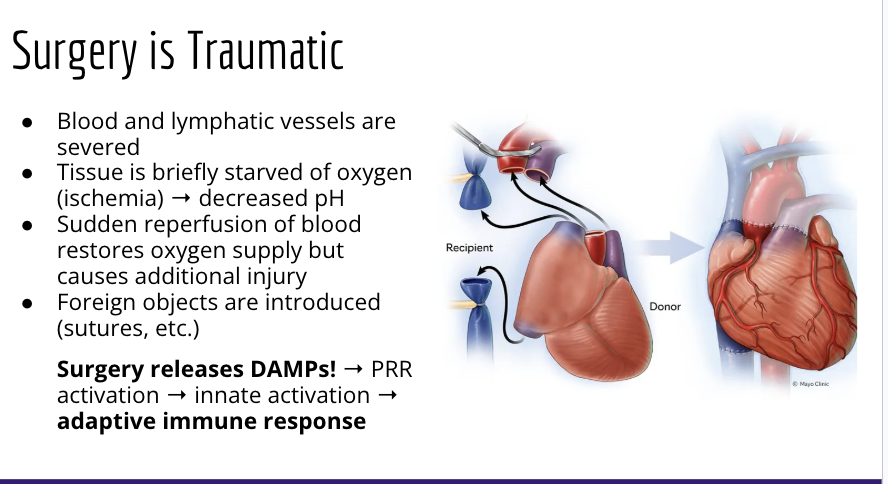
What is the human leukocyte antigen? what is the major histocompatability complex? what dos it incod? whats the difference between MHCS
Major histocompatibility complex (MHC): that thing that presents antigens!
“H2 region” in mice encodes MHC-I and MHC-II
Human leukocyte antigen (HLA): the human MHC
most polymporphic genes of human genome
HLAs are the human MHCS
MHCs
1 has 3 alphas 1 beta
HLA a-c
2 has one half beta and one half alpha
HLA-DR, DQ, and DP
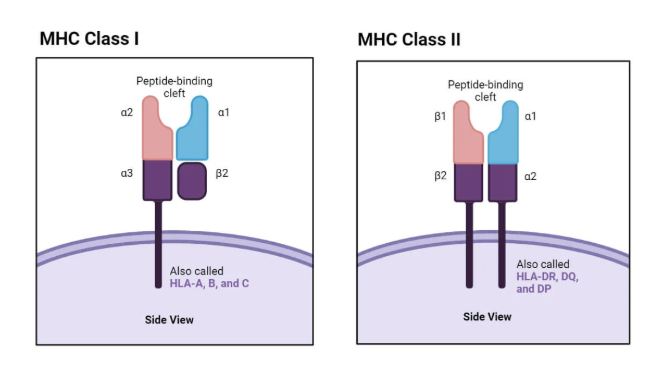
What are the different types of allorecognition? direct, indirect, and semi direct pathways. what does it involv, what are the receptors? what are they responsibl for
Direct pathay
Donor APcs connect and prosent dornor antigens to host t cell
uses hla/peptide complex
results in activation of large polyclonal T cell response
responsible for acute rejection
indirect =
recipient apc presents donor antigen using its own presention receptor to recipent t cell
Recipient apcs present donor antiges (both MHC and minor self antigens) on self to t cells = responsible for chronic graft rejection
semidrect pathway
recipent apc obtains donor MHCs from plasma membrane trasfer
Semi-direct allorecognition relates to acute rejection because recipient antigen-presenting cells (APCs) acquire and display intact donor MHC molecules, activating T cells that damage the graft.
they present forgien MHC: antigen to T cells = assive polyclonal response
robust, rapid, polyclonal response
responsible for acute rejection
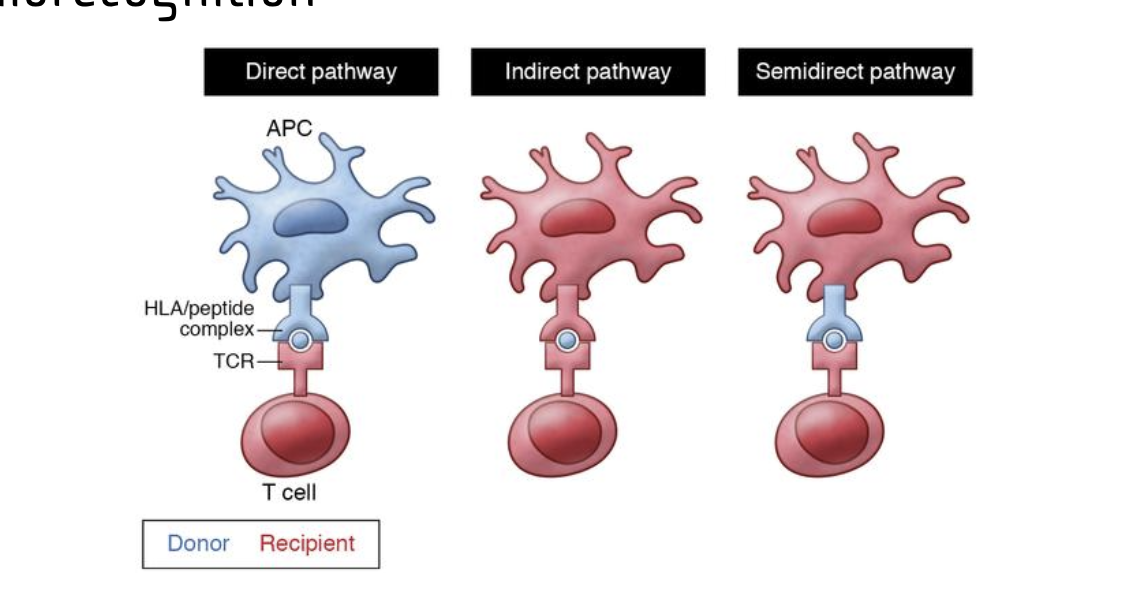
What is the mechagnism of direct allorecogition? how does it relate to cross reactivity
TCRS are specific to MHC: antigen complex not just the antigen
self and nonself mhcs are pretty similar but mosty differ in antigen binding rations
Cross reactivity:
the same TCR can reconize different antigesn on different MHCs (ie slf or donor mhc)
Lots of foreign MHCs & minor alloantigens = lots of distinct MHC:antigen complexes
ex: TCR can bind to the antigen presenting MHCs of both self and donor apcs for recogntion
1-10% of T cells in an individual can directly engage intact foreign MHC
Highly diverse polyclonal adaptive immune response is immediately activated
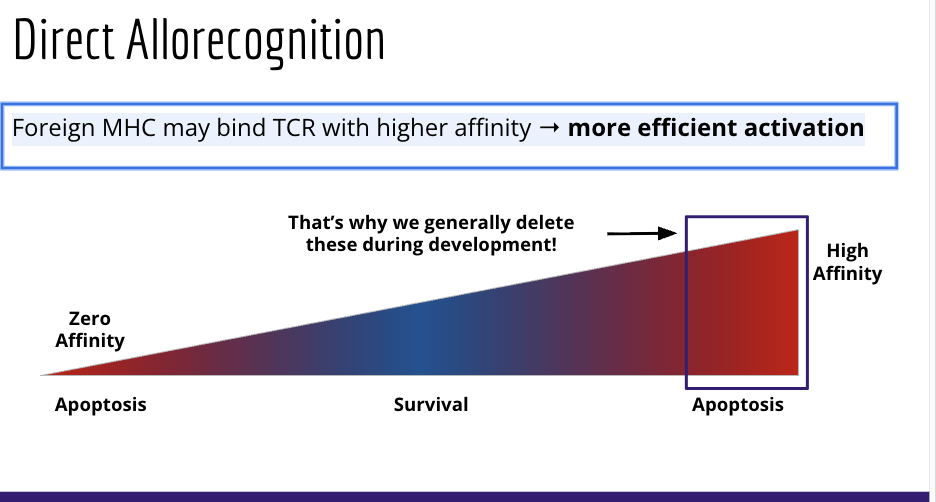
What is the binding affinity of direct allorecognition
Foreign MHC may bind TCR with higher affinity → more efficient activation
What is cross matching, ABO incompatable transplants, and Xenografing in relation to hyperacute graft rejection? What mehtods are used to help with ABO inompatable transplants
Cross-matching: test whether donor & recipient have antibodies reactive to the other
ABO Incompatible Transplants: now possible with preconditioning
Plasmapheresis: Remove plasma (contains antibodies) from blood
Immunosuppressive drugs
Especially possible in livers, which seem less sensitive to hyperacute rejection (for some unknown reason)
Major problem for xenografting
Host cells express complement protein competitors & proteases to protect themselves
Complement-regulatory proteins work less effectively across species barriers
Current attempts to genetically modify pigs to be less susceptible to hyperacute rejection from human recipients!
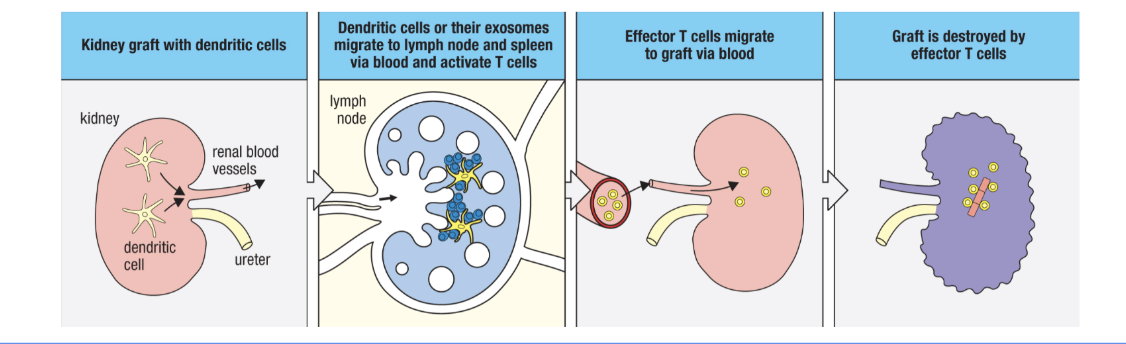
What is an example of direct and semidrect allorecognition in the kidnesy? how does this activate iune repsonses? how does this play out longtrm? what are passenger leukocytes
passenter leulocites = donor dendritic cells that travel to lymph
passenger leuokocytes eventual die off = lower direct and smidrect allorecognition risk and acute rejection risk (direct)
some long lived tissue residnt cells (tr macrophages, innate lymphoid cells, memory cells, ect) can still persists (Semidirect)
What is chronic graf rejection? how doesi schemia reperfusion injuries relae to this
chornic graft rejection
specific immune alloreactivity and/or non-immune injuies
viral infecitosn resulting from immunosuppression can contribute to this (similar to autoimmunity)
recurrence of teh same disese that destroyed the og organs - note there are different responses in different organs
Ischemia reperfusion injuries; provid dampsat tie or transplant to start innate repsonses —> Adaptive immune response) induces crhronic response
What is chronic allograft vasculopothy? donor specific antibodies (DSA’s) and how does it relate to chronic graft rejectionq
Chronic allograft vasculopathy: chronic arteriosclerosis of graft vasculature resulting in reduced blood flow, fibrosis, and atrophy
major component of chronic graft rejection, especially in the kidney and heart transplant
develops DSA’s = aloreactive t cell to graft vascular endothelium (alloreacive = reacting to forgein bodies)
endotheilial injury = immune infiltraiton into allograft
some immunosupresive drugs cause vascular injury
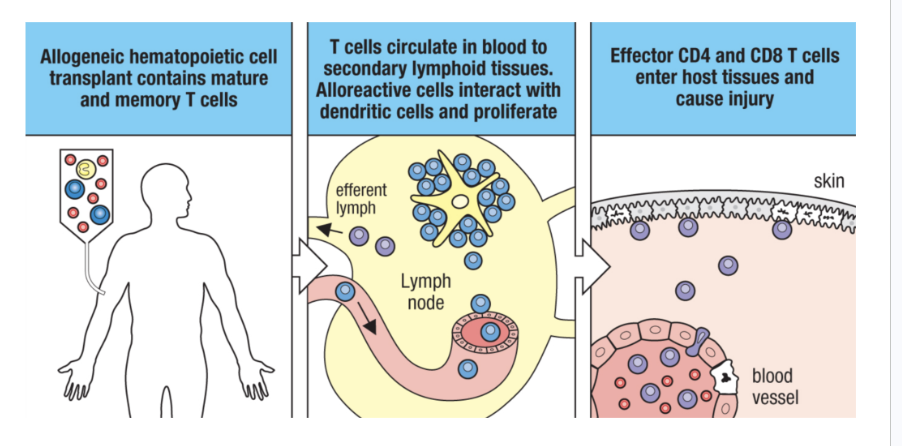
What is graft vs host disease? what is the mechanism? how do you resolve this
Bone marrow transplant is generally used to treat leukemias and lymphomas
rejection of recipient tissues by donor immune cells followign bone marrow
1impacts the skin, intestines, and liver (alrady damaged by chemotherapy and raidation)
thats why attempts at inducing adult tolerance didnt go well
mechanism
1.. allogenic hmatopoetic cell transplant contains mature and memory t cells
t cells circulate in blood to secondary lymphoid tissues = alloreactie cells interact with dendritic cells and proliferate
effector cd4 and cd8 t cells enter host tissues and cause injuy
What are the different types of T cells 8 and 4. what are their differentations
CD8+ Cytotoxic: mediate cell killing during rejection
May promote Th1 response
Th1 CD4+: generally promote rejection
Increases CD8+ function, activates macrophages, recruits T cells
Th2 CD4+: generally protective
Antagonizes Th1 responses, stimulates wound healing
Th17 CD4+: generally promote rejection
Antagonizes Treg responses, recruit neutrophils
inflammaiton, attachsk mucosal sites
Tregs: critical for establishing graft tolerance
What are b cells and what type of graft rejection are they typically responsible for? what do they produce? what can be done to help promote tolerance
they produce DSA’s -Donor specific antiboidies = prootes graft rejection
genearlly hyepracute and chronic
they support t cell differentation and memory formation
indirect antigen presentation = indirect allorecogntion
accommodation: physiological changes that allow regeneratino of tissue function after grafting
30 persent of pateitns generate DSAs without other signs of rejection
ex class switching from IgG3 to IgG2 for less efficeint complement activation
subset of regulatory B cells (bregs) help promote tolerance
What are neutrophils and how do they realte to graft rejection? what do they do what activates the
= damps (ischmia-reperfusion injury) relase = tissue resident myeloid cell actiaiton = recruitent of neutorphils
generally pro-inflammatory (promotes rejection)
inflammatory cytokien production skews APCs to induce Th1 and Th17 t cell phenotypes
th 17 recute neutrophils chronically (positive feeback loop)
may provide costiulatory signals to T cells in graft
one neutrophil su bset may be later involvd in revascularization
Phagocytes. What are they and how to they relate to rates of graft rejection. What is the mechanism behind that? what signaling molecule is beign used
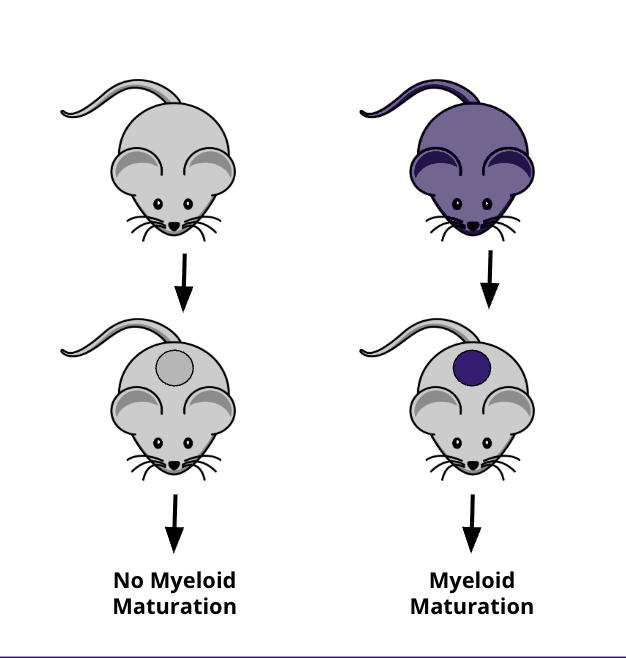
Damps may not be sufficeint for activating the innate immune system during graft rejection
allogenic, but not syngeneic grafts in mice induce monocyte maturation or innate cell activation
innate cells must be recognizing non self rn (how? phagocytosis eat me eat me)
CD47 = dont eat me signal = signals that prevents phagocytosis of healthy self cells
Sirpa on phagocytes bind to CD47 on other ells = inhibits phagocytosis x
one is missing = its eaten
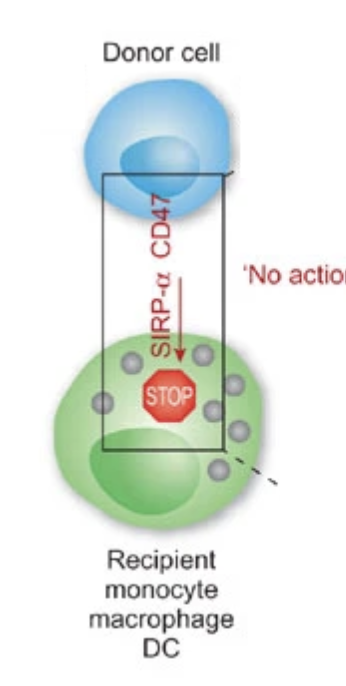
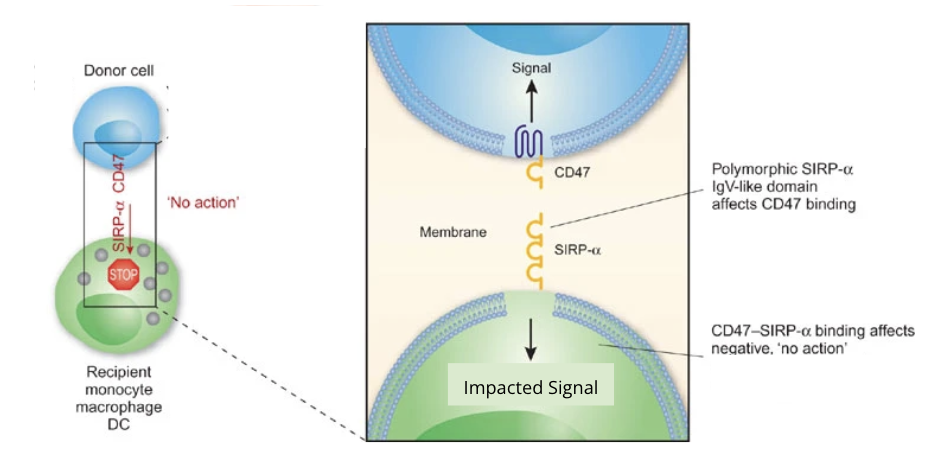
What is the function of cd47 and sirpa? how do differneces alter binding affinity
used to recognize CD47 signaling to preent phatgocytosis
highly polymorphic between individuasls
differences in SIRPa between individuals = differencs in binding intensity = differences in myeloid cell activation
not alwasy functional due to bidning affinity
myloid cells may mature, traffic to lymph nodes, and activate t cells even if activated
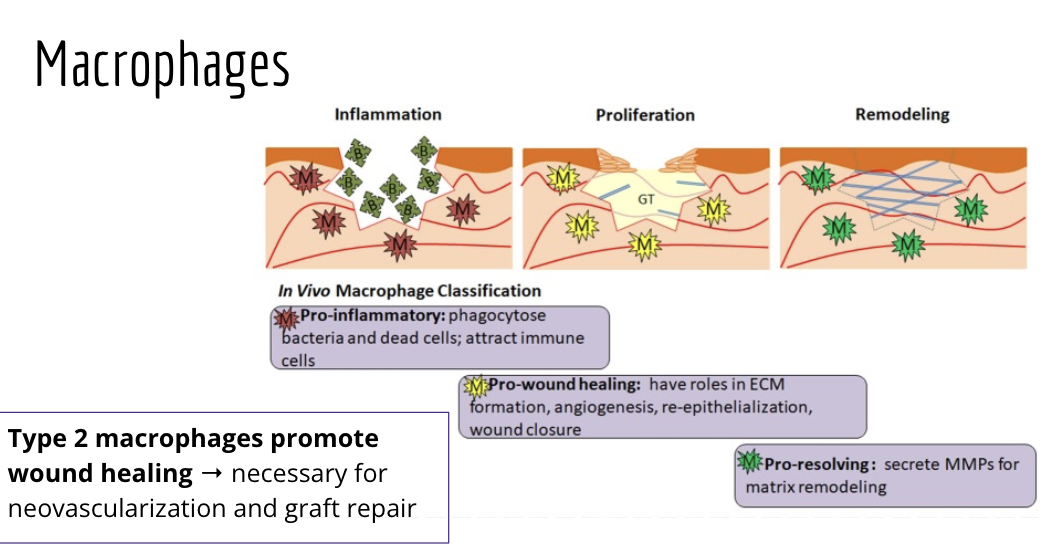
What are the function of macrophages? do they help or prevent graph rejection
they are pro inflamatory, pro-wound healing, pro-resolving
pro inflamatory: phagocytose bacteria and dead cells to attract immune cells
bad for grafts
pro wound healing: have roles in ECM formation and wound closure
mediu, for grafts
pro-resoliving: they secrete mmps for atrix reoeling
pro graft rejection
type 2 machrophages promote wound healing = necessary for neovasucalirzation and graft repair
What are some of the characthersitcs of innate lymphoid cells
lyphoid cells with innate charachteristics
they are generally tissue resident
nk cells circulate with them as well
they are involed in tissue homeostasis and have specific function dependent on the tissue
they are often protective but not alwasy
can be donor or recipient derrive = some donor dervied cells may stay longterm
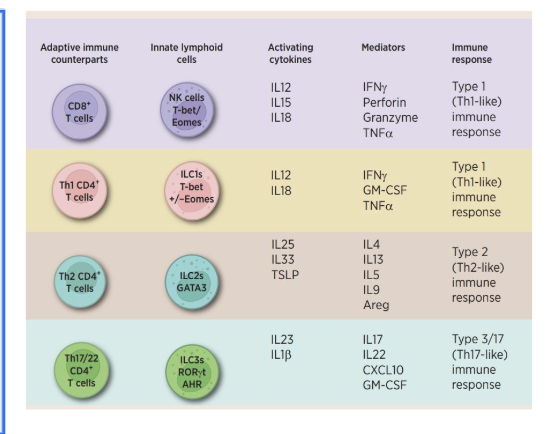
What are some of the innat lymphoid cells and what are their function. different types of nk cells, gama cells, ilcs, nkt cells
NK cells → analogous to cytotoxic CD8+ T cells
activated independent of antigen
integrates many activating and inhibitory signals = quantity and quality of donor cell signals
is donor and tissue stressed = morea ctivation
the target cell, whether the MHC1 is signaled or not tells the nk cell to kill it or not to kill it
ILC1/ILC2/ILC3 → analogous to helper CD4+ T cell subsets
il2 = th1, il2 = th2, il3 =th17
they are activated independent of antigen
NKT & 𝛾ẟ T cells: CD1-restricted lipid antigen recognition
Type 1 NKTs → analogous to CD4+ T cell subsets
often activated independant of antigen but can be activated by lipid antigens
Type 2 NKTs: mostly immunoregulatory
generally activated by lipid antigens
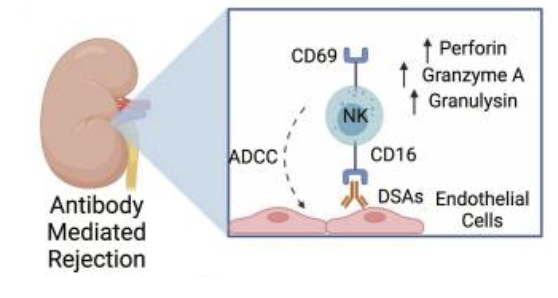
how are NK cells educated? describe both of their roles in graft protection and rejection
Protective mechanisms
Nk cells learn waht self is via MHC an inhibitory olecules during development
Recipeitn NK cells recognize missing self on donor apcs = hinders direct allorecognition (protective)
non-self MHC may act as an activating lignad for NK cells
rejection mechanisms
non-immune cells in the graft dont express recipeint-MHC
Mechanisms that mediate rejection
non-imune cells in the graft also doesnt express recipient-MHC
Donor-specific antibodies (DSAs) against vasculatore can trigger ADCC
other mechanisms may also be involved = imbalance and activating and inhibitory receptors
What are the different types of NKT cells and are tehy protective or adverse for grafts
NKT cells are generally protectiv
they are beneficial in pancreatic islet, cardiac, the liver, and the kidney
apcs and injured cells upregulate CD1d= damp production + lipid antigen presentation = nkt cell activation = anti inflammatory cytokine production = immunosuppresion
Type 1 NKT cells
nkt1s = proinflammatory and promote ischemia-reperfusion injury, can induce cell death
NKT2s and NKT10s = immunosuppresive cytokin production = tolergenic
Type 2 NKT cells
they inhibit type 1 = reduced macrophage and neutorphil accuulation = reduced ishceia-reperfusion injury
Similar to CD4+ subsets
γδ T Cells. What do they do and do they promote rejection or torance
can mediate both depending on the tissue and circumstances
can behave like a Treg or a Th17 cell depending on the situation
Th-17 like pheonotype = inflammatory cytokine produciton and neutrophil recruitment (promotes rejection)
treg-like phenotype = antiinflammatory cytokine produciton and T cell inhibition = promotes tolerance
What makes liver transplants so succesful
they are constantly being exposed to environmental antigens from GI tract and spleen via the portal vein = generally tolerogenic area
imunosuppresive masures can be seased for some and some may be able to achive spontanious operational tollerance
they are highly regnerative = you can recive a partial liver from a living donor or a total liver from deceased donor
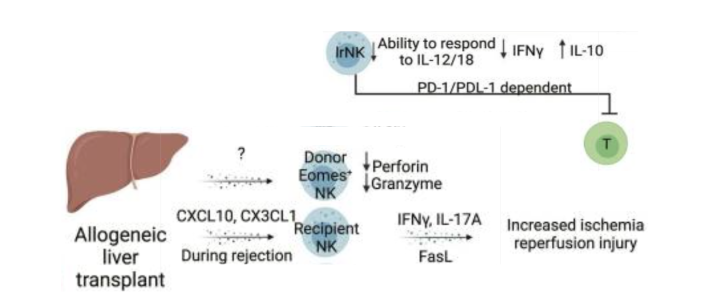
What immune cells allow for liver transplant protection or rejcection
Donor-derivd NK cells can persist for over a decade and are generally protective
reduced pro-inflammatory and increased anti-inflammatory cytokine production
inhibits T cells via PD-D1/PD-L1 axis
infiltrating recipeint NK cells may promote ischemia-reperfusion injury rejective
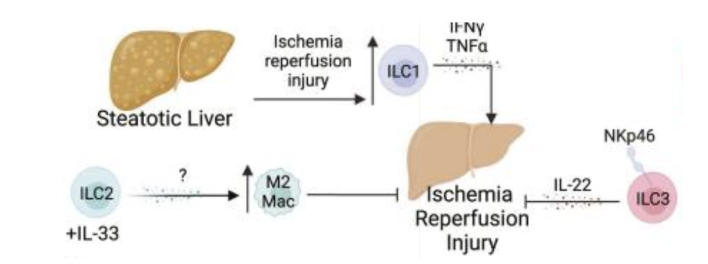
What are teh effects of the ILCs on lier transplants? protection or rejectin?
ILC1s
Worsen ischemia-reperfusion injury (IRI)
ILC2s
Induce type 2 (wound healing) macrophage activity
May contribute to chronic fibrosis - can lead to chornic graft rejection
ILC3s
Protective against ischemia-reperfusion injury
May promote regeneration
Most data comes IRI model of steatotic liver, limited allogeneic data
What are the effects of γδ 1 and 2 cells on liver transplants
γδ 1 cells: generally immunosuppressive
γδ 2 cells: generally inflammatory
Spontaneous tolerance observed in recipients where γδ 1 > γδ 2
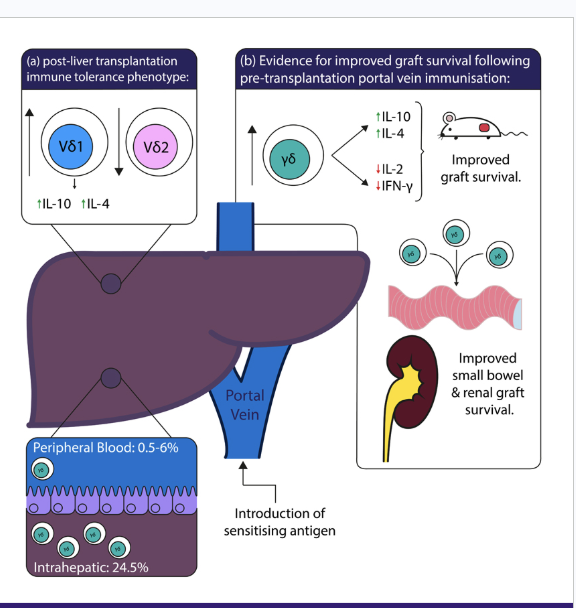
What are some of the charachteristics of bone marrow transplatns (HSC)
you dont really ahve to take the bone marrow anymore
G-CSF (granulocyte colony stimulating factors) produced by cells throughout the body to induce HSCs to movilize during homeostasis and inflammation
80% of bone marrow donationsa re now given thorugh a peripheral blood stem cell transplantation
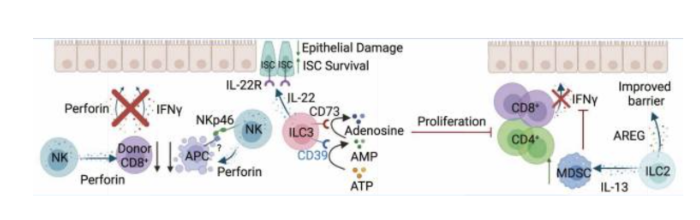
What are the function of nk cells, gamma cells, and ILCS in bone marrow transplant. What is the mechanism
NK cells
protect from graft vs host disease and promote graft vs host leukemia
impare APC and T cell funciton
ILC2s
Barrier maintenance
Induce anti-inflammatory myeloid cells (myeloid-derived suppressor cells, MDSCs)
ILC3s
Recipient-derived ILC3s protect gut tissue from GVHD
Barrier & stem cell protection
Inhibit T cells
γδ T cells may help engraftment & be protective against GVHD (conflicting data)
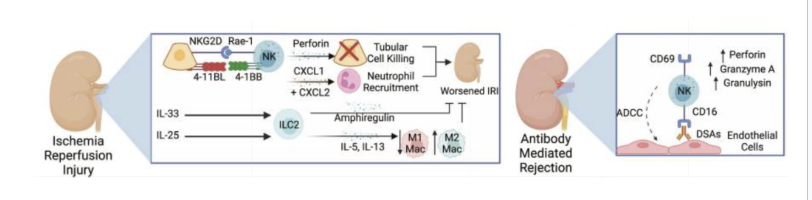
What are the stats for kidney transplants. What are the effects of NK cells, ILC2s and gamma cells
About one third of transplants are from living donors
Better immediate kidney function & less risk of rejection from living donor
Circulating recipient NK cells
Recruit neutrophils & kill renal tubule cells → exacerbates ischemia-reperfusion injury
Kill graft vascular cells via ADCC (donor-specific antibodies)
ILC2s
Stimulate cell growth & survival → minimizes ischemia-reperfusion injury
Promote type 2 (wound healing) macrophage activity
γδ T cells
Recruit αꞵ T cells & cause renal tubule damage
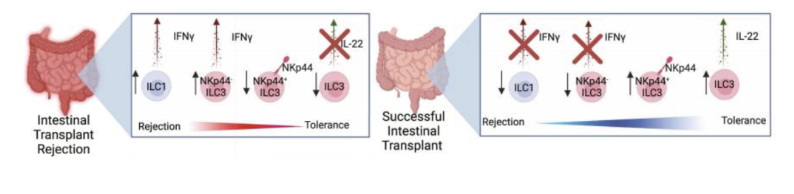
What are the stats for intestial transplants? What are the efeects of NK cells and ILcs
intestines house large an complex lymphoid tissue
there is a substancial number of donor derived ILCs sustaind for years = gradually transitions to recipient derived ILCS
Most donor-derived ILCs are NKCls, ILC1s, ILC33s,
ILC1s may facilitate rejection
ILC3 and gama t cells seem protective
What are the stats for Lung Transplatns? What are teh effects of NK cels and ILCS
Nk cells exacerbate iscehmai reperfusion injury but may also kill donor APCs
ILC3 = tolrance
ILC1 = protective
ILC2 - recrut eosinophils = tolernace
gama t cells = niflammatory (rejective)
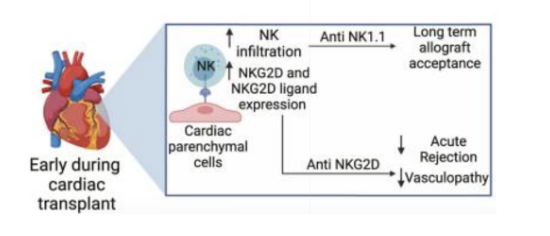
Heart transplants what are the effects of ILC2, nk cells, and gamma bea cells
Difficult to study → limited samples, mostly mouse
Most helper ILCs are donor-derived ILC2s → little infiltration of other recipient helper ILC subsets or precursors
NK cells
Accelerate both cellular- and antibody-dependent graft rejection
Contribute to vasculopathy
γδ T cells generally inflammatory
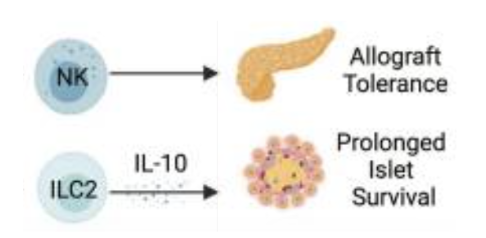
What are islet transplants (pancreatic) vs whole pancreus transplants? what are teh differences in procedure and risks? What are teh effects of nk cells, ilc2s and gama theta t cells
used to treat patients with type 1 diabetes
Can be either a major surgery or just islets
Islet transplant: lower surgical risk, successful at eliminating severe hypoglycemia & glucose changes but may require multiple donors to achieve insulin independence
Whole pancreas: higher surgical risk, but higher likelihood of insulin independence after single transplant
NK cells generally promote tolerance, likely by killing donor APCs
ILC2s generally protective & can induce myeloid cells to secrete retinoic acid, which activates insulin secretion
γδ T cells may be protective
How does microbione impact transplatnation
micorbionme at mucosal sites (lungs, gut, skin eyes, ect) are ky to tissue homeostatsis both locally or systematically
recipeint receives donor microbiome of mucosal organs
changes dynamically following transplatnation = local and sytemic physiologicy can be impacted
microbial communities and metabolites produced = behavior of tissue and sytemic cells (both immune and nonimmune)
ability and metabolism of immunosuppresive drugs
can affect transplanatation outcome (tolerance and rejection
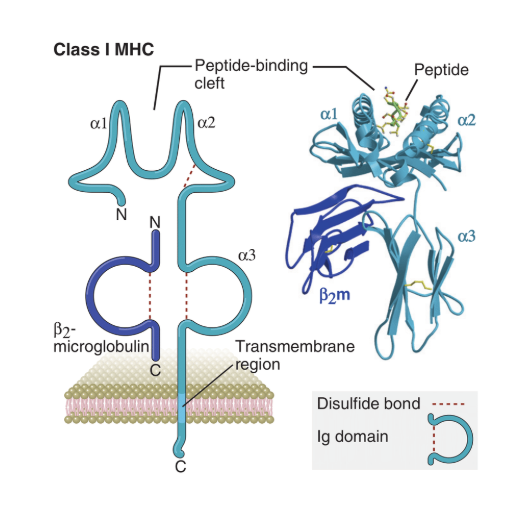
MHC and HLA diversity - What makes them polymorphic, what parts changes,
MHC proteins bind a restricted number of antigens based on their structure
HLA alleles are the most polymorphic genes of a person (humans are 99.9% identical to eachother)
its a 3 domain protein with 3 alpha helixes and beta sheets
Broad antigen coverage within the population to poulation-level protection
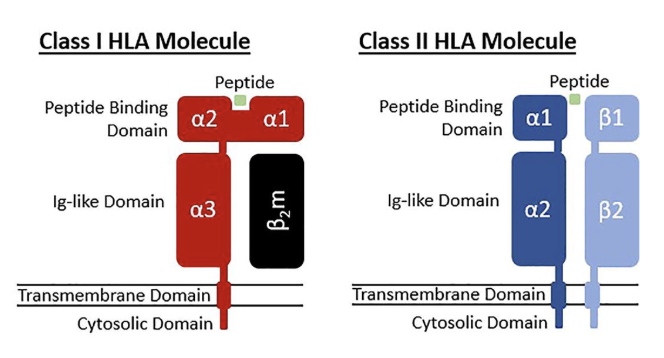
What are the different types of HLAs and which part differentiates them
HLA class I: HLA-A, HLA-B, HLA-C
>28,000 known alleles
they differentiate in their alpha chains but constant beta domain
HLA class II: HLA-DR, HLA-DQ, HLA-DP
>12,000 known alleles
Separate genes for alpha (A) and beta (B) chains
Non-classical HLAs: HLA-E, HLA-F, HLA-G
>500 known alleles
How does HLA match? What are the most importaint chains of hlas? Where are the Gene locuses, the serological group, designated allele, and genetically distinct area: HLA-a*01:01:01:01
Most important = HLA-A, HLA-B, HLA-C, HLA-DRB1 (Beta cahin 1 of HLA-DR)
rapid in depth sequencing of HLA genes at nucletide level
hla genes with similar reactivity to certen sera were grouped
Genetically dsing area
they are genetically distinct but phenotypically identical alleles
noncoding dna changes or those that dont result in differnt amino acids

How are HLA genes distributed from parents? How are HLA matching happen per parents? what are the ratios between siblings
1 hla from each parent is distrubuted both HLA 2 and HLA 1
Parents and children
Half-atch
Full siblings
25 chance of perfect match
50 haploidentical
25 chance of no match
Crossing over happens (1%) —> reare to get new haplotypes within family
Chance of an exact match with unrelated donor: <1 in 100,000
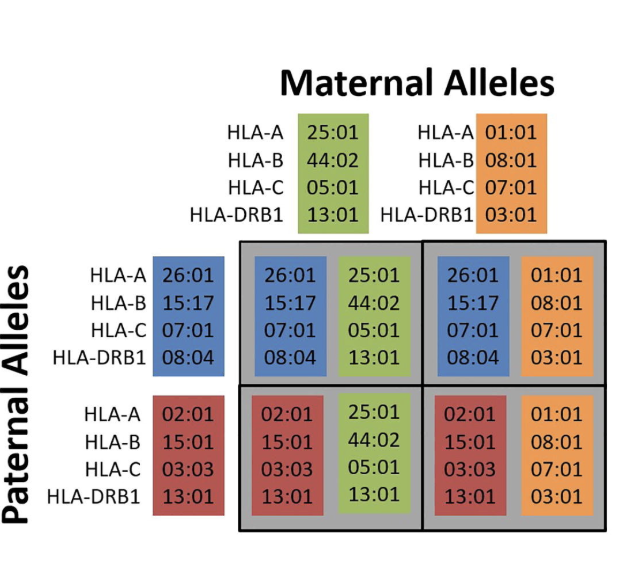
What is linkage disequilibiru? What does it mean with HLA matching
Linkage disequilibrium: Some combinations of HLA alleles are more common than others because of mechanism of inheritance
not necessarily matching 8 genes independently
Linkage disequilibrium means that certain genetic variants tend to be inherited together more often than expected by chance.
Why this happens
Genes or genetic markers that are physically close together on a chromosome are less likely to be separated during recombination (crossing over) in meiosis. Because of this, they often travel together from parent to child.
some haplotypes are more common = easer for some recipients to find donors thatn others
note that databses skew towards white people of european descent
What are the different types of HLA matching in different organs: Bone marrow, kidney, heart and lungs, liver, and cornea
Bone marrow: requires strict matching to prevent graft-vs host disease
Kidney: HLA matching is used becuase patients can remain on dialisis for long periods of time
Heart and Lungs: frequently ismatcehed; would benefit from bettter HLA matching, but trnasplatns are. urgent and organs are limid
Liver: not currently clinically considered because liver is more tolerogenic but research suggest HLA matching could be beneficial
Cornea: not requried (no vascularization so its not needed)
improved immunosuppresion protocas are bridging the gap between matched and mismatched graft survival
What are the effects of minor Histocompatability antivens on HLA matching? when are HLA matchs 100%? When is immunosuppression ecessary
no perfect matching unless identical twins
you ned immunosuppresion is essentail for everyone else
Minor histocompatability antigens: antigens that are different between recipeint and donor are not HLA
Y chromosome genes transplanted into an XX recipient are foreign
Mostly autosomal & mostly unknown
Reject more slowly than HLA mismatches
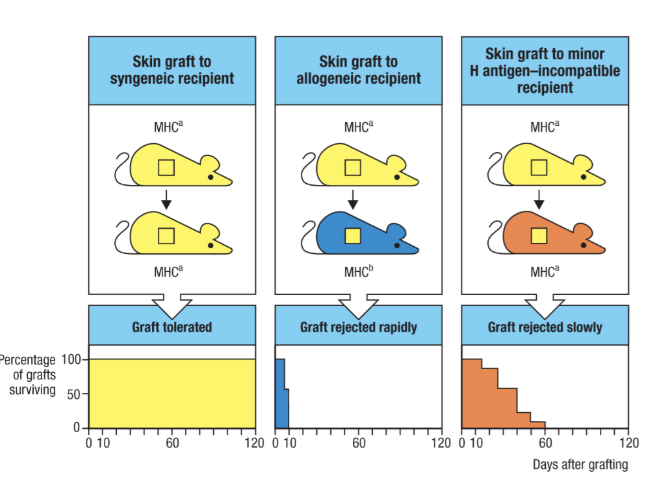
What are some challengs to HLA matching
Some haplotypes are more common than onters = there may not be many mathces avaliable
some transplants are too. urgent = no time to wait for a perfect match
even perfect HLA match is allogenic (aside from identical twins) immunosupprefsion is essential regardless
What are the results of early high does radiation studies
high level radiation exposured resulted in repressed bone marryow, low white and red blood cell counts, increased chance of infection
bone marrow transpalants from any strain (chimerism) could restroe functioning, but only skin grafts from the donor strain would be tolerated
thre were attempts to induce adult tolerance but that resulted in graft vs host diease
What were the conditions for the first succesfful kidney allograft
fraternal twins, oner eicived total body irridation but no other immuno suppresion
most likey benefitted from HLA matching
What is high does readiation used for an dnot used for
NOT used for…
General immunosuppression
Inducing chimerism or adult tolerance
Used for…
Treating blood disorders (especially leukemias & lymphomas)
Ablating recipient bone marrow prior to receiving a bone marrow transplant
What is tolerance vs immunosuppression
The Ultimate Goal:
Tolerance: immune system recognizes graft as healthy/self and does not attack it
But beggars can’t be choosers…
Immunosuppression: immune system recognizes graft as dangerous/non-self but is unable to effectively reject it
Also less able to reject things like pathogens and cancer cells…
What are some of the early immunosuppression methods and what are their drwabacks? Benzol? Nitorgen mustard
benzol.
usd as an industrial sol;ivent in the late 19th centrury
stopped when learning it casues leukimia
Nitorgen mustard
used as a poison during WW1
used experientally to treat mous and human lymphomas and leukimia but difficult to used
What is 6-mercaptopurine (6-MP)- early immunosuppresision methods
used as a potential suppressant
chemotheropy drug that cuases bone marrow suppression
more predictable effects tahn random conntinous ise = could extend survival of skin grafts survival
iniital use were a faleure b/c the patients it were used on all died
What steriods were used during WW2 and what did they do: Cortisol
Cortisone was initaually used to counteract. hypoxia so pilots could fly higher at altitudes
war use was doubtful btu seen to have other theraputic benifits
Used to treat arthuritis
it reduces antibody functions and allows grafts to last onger even afther withdrawl
results wer inconsistent and not dramatic enough for continued research
What is thomas starzl’s contribution to organ transplantation resaerch. what steroids did he use
Azaathioprine and Prednisone
used together improved graft survival in dogs (especially in the liver)
he used studies in the liver = less powerful rejection and occasional spontaneous reversal of rejection
rejection seen only with azathiroprine but uses of predisone fixies that
reccomended flusing and cooling of donor organ
His contributions
he is considered the father of rogan trasnplantations and his protocaol became standard for the liver kindey and heart for 32 decades
new transplantation centers
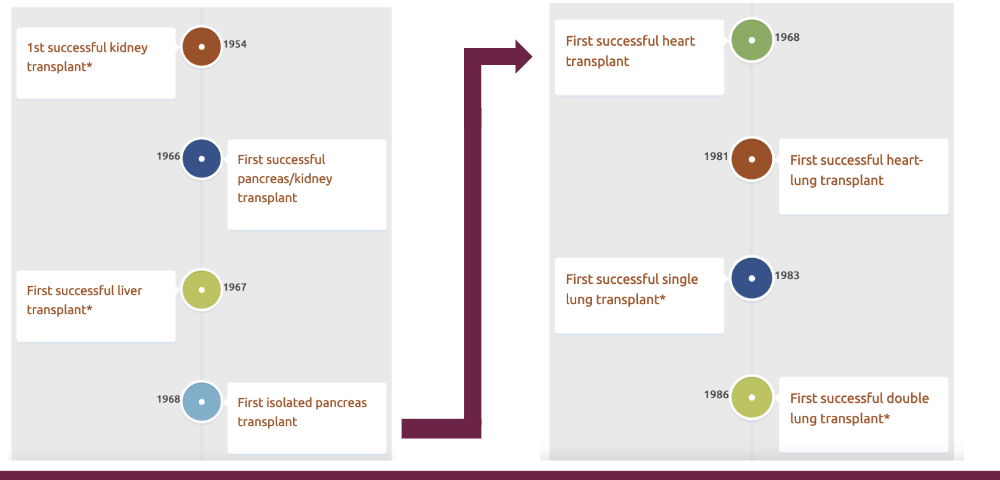
What was the order of the first transplantations. waht came first waht is most recent
look at diagram
How do you treat chronic graft rejection? what is usually seen
steroids
need for new protocols for chronic care
Chronic issues:ucommon infections (
Chronic issues: uncommon infections (usually controlled in immunocompetent patients), reactivation of infections (tuberculosis, herpes, CMV, etc.), vascular stenosis at sites of anastomosis
Chronic side effects of steroids: poor wound healing, diabetes, pancreatitis, gastrointestinal hemorrhage, muscle loss
Need to balance risk of graft rejection with risks associated with treatment
What are the steps to. modern day intervention
Induction
Immediately after transplantation to prevent acute rejection
Not for long-term use
Includes: OKT3, Basiliximab, and others
Maintenance
Before, during, and/or after transplantation for long-term maintenance
Includes: Prednisone, Cyclosporine, Tacrolimus, Azathioprine, Rapamycin, and others
Anti-Rejection
Treating acute rejection episodes during initial post-transplant period or later follow-up periods
Same as induction drugs
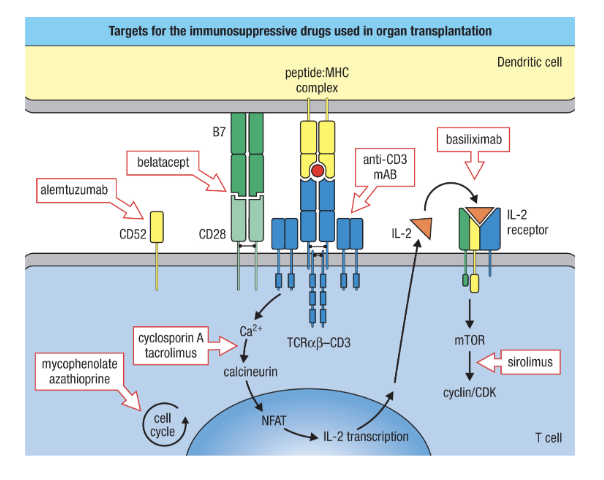
What are the steps to modern immunosuppression. Nechanisms - draw it out
they inhibit broad general inflammatory gene transciirption
sterridos (prednstone)
target T cells for destruction
anti CD3
Alemtuzumab
Prevent T cell activation and proliferation
Belatacept (synthetic CTLA-4)
Basiliximab (IL-2R antagonist)
Mycophenolate & azathioprine (cell cycle inhibitors)
Cyclosporin A & tacrolimus (Ca2+ signalling inhibitors)
Sirolimus (“rapamycin”) reduces IL-2 signalling
Provide at least two examples of how World War II impacted transplantation research. Consider impacts during and immediately following WWII (the Cold War Era).
1939 - 1945
Increased mechanization of warfare → aerial combat
Fighter pilots often suffered burns from engine fires located in front of cockpit
~4,000 aerial burn cases alone in France & Britain
Large number of burns during naval ship sinkings
Required emergency care & plastic surgery
Britain prioritized the study of burns
Cold war
competition between soviets and the us
fear of nucelar war = increased reserach into the effects of radiation and radiation exposure
increased intrerest in surgical techniques and developkent