CHEM 2 TEST 2
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
Arrhenius Acid and Base definition
Acid
Source of H+ (i.e., HCl)
Base
Source of OH- (i.e. NaOH)
Brönsted-Lowry Acids and Bases definitions
Acid
Proton (H+) Donor (i.e. HCl)
Base
Proton (H+) Acceptor (i.e. NH3)
Lewis Acids and Bases definitions
Acid
Electron Pair Acceptor (i.e. BF3)
Base
Electron Pair Donor (i.e. NH3)
Which one is most commonly used?
Brönsted-Lowry Acids and Bases
Acid
Proton (H+) Donor
Base
Proton (H+) Acceptor
In an acid-base reaction, H+ is transferred from the acid to the
base. Water is frequently a product.
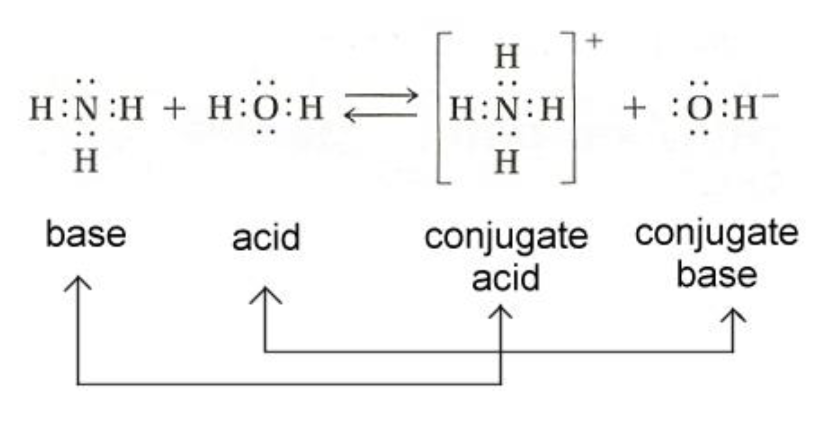
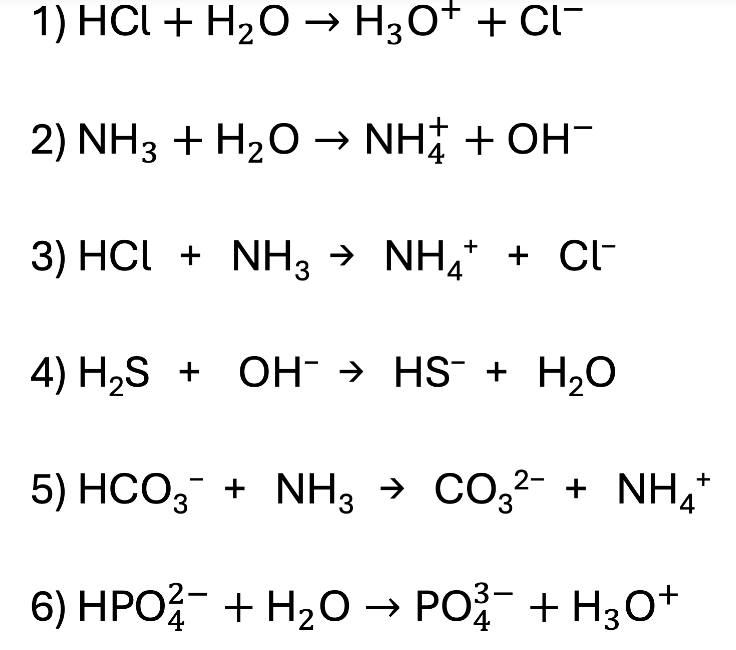
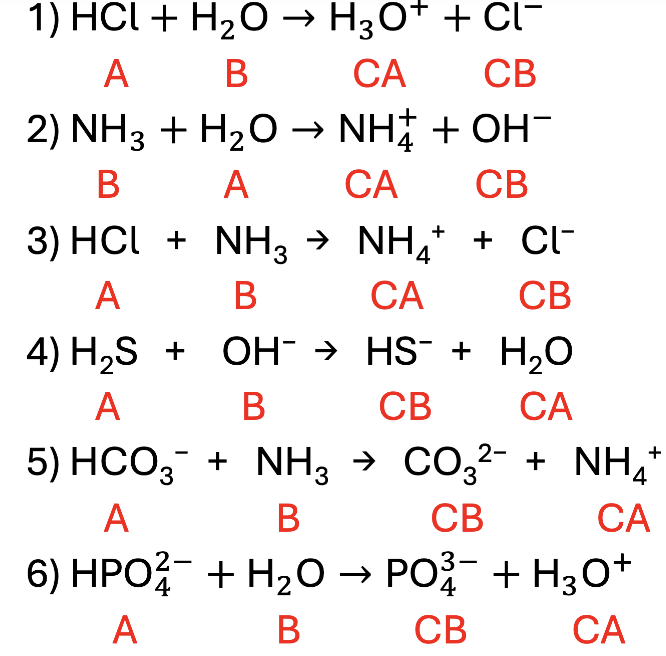
Practice: Label the Acid, Base, Conjugate Acid, and Conjugate Base
Acid Base Answers!
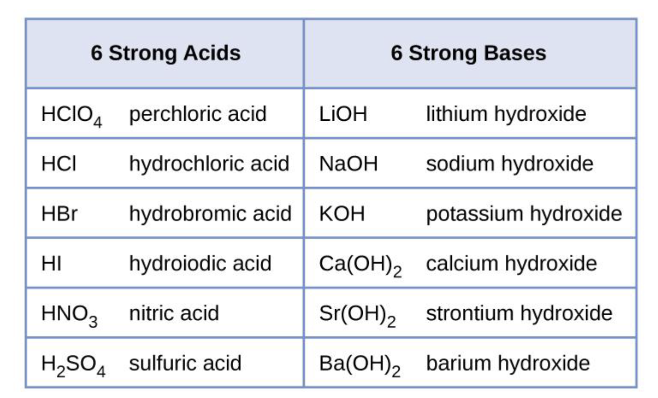
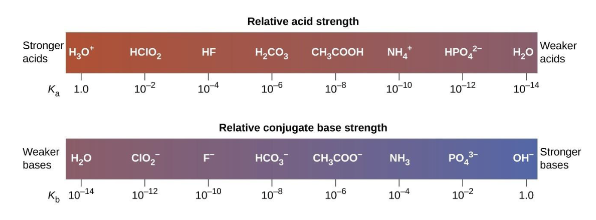
Strong Acids and Strong Bases: What’s unique?
Strong acids and bases are defined as those that fully dissociate in water (break apart into ions)
HCl → H+ + Cl- (This happens fully in water, Ksp is >>>>1)
The _____ the acid, the _____ (____ ____) the conjugate base
Stronger, weaker (more stable)
What is unique about water?
Water is unique because it is amphoteric (or amphiprotic)
Means it can act as both a Brønsted-Lowry acid (donor) and a base (acceptor) depending on the substance it interacts with.
It has a neutral pH of 7, yet it self-ionizes into hydronium (H3O+) and hydroxide (OH-) ions.
What are neutral conditions?
Neutral conditions occur when an acid and a base react to perfectly balance each other ([OH-] = [H+]), resulting in a pH of 7, typically forming water and a salt.
In this state, the hydrogen ion (H+) concentration equals the hydroxide ion (OH-) concentration. Examples include pure water, sugar solutions, and common salt water.
Explain the auto-ionization of water, kw, and shifting equilibrium/pH chnages.
Water autoionization is an endothermic process (2H2O = H3O+ + OH-) where water acts as both acid and base, forming ions.
The equilibrium constant, kw= [H3O+'] [OH-], is 1.0 × 10-14 at 25*C .
As temperature rises, the endothermic reaction shifts right, increasing kw, increasing H+ concentration, and lowering the pH (making it < 7), while remaining neutral [OH-] = [H+].
![<ul><li><p><span>Water autoionization is an endothermic process (2H<sub>2</sub>O = H<sub>3</sub>O<sup>+</sup> + OH<sup>-</sup>) where water acts as both acid and base, forming ions. </span></p></li><li><p><span>The equilibrium constant, k<sub>w</sub>= [</span>H<sub>3</sub>O<sup>+'</sup>] [OH<sup>-</sup>]<span>, is 1.0 × 10<sup>-14</sup> at 25<sup>*</sup>C . </span></p></li><li><p><span>As temperature rises, the endothermic reaction shifts right, increasing k<sub>w</sub>, increasing H<sup>+ </sup>concentration, and lowering the pH (making it < 7), while remaining neutral </span><mark data-color="rgba(0, 0, 0, 0)" style="background-color: rgba(0, 0, 0, 0); color: inherit;">[OH-] = [H+]</mark><span>.</span></p></li></ul><p></p>](https://assets.knowt.com/user-attachments/f74c6296-6f7a-49e6-a9fe-88edbdaf6f0a.png)
Practice: If Kw becomes 1 x 10-12 at a higher temperature, what is the pH of pure water at this temperature?
6!
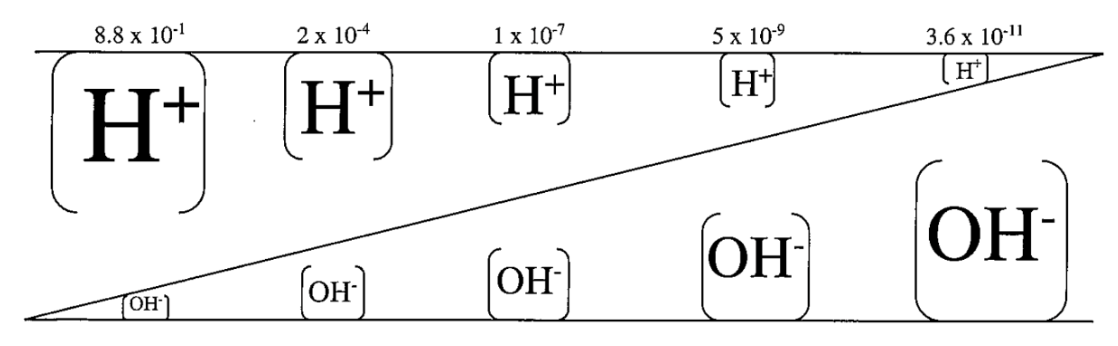
H+ and OH- are _____ proportional. Explain
Inversely!
As the [H]+ increases in water, the [OH-] goes down
1x10-14 = [H+][OH-]
14 = pH + pOH
What is the pH if the [OH-] = 1 x 10-3?
11
Explain converting: Ka or Kb to pKa or pKb
What is the relationship between the two?
Acid dissociation constant:
HA⇌H++A−
Ka= [H+] [A−] / [HA]
Relationship:
pKa=−log(Ka)
For bases:
pKb=−log(Kb)
Important relationship:
Ka x Kb=Kw
Explain converting: [H+] or [OH-] to pH or pOH.
What is the relationship between the two?
Definitions:
pH= −log[H+]
pOH= −log[OH−]
Relationship:
pH+pOH=14
1x10-14 = [H+][OH-]
(Trick is that the negative exponent of a conc is the pH for 1.0×10^).
How do you calculate the pH of a strong acid or base?
They fully dissociated, so find [H+] or [OH-] then take -log to get
the pH or pOH
Example
[H+]=0.01M
pH=−log(0.01)= 2
Strong vs. Weak Acids and Bases
Strong – Fully Dissociate
HCl(aq) → H+(aq) + Cl-(aq)+
Assume Kc >>>> 1
Weak – Very little dissociates (< 5%)
HF(aq) ⇄ H+(aq) + F-(aq)
Kc matters!!
How do you determine the pH of a weak acid or base?
RICE tables!
Assume the amount dissociated is small relative to the starting concentration
What is the pH of 0.850 M HF?
HF(aq) ⇄ H+(aq) + F-(aq) Ka = 6.8 x 10-4
We still need to determine the equilibrium [H+], but we can’t assume full dissociation, like with strong acids
Need to use K to determine [H+]
Ka = [𝐻+][𝐹−] / [𝐻𝐹]]
6.8 x 10-4 = (𝑥2)/(0.850−𝑥)
Since x is small compared to 0.850M(definition of weak acid), we
can simplify the math by dropping the x in the denominator
6.8 x 10-4 = (𝑥2)/(0.850)
x = 2.4 x 10-2 M = [H+]
pH = -log(2.4 x 10-2) = 1.61
![<ul><li><p>We still need to determine the equilibrium [H+], but we can’t assume full dissociation, like with strong acids</p><ul><li><p>Need to use K to determine [H+]</p></li><li><p>Ka = [𝐻+][𝐹−] / [𝐻𝐹]]</p><ul><li><p>6.8 x 10<sup>-4</sup> = (𝑥<sup>2</sup>)/(0.850−𝑥)</p></li></ul></li></ul></li><li><p>Since x is small compared to 0.850M(definition of weak acid), we</p><p>can simplify the math by dropping the x in the denominator</p><ul><li><p>6.8 x 10<sup>-4</sup> = (𝑥<sup>2</sup>)/(0.850)</p></li><li><p>x = 2.4 x 10<sup>-2</sup> M = [H+]</p></li><li><p>pH = -log(2.4 x 10-2) = 1.61</p></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/20dbe313-2bf3-4c34-afb3-6408cd56deb4.png)
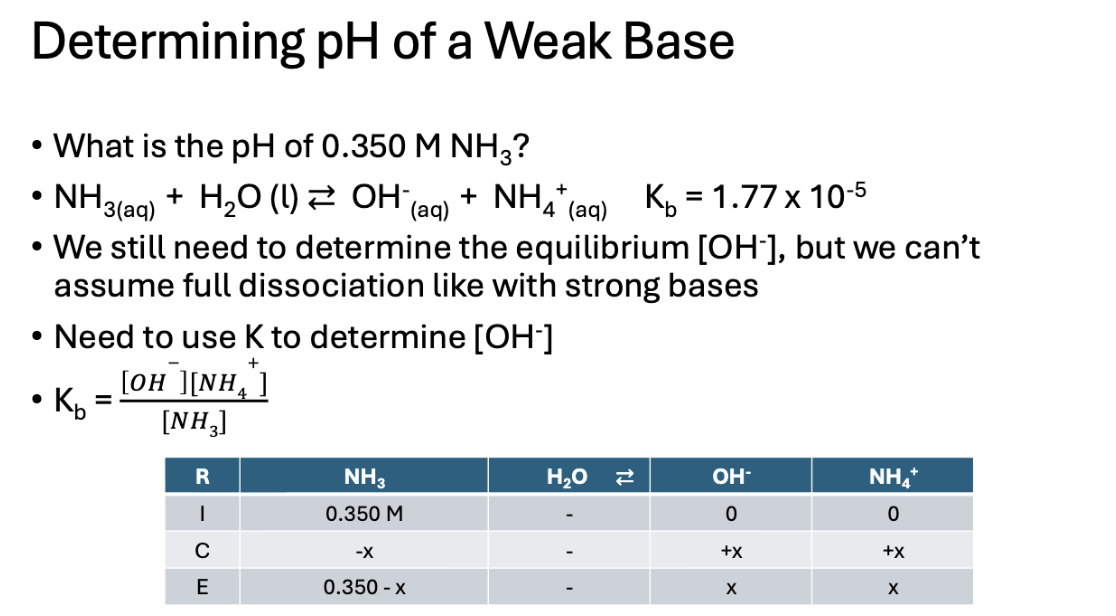
pH for a weak base
pH for a weak base answer

What is a shortcut for weak acids and bases that comes out in determining their pH?
Ka = x2 / [Acid] where x = [H+]
Kb = x2 / [base] where x = [OH-]
![<ul><li><p>Ka = x<sup>2</sup> / [Acid] where x = [H+] </p></li><li><p>Kb = x<sup>2</sup> / [base] where x = [OH-] </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/3bdc884e-7a88-4a26-96cf-c46376e47908.png)
What is percent dissociation?
% dissociation is another way to measure the relative concentration of H+ and the original weak acid
% dissociation = [H+] / [Acid] x 100%
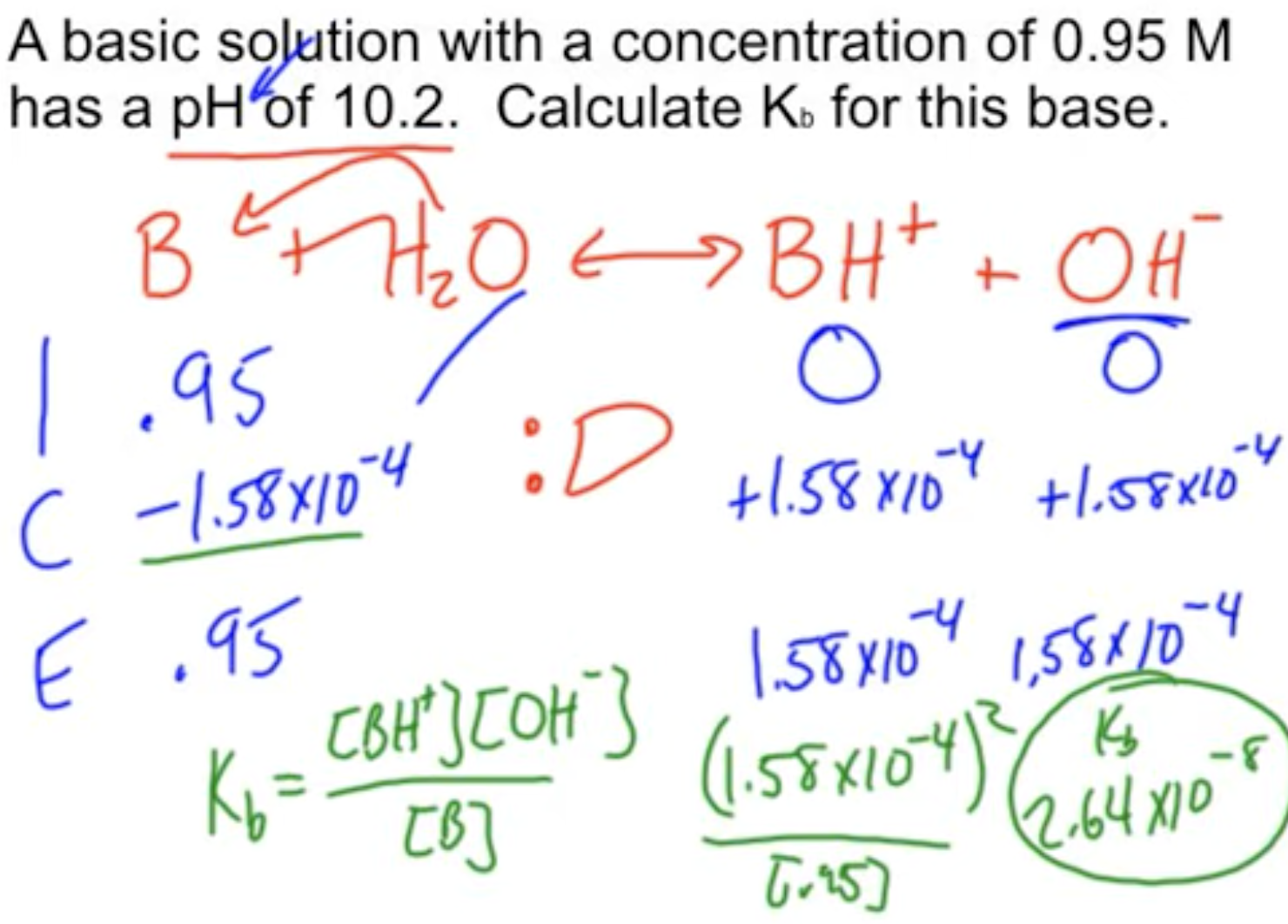
How to determine Ka or Kb from pH?
See the description here.


Ka from pH exmaple
Ka from pH exmaple

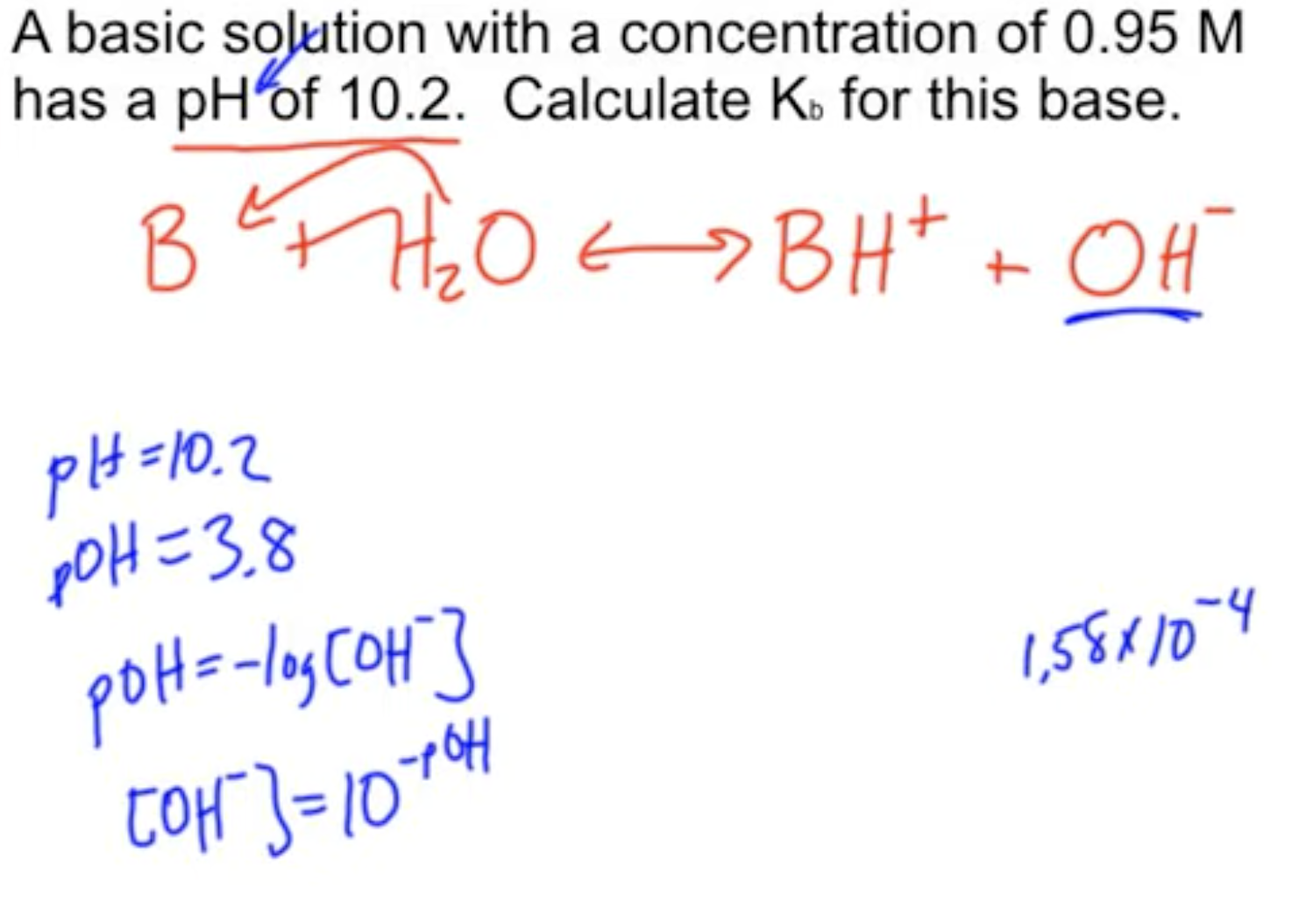
Kb from pH exmaple
Kb from pH exmaple
When do neutralization reactions occur?
When there is something of excess left over and we must determine the new pH.
Neutralization when the acid or base is strong
Find concentration and take -log to get pH or pOH
Neutralization Reactions
Steps:
Convert to moles
Determine limiting reagent
Find excess acid/base
Calculate concentration
Convert to pH/pOH
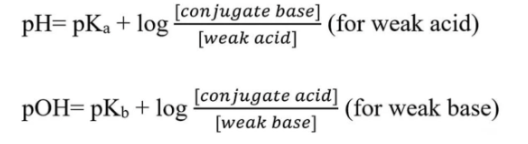
Neutralization when the acid or base is weak. What is the important conjugate concept with this?
Weak Acids Have Strong(er) Conjugate Bases.
Therefore, when we run a neutralization reaction of a weak acid or base, we need to consider the impact of both the acid and the conjugate base.
Whenever there is a mixture of a weak acid and its conjugate base (or weak base and its conjugate acid), we use the Henderson-Hasselbalch Equation to capture the impact of both species.
Look at the slides for practice on neutralization!
Look at the slides for practice on neutralization!

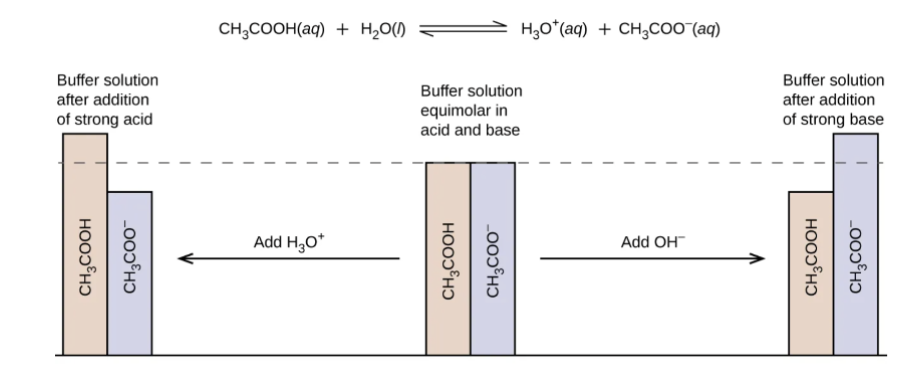
What are buffers?
Buffers are a solution that contains a weak acid/base and its conjugate.
Both the weak acid and the conjugate base can exist in solution together without neutralizing each other.
This creates a solution that is resistant to pH change when either a strong acid or a strong base is added to the solution.
Ideally, the buffer is made at the mid-equivalence point
What should the pKa of the acid be equal to? What should be used to calculate?
The desired pH of the solution!
The pH of the buffer will be equal to the pKa of the weak acid. Since the weak acid and CB are in a 1:1 ratio, the pH = pKa.
Remember, if Ka of the acid is 1 x 10-5 then the pKa is 5.
Henderson-Hasselbalch equation should be used for calculations
What is the ideal buffer and three ways to make it?
The ideal buffer will be a 1:1 combination of a weak acid/base and its conjugate (mid-equivalence point on the titration curve)
Three ways to get the 1:1 ratio (for a weak acid and CB)
1) Just mix a 1:1 mole ratio of the weak acid and its conjugate base.
(i.e. HF and NaF)
2) Mix a 2:1 mole ratio of weak acid to strong base. This will convert half of the weak acid into its conjugate base.
(i.e. 2:1 HF:KOH)
3) Mix a 2:1 mole ratio of the conjugate base with a strong acid. This will convert half of the base into the weak acid.
(i.e. 2:1 NaF:HCl)
Why are buffers important/ what is their primary purpose?
Buffers are resistant to pH change from a strong acid or base stress because the buffer components just change ratios.
Changing the ratio of weak acid and CB (even up to 10:1) has a relatively minor impact on pH
The weak acid will react with any strong base and convert it into the CB, which just changes the ratio in the HH equation
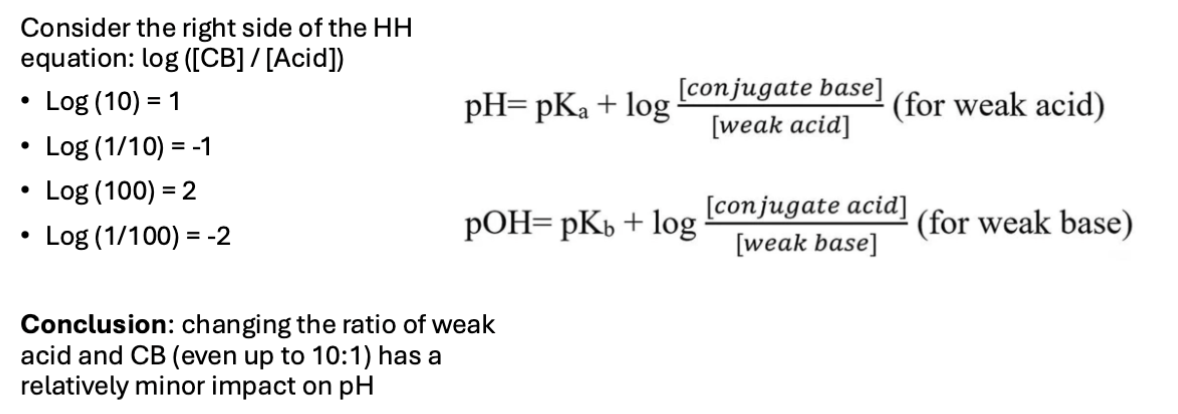
What is a buffer capacity?
A buffer can’t resist a pH change forever. Enough strong acid or base will eventually overwhelm the buffer and create a big pH change
The capacity of the buffer is simply the number of moles of weak acid or conjugate base in the solution.
Once it is all consumed, the strong acid/base will overwhelm the buffer and cause a big change in pH.
These require mole calculations!!
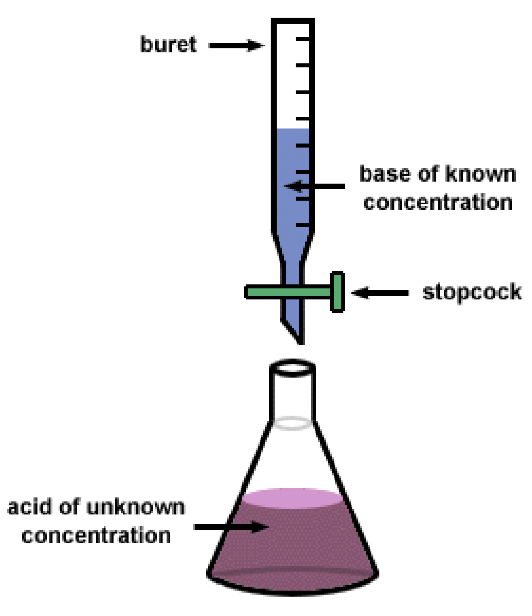
What is a titration? What is it often used for?
Titrations are a way of measuring the amount of a reactant needed to consume another unknown reactant
Uses a buret to measure the amount of reactant
Often used for Acid/Base and Redox Reactions
Color Indicators are used to determine the endpoint of the reaction
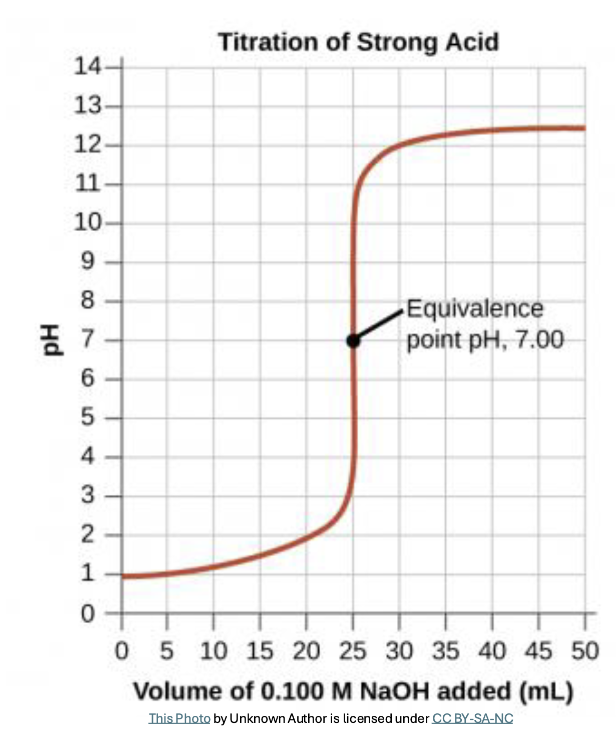
Strong Acid-Base Titrations. What do they look like, and what is the difference between the endpoint and the equivalence point?
A pH indicator is used to change the color to indicate the end of the reaction
Phenolphthalein turns pink at pH=10
This is called the endpoint, which is a real-world surrogate for the equivalence point
The equivalence point in a titration is the theoretical point where the moles of titrant exactly neutralize the analyte according to stoichiometry.
The endpoint is the practical, visual point where an indicator changes color
If the acid and base are 1:1 in the balanced chemical equation, you can use MAVA = MBVB to calculate the molarity of the unknown acid
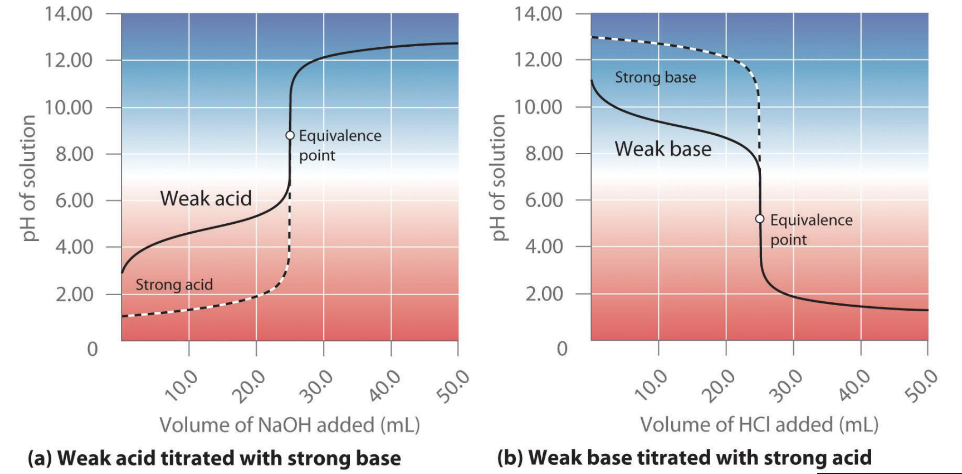
Titration of a Weak Acid with a Strong Base. How is this a different graph, and what is significant about the buffer region?
The equivalence point is still where moles of acid = moles of base (MAVA = MBVB)
Weak acid and CB exist together before the equivalence point (Buffer Region)
Mid-equivalence point (middle of buffer region), pH = pKa of the acid (HH equation)
At the Mid-Equivalence Point [Acid] = [CB], Log (1) = 0
![<ul><li><p>The equivalence point is still where moles of acid = moles of base (MAVA = MBVB)</p></li><li><p>Weak acid and CB exist together before the equivalence point (Buffer Region)</p></li><li><p>Mid-equivalence point (middle of buffer region), pH = pKa of the acid (HH equation)</p><ul><li><p>At the Mid-Equivalence Point [Acid] = [CB], Log (1) = 0</p></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/ae66e0da-a73d-4717-b85f-9e62c73f3be9.png)
What is unique about titrating a weak A/B with a Strong A/B? What do the curves look like, and what do they have?
They are slightly above or below standard equivalence at a pH of 7.
They also contain buffering regions before the equivalence point that resist sharp changes in pH.
Mid-equivalence point (middle of buffer region), pH = pKa or pOH = pKb
Titrating a polyprotic acid or base. Explain the basics and what it should look like.
A polyprotic acid is an acid that can donate more than one H⁺.
Each proton has its own dissociation step and own Ka value.
Important concept:
Each step becomes less acidic.
Ka1>Ka2>Ka3
Because removing a proton from a negatively charged molecule is harder.
Has more than one curve on the overall titration curve.
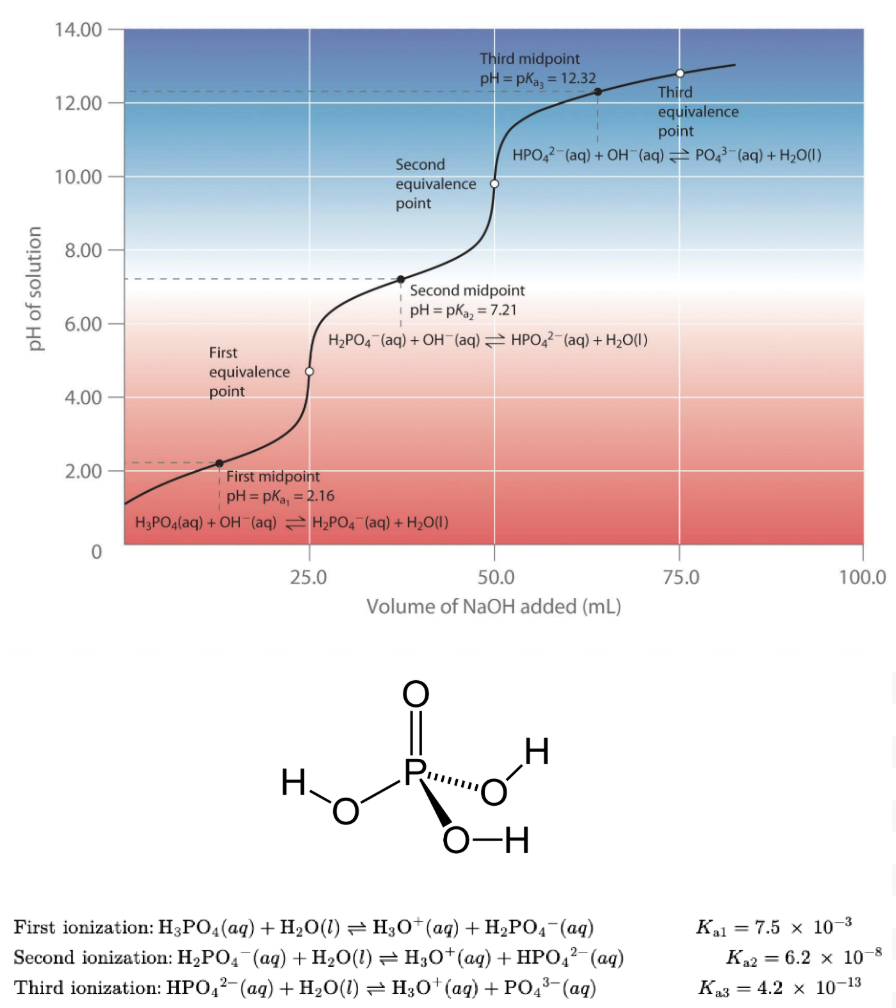
How should one chose out a good color indicator?
Color indicators should be appropriately chosen for an acid-base titration to change color at or just past the equivalence point
Choose an indicator with a pKa ~ pH at the equivalence point
This allows a sharp color change when you want it to occur
They themselves are weak acids whose equilibrium will shift according to LeChatelier’s Principle
In the graph, all three indicators work for the strong acid, but only phenolphthalein works for the weak acid.
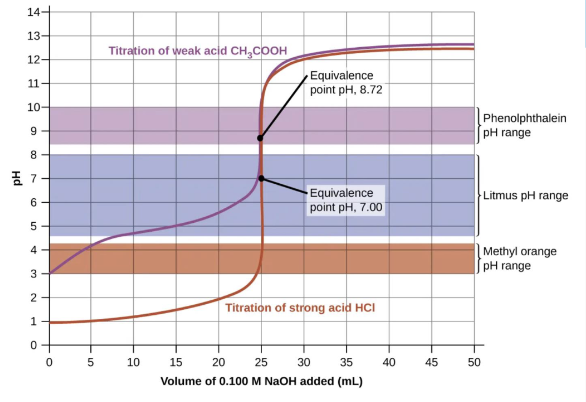
Weak acid with a strong base scenarios, how do you solve?
Initial pH
Before the equivalence point, where the WA is still in excess
pH at the equivalence point
Beyond the equivalence point
(WARM UP REVIEW SLIDE DAY 1, LOOK AT PAST QUIZ, DAY 18 NOTEBOOK, OR CHAT GPT FOR PRACTICE OF THESE!!!)
Weak acids do not fully ionize, so you must use equilibrium calculations. pH of a weak acid question. Shortcut!!
Buffers resist pH change because: Added acid reacts with A− and added base reacts with HA. So, pH changes slowly in this region. Henderson-Hasselbalch / Buffer type question.
This is where moles of base added = moles of acid originally present. All weak acids have now become the conjugate base. You can find the Kb of the conjugate base from the Ka of the weak acid. You can find concentration and solve. The conjugate base hydrolyzes water, producing OH⁻. That is why weak-acid titrations have an equivalence pH > 7.
Once a strong base is in excess, it completely controls the pH.
The weak conjugate base becomes irrelevant. Strong base in excess type problem.
Equilibrium will always favor the formation of the ____ acid. How can you know this? What about their conjugates?
Weaker
pKa table
Weaker acids will always have the larger pKa.
Will have a stronger conjugate base
Stronger acids will always have the smaller pKa.
Will have a weaker conjugate base
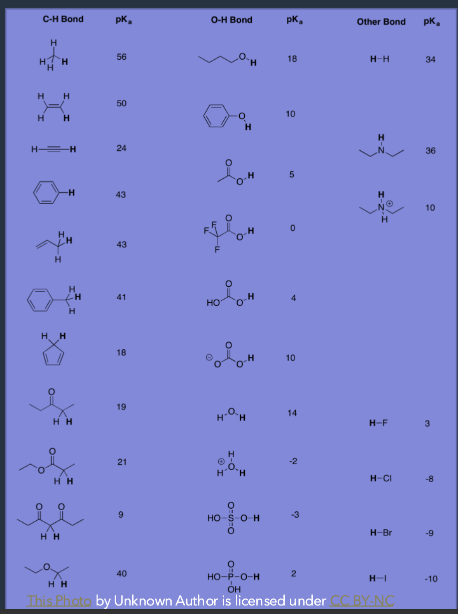
What is the ARIO method?
ARIO is a qualitative way to determine relative acid strength
If we can determine the most stable conjugate base, then we will know which acid is the strongest
Remove a proton (H+) from each to start the process
The ARIO method works by comparing the following:
Atom: What atom has lost the Ht, and can best accommodate the negative charge?
Resonance: Can the resulting charge be delocalized through resonance?
Induction: Are there electronegative elements available to help stabilize the charge?
Orbital - What is the hybridization of the atom that has the negative charge?
ARIO - Atom: Left to Right and Up to Down trends
Left to Right on Periodic Table
When comparing the conjugate bases of two acids, look at the atom that has the negative charge.
When comparing the stability of the anion, the atom that is more electronegative will be more stable
The more stable conjugate base corresponds to the stronger acid
Which is the stronger acid: H2O vs. HF
Look at the conjugate bases: OH vs. F-
F is more electronegative and therefore more stable so HF is the stronger acid
Up and Down on Periodic Table
When comparing the conjugate bases of two acids, look at the atom that has the negative charge.
When comparing the stability of the anion, the atom that is larger will be more stable
The more stable conjugate base corresponds to the stronger acid
Which is the stronger acid: HF vs. HCI
Look at the conjugate bases: F-vs. CI-
Cl is larger and therefore more stable so HCl is the stronger acid
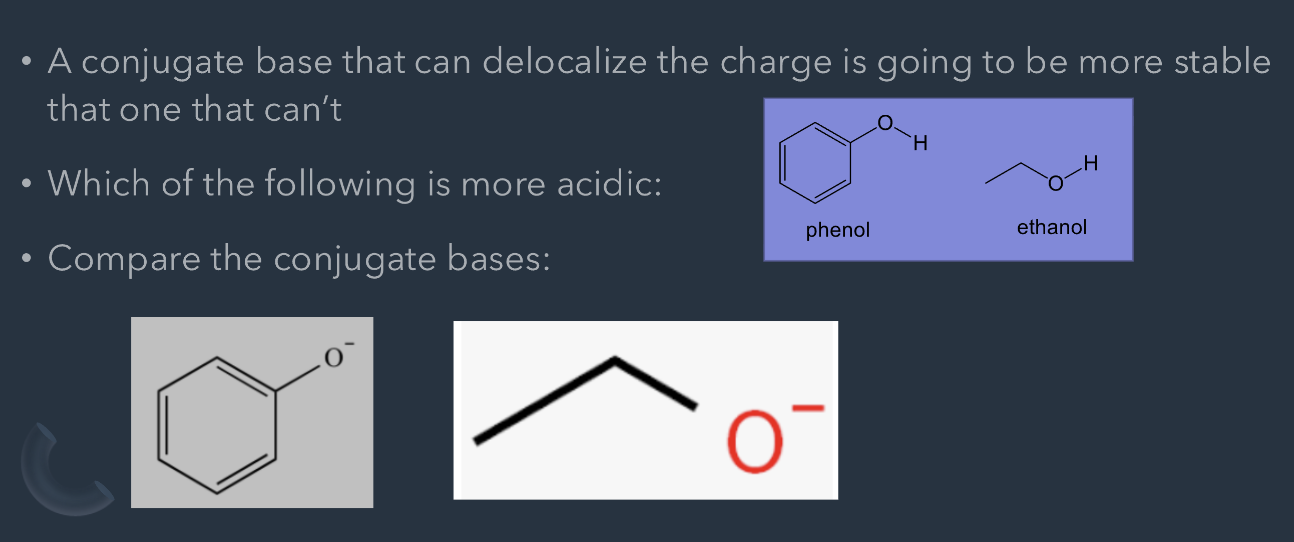
ARIO - Resonance
A conjugate base that can delocalize the charge is going to be more stable that one that can't
Which of the following is more acidic:
phenol
ethanol
Compare the conjugate bases:
Phenol is more acidic as it can be stabilized in resonance
ARIO - Induction
Electronegative elements that are several bonds away from a negative charge can still
help stabilize the charge through inductionWhich of the following is the strongest acid?
Il has the most electronegative element at a position close to the anion, which helps stabilize the conjugate base
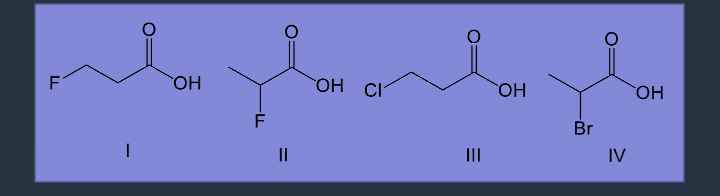
ARIO - Orbital
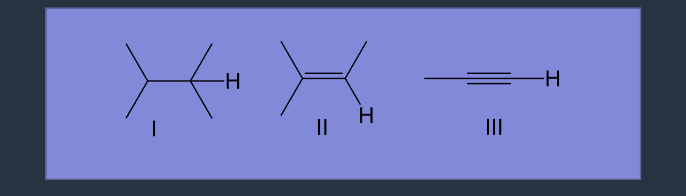
The type of orbital accommodating the anion on the conjugate base can impact the stability of the anion
sp> sp2 > sp3
Which compound has the most acidic hydrogen?
III
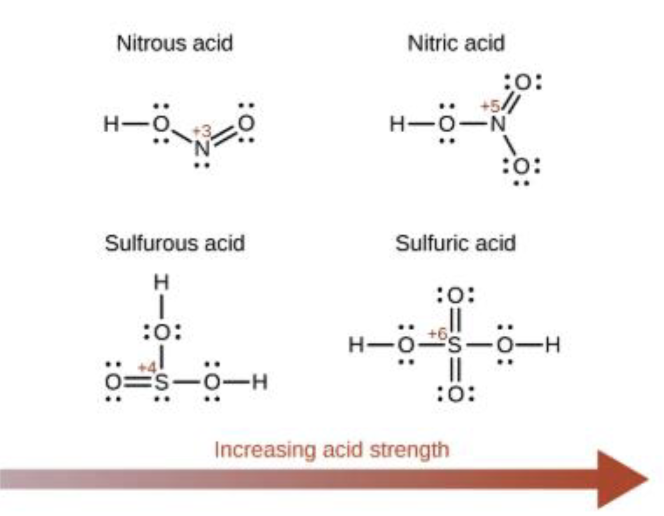
Oxy acids rule
Adding oxygen atoms to polyatomic ions improves the stability of the anion
More oxygen makes it more acidic
Combination of resonance and induction
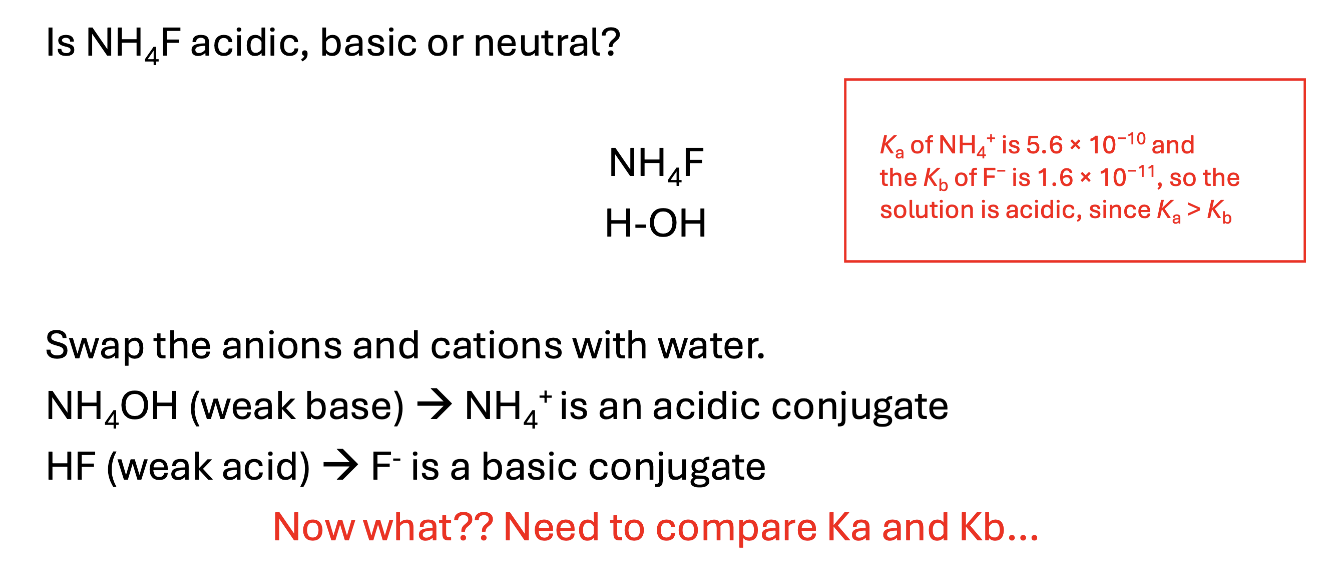
Given a salt, how to determine if it is acidic, basic, or neutral? What is significant about anions and cations?
Salt is a broad term for an ionic compound that contains a cation and an anion
Salts are often one of the products in an acid base reaction that can be isolated by removing the water at the end of the reaction
HCl(aq) + NaOH(aq) ⇆ NaCl(aq) + H2O(l)
Salt: NaCl
Neutral ions are derived from the strong acids and bases
Essentially, add OH - H to the two elements that make up the salt.
If they both make a SA/SB the salt is neutral
If they make one strong acid or one strong base, that overpowers and makes the whole thing acidic or basic.
Cations have the ability to be acidic and anions have the ability to be basic.
Cations: X+ + H2O ⇆ XOH + H+
Anions: X- + H2O ⇆ HX + OH-
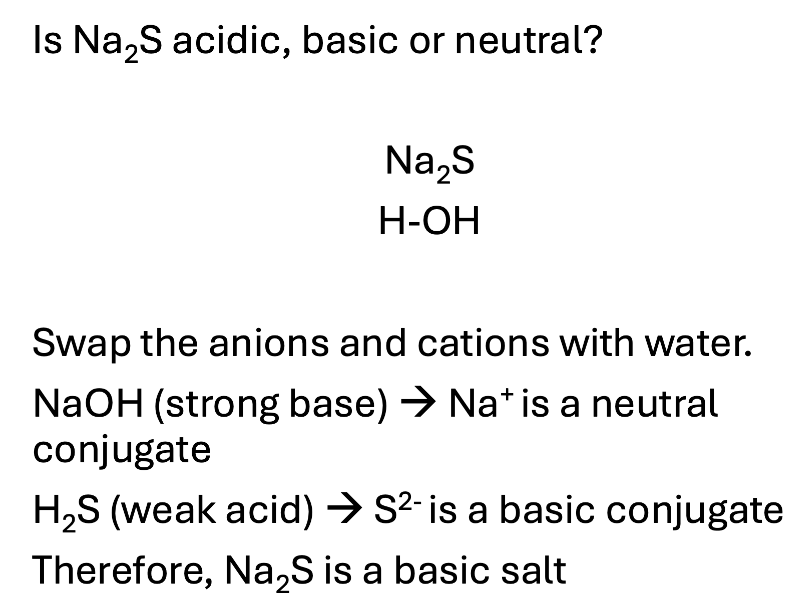
Given a salt, what happens if it contains both an acidic and basic ion?
Initially, carry out the same process.
If nothing strong is created on either ion, then you must compare kA vs kB
Whatever one is bigger (kA vs kB) determines which overpowers the salt and makes it more acidic or basic.

END OF ACIDS AND BASES + RECAP!
END OF ACIDS AND BASES + RECAP!

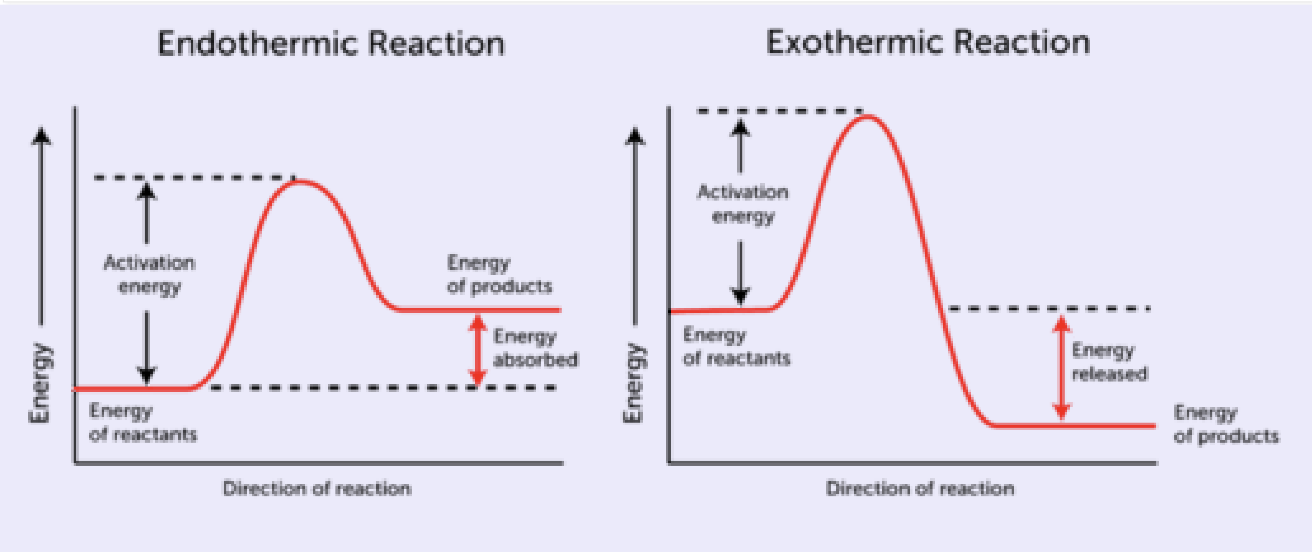
What is enthalpy: definition, symbols, and general concept. Reaction coordinate (graph)
Concerns the amount of heat released or absorbed during a chemical reaction at constant pressure
Represented as ΔH
Reactions that release heat
Exothermic
Get hot (the surrounding temperature goes up)
ΔH= negative #
Reactions that absorb heat
Endothermic
Get cold (surrounding temperature goes down)
ΔH = positive #
(Δ is always final - initial)
What is the kinetic energy equation? What about a joule and why they are important?
Kinetic Energy – KE = ½ mv2
m = mass (kg)
V = velocity (m/s)
Joule = 𝑘𝑔∗𝑚2 / 𝑠2
4.18 J = 1 calorie = amount of heat needed to warm 1.00 g H2O up by 1.00oC
Temperature v. Heat v. Enthalpy and their units
Temperature (T) is a measure of the average kinetic energy of the molecules in a sample
Units of Celsius / Kelvin / Fahrenheit / Rankine
Heat (q): A form of energy often studied in chemistry. Heat energy can cause an increase in temperature. q = mCΔT
Units of Joules (J), kilojoules (kJ), calories (c), or Calories (C)
Enthalpy (H): A state function describing the sum of the internal energy of a system
Units kJ/mol
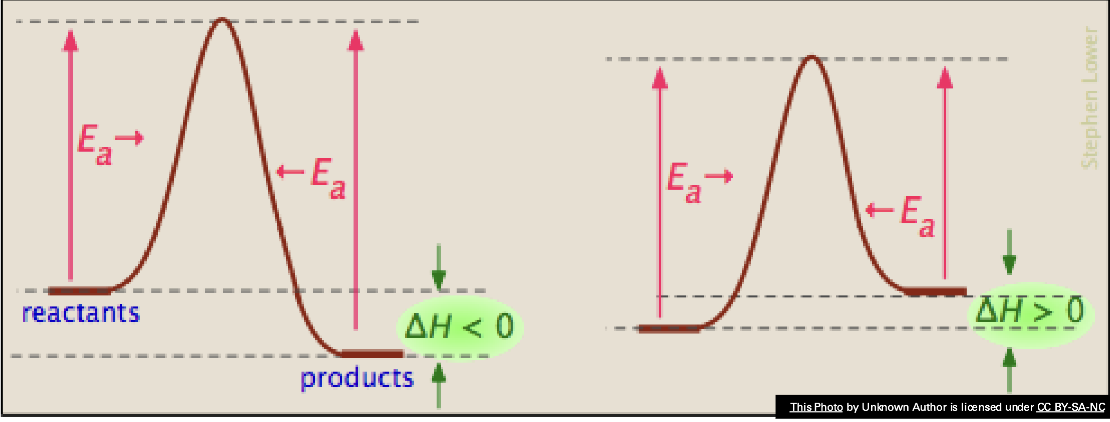
Enthalpy is a ____ function. How do you calculate these? What are the thermodynamically favorable (spontaneous) conditions?
State
Matter in its elemental state has a ΔHof of zero by definition (Fe (s), O2 (g), I2 (s))
The ΔHof of a compound can be calculated based on how much heat is gained or lost making that compound from its respective elements
The compound data will be provided to you in a data table
Enthalpy is an extensive property so you must remember to multiply by the number of moles
ΔH = Hfinal – Hinitial = ΣΔHof products - ΣΔHof reactants
Exothermic reactions (ΔH = ⊝) are typically more favorable to
happen than endothermic reactions. Releasing heat energy is a very
helpful contribution to causing a reaction to happen.
What is Entropy: definition, symbols, what does zero mean, general concept, what conditions favored?
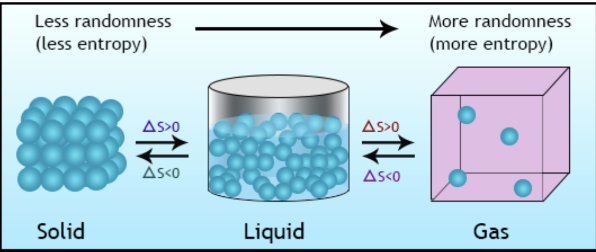
Entropy (ΔS) is a measure of randomness or disorder of a system – Units J/(mol*K)
Zero entropy is defined as being a perfect, solid crystal at absolute zero (0 Kelvin)
All real-world substances have S > 0 because they all contain some level of imperfection/randomness
A large entropy value is associated with more randomness
ΔS can be positive or negative!
+ ΔS means it becomes more random
– ΔS means it becomes more ordered
+ΔS is more likely to be a spontaneous process – The universe likes disorder (2nd Law of Thermodynamics)
What are the three popular trends in entropy (states, form, #moles)?
How is decomp and synthesis reactions impacted?
What kind of function is this, and what does it imply?
SGases > SLiquids > Ssolids
SDissolved > Ssolid
S2 moles > S1 mole
Decomposition reactions AB → A + B typically have an increase in entropy
Synthesis reactions A + B → AB typically have a decrease in entropy
Since Entropy (S) is a state function, it can be calculated using the following:
ΔSo = ΔSoproducts - ΔSoreactants
Applying Entropy:
Combustion of Butane
2 C4H10(g) + 13 O2(g) → 8 CO2(g) + 10 H2O(g)
15 moles of gas → 18 moles of gas
ΔS = + as there was more moles and hence more randomness.
What is Gibbs Free Energy: definition, symbols, general concept, favored state, and spontaneity.
Measure of the overall assessment of a process’s favorability/spontaneity to happen
ΔG (Units: kJ/mol)
Specifically, it is a measure of whether something will happen without added energy (spontaneous), then ΔG will be negative
Exergonic, this is the favored state
If something will not happen (non-spontaneous), then ΔG will be positive – Could still happen if outside energy is added
Endergonic, this is the unfavorable state
If a process is spontaneous, the reverse process is not spontaneous
What kind of functions are H,S, and G? What does this imply and mean for them?
H, S, and G are all state functions
Known as a thermodynamic property of a system that depends only on its current state (initial and final points), not on the path taken to reach that state
Therefore, ΔH, ΔS, and ΔG can be calculated using the ΔHfo, ΔSo, or ΔGfo data from a table.
Hence, ΔHorxn = ΣΔHof products – ΣΔHof reactions, and etc for each.
ΔH, ΔS, and temperature are the components of Gibbs Free Energy according to what equation?
ΔG = ΔH = TΔS
This is known as the Gibbs free energy equation
T in this equation is the temperature in Kelvin. Δ is final – initial.
What are more likely to be associated with a spontaneous and favorable reaction for the Gibbs free energy equation?
A negative ΔH (exothermic)
A positive ΔS (more randomness)
A negative ΔG (spontaneous/favorable)
If ΔH is negative and ΔS is positive, what will the reaction be?
If ΔH is positive and ΔS is negative, what will the reaction be?
If ΔH is negative and ΔS is negative, what will the reaction be?
If ΔH is positive and ΔS is positive, what will the reaction be?
If ΔH is negative and ΔS is positive
ALWAYS SPONTANEOUS
If ΔH is positive and ΔS is negative
NEVER SPONTANEOUS
If ΔH is negative and ΔS is negative
Spontaneous at lower temperatures
If ΔH is positive and ΔS is positive, what will the reaction be?
Spontaneous at higher temperatures
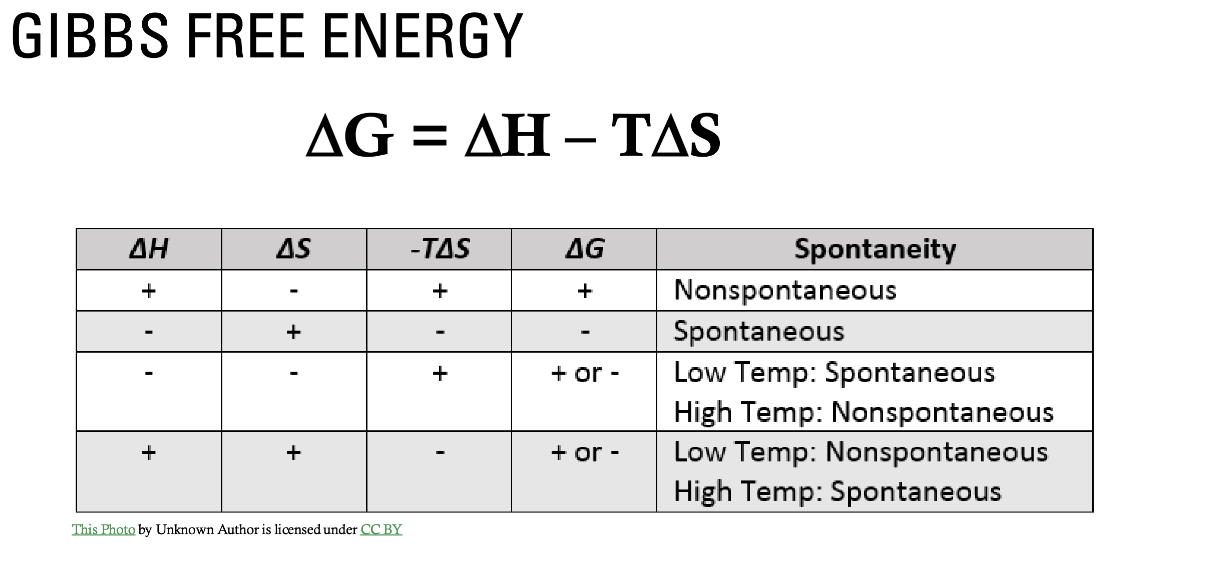
Before S can be plugged into the Gibbs free energy equation, what must happen?
S has units of Joules / mole-K, so it must be converted into kilojoules in order to plug into the equation
What is unique to Gibbs free energy when the reaction is at equilibrium? How can it be simplified?
If the reaction is at equilibrium (a change of physical state like liquid to a gas), then ΔG = 0
The equation can be simplified to TΔS = ΔH.
How to encounter a Gibbs free energy equation question when it asks, “At what temperature does a process become spontaneous?” What does this imply?
This implies that at a certain temperature, you will cross over from a positive to a negative ΔG.
Set ΔG = 0 and solve for T.
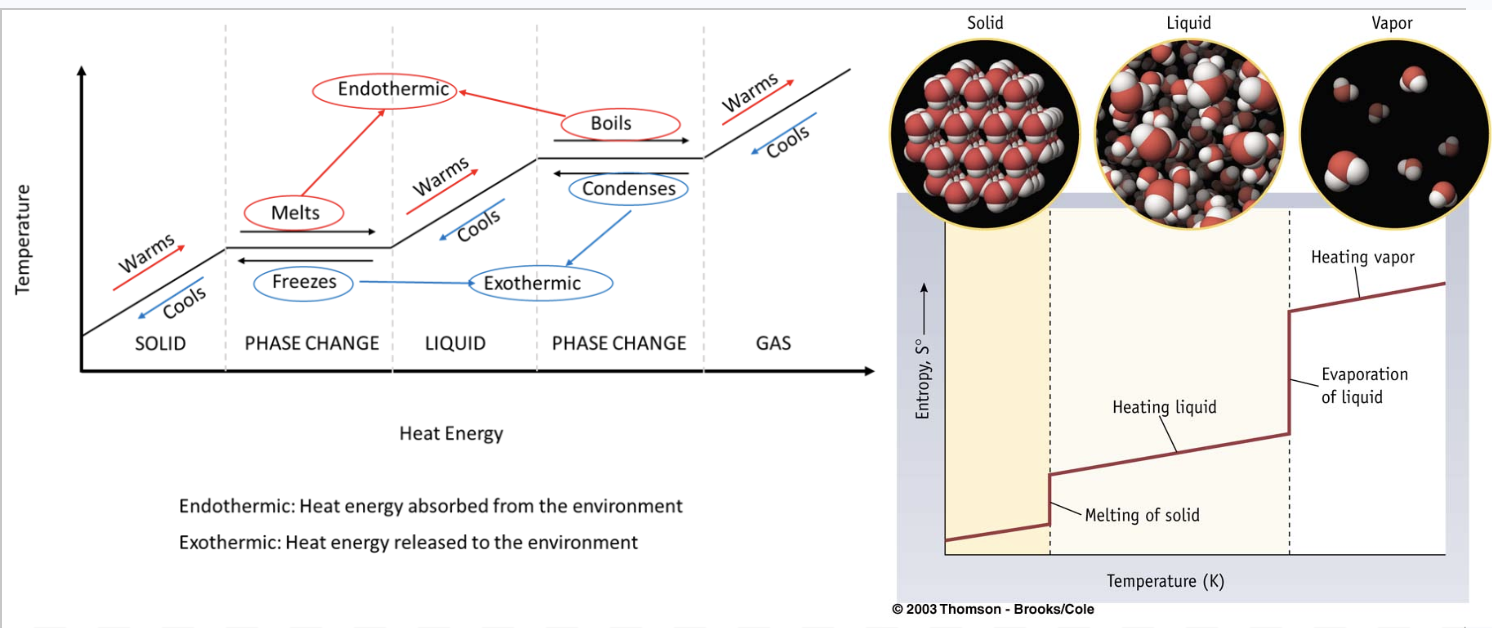
Heating and cooling curves: what phase changes are exo/endothermic? How does entropy change along with the phase changes/curves?
Heating and cooling curves illustrate the energy changes that occur during phase transitions.
Endothermic processes, which absorb heat, occur on heating curves (solid → liquid → gas)
Exothermic processes, which release heat, occur on cooling curves (gas → liquid → solid).
Entropy increases as a substance becomes more disordered, specifically when changing from solid → liquid → gas.
How can a change in Gibbs free energy (ΔG) be related back to the equilibrium constant (keq), both qualitatively and quantitatively (favorable vs not)?
Quantitatively
ΔG = − RTln(K)
R = Universal Gas Constant = 8.31 J/(mole*K) – Watch your units!!
T is temperature in Kelvin
K is the equilibrium constant
Qualitatively
If you plug in a value for K >1, the value of ΔG will necessarily be negative (favorable)
If you plug in a value for K <1, the value of ΔG will necessarily be positive (not favorable)
This should make sense! If your equilibrium favors products, then the reaction will proceed!

What is the math to undo the natural log? When would you need to do this?
Undo the natural log with (k = eln(k))
This applies to Gibbs free energy when you are calculating the equilibrium constant (K) from the standard Gibbs free energy change (ΔG).
Because ΔG is related to the natural log of the equilibrium constant via the equation ΔG = -RTln(k), you must use the inverse natural log function (exponential function ex) to isolate K and solve for it.

What is the equation used to calculate ΔG under nonstandard conditions? Why is this used, and how is it solved for?
What happens when the reaction reaches equilibrium?
ΔG = ΔGo + RTln(Q)
ΔG: Standard Gibbs free energy change (at 1atm,1 M, 298 K).
R: Ideal gas constant (8.314 J/mol x k)
T: Temperature in Kelvin
Q: Reaction quotient, calculated as [products]/[reactants], (or partial pressures), at nonstandard conditions
The value of Q corrects for the deviation from the standard conditions.
Q(reaction Quotient) is the same value as from the Equilibrium chapter and is solved the same way as Keq, but under the nonstandard conditions.
When the reaction reaches equilibrium, ΔG = 0 and Q = K, which leads to the related formula: ΔG = − RTln(K)
![<ul><li><p>ΔG = ΔG<sup>o</sup> + RTln(Q)</p><ul><li><p>ΔG: Standard Gibbs free energy change (at 1atm,1 M, 298 K).</p></li><li><p>R: Ideal gas constant (8.314 J/mol x k)</p></li><li><p>T: Temperature in Kelvin</p></li><li><p>Q: Reaction quotient, calculated as [products]/[reactants], (or partial pressures), at nonstandard conditions</p></li></ul></li></ul><ul><li><p>The value of Q corrects for the deviation from the standard conditions.</p><ul><li><p>Q(reaction Quotient) is the same value as from the Equilibrium chapter and is solved the same way as Keq, but under the nonstandard conditions.</p></li></ul></li></ul><ul><li><p>When the reaction reaches equilibrium, ΔG = 0 and Q = K, which leads to the related formula: ΔG = − RTln(K)</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/d403d78b-f98d-42b4-89f8-115e4012d5b3.png)
A student calculates the ΔG of a chemical reaction to be negative, and he knows this means the reaction is therefore thermodynamically favored to occur. He sets up the reaction, and the next day, he evaluates the mixture to find that it is all starting material. What’s the deal?
The reaction is under kinetic control!!
Even as a thermodynamically favored reaction, the activation energy is too high, even if the ΔG is negative!
The student needs to add energy (e.g., heat the mixture) or add a catalyst to lower the activation energy and allow the reaction to proceed faster.
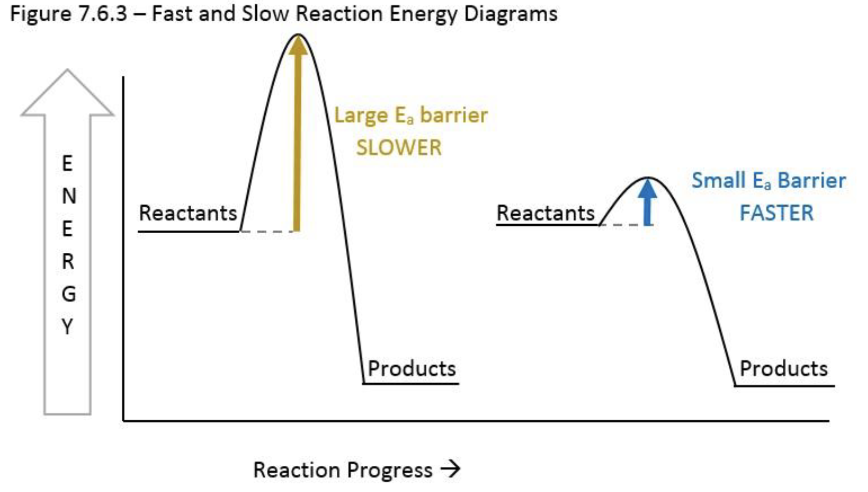
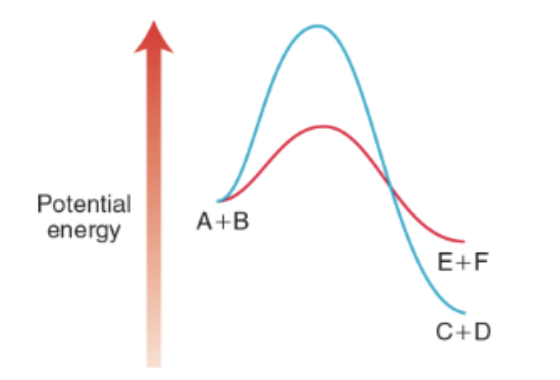
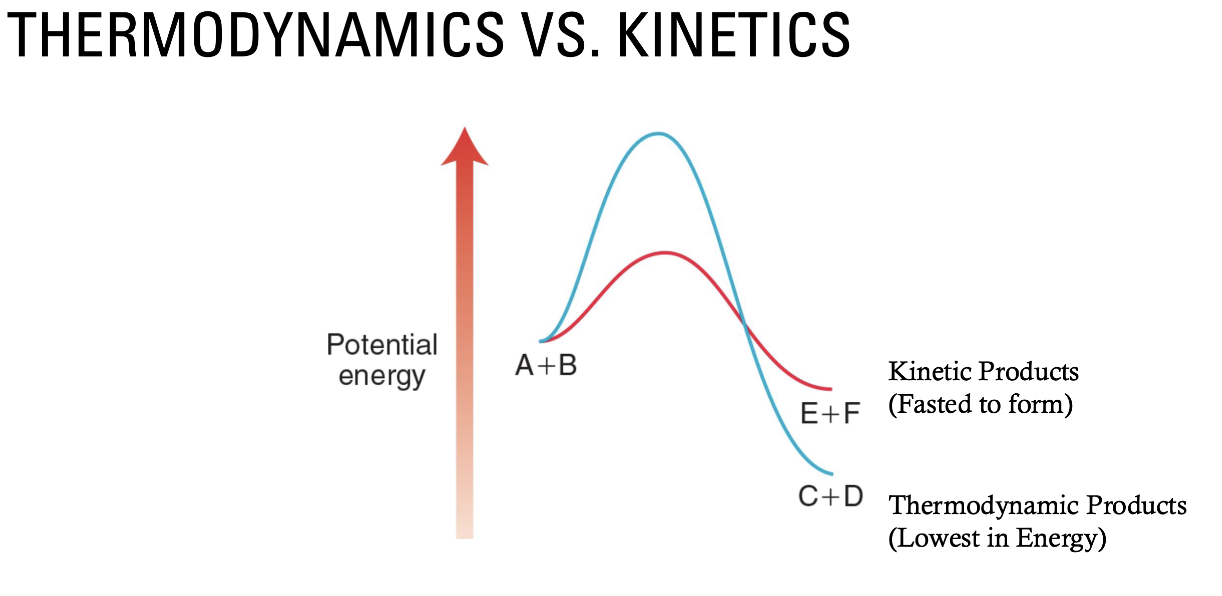
Thermodynamics vs kinetics, describe this graph. What about reactions that are not favored?
In the diagram below, the blue line creates the thermodynamic products (lowest in energy), and the red line is the formation of the kinetic products (lower activation energy).
This can be controlled with Temperature.
Higher T will push the reaction to the thermodynamic products because it will help molecules get over the activation energy.
Reactions that are not favored can still occur if energy is applied to them.
GIBBS FREE ENERGY RECAP
Exothermic reactions that become more disordered are always going to be thermodynamically favored (negative H and positive S)
Temperature influences the entropy portion of the Gibbs Free Energy function, so high temperature will weigh the entropy part of the equation
If a process is favorable (spontaneous), the reverse process will be unfavorable (nonspontaneous)
Unfavorable processes (+G) can still occur if energy is provided to do so
If a process has a – G and no or slow-forming product is observed, the process is likely under kinetic control, meaning that even though it is thermodynamically favored, the activation energy is too high
END OF TEST 2!