Lecture 1: Ideal gas law, chemical equilibrium
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
what determines the state of a gas?
pressure
volume
temperature
number of particels (n)
what is one celcius in Kelvin?
what is one Kelvin in celcius
Beispiel 25 Celcius in Kelvin
K= °C+ 273,15
°C = K - 273,15
Beispiel: Wenn Sie 25 Grad Celsius haben, beträgt die Umrechnung in Kelvin 25 + 273,15 = 298,15 K.
atm in Pa
Pa in atm
atm*101325
Pa/101325
Torr in Atm
Atm in Torr
Torr= Atm * 760
Atm = Torr/760
Ammonia
Chlorine
Hydrogen
NH3
Cl2
H2
H2S
CH4
NO2
Hydrogen sulfide
Methane
Nitrogen Dioxide
postulates of the kinetic theory of gases
most volume is empty space
collisions account for the pressure of the gas
perfectly elastic =) no kinetic energy lost. Energy remains stable
No attraction or repulsion between molecules unless collision
Standart Temperature and Pressure (IUPAC and DIN)
Standart Ambiente Temperature and Pressure
0 °C (273,15) and 10^5 Pa
Boyles law
the volume is inversely proportional to the presure
v= 1/p and vice versa
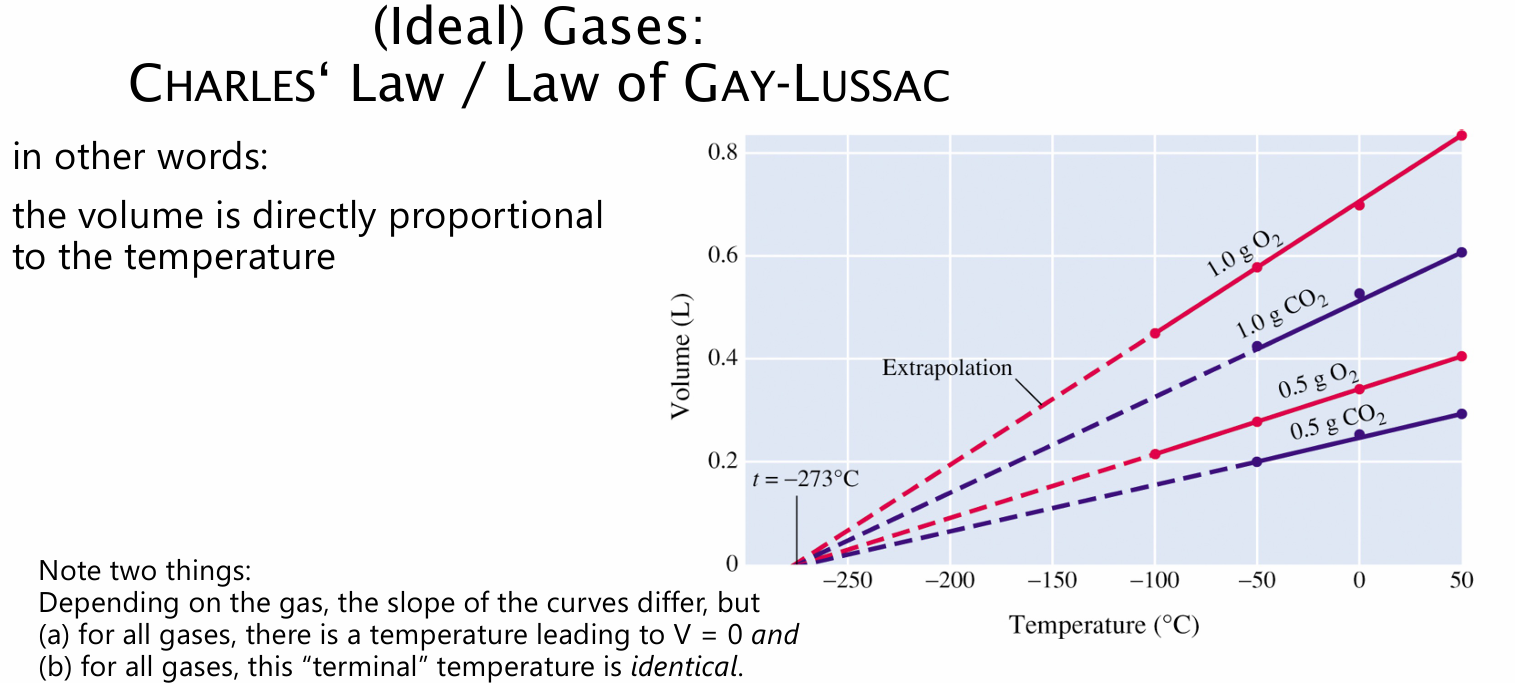
Gay-Lussac law
Avogadros law
equal volumes of any two gases at the same temperature/pressure contain the same number of particles
the molar gas volume
Vm
The volume occupied by one mole of an ideal gas
At 0 degrees = 22.4 L/mol
at 20 degrees = 24.0 L/mol
at 25 degrees = 24.8 L/mol
how do you calculate V out of Vm
V= Vm times n
Increasing the number of particles leads to increased _____ via ________
Increasing the number of particles leads to increased pressure/volume via fully elastic collisions
BOYLE'S law CHARLES' law AVOGADRO’s law short proportions
V ~ 1/p p *v = constant
V ~ p and t V/t = constant
V~ n
The Ideal Gas Law
p · V = n · R · T
molar gas constant
R = 8,3145 J/Kmol
R= 0,0820 Latm/Kmol
pressure times volume =
energy in Joule
Pa m³ = N/m² * m³ = Nm = J
Real gases
consist of atoms/molecules with intermolecular attraction/repulsion
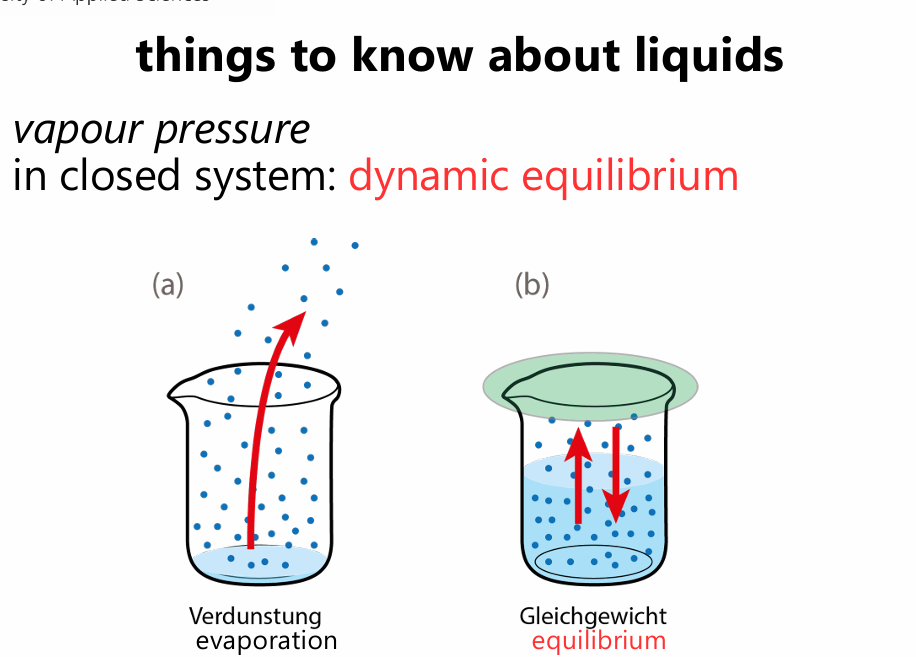
vapour pressure
Osmosis vs diffusion
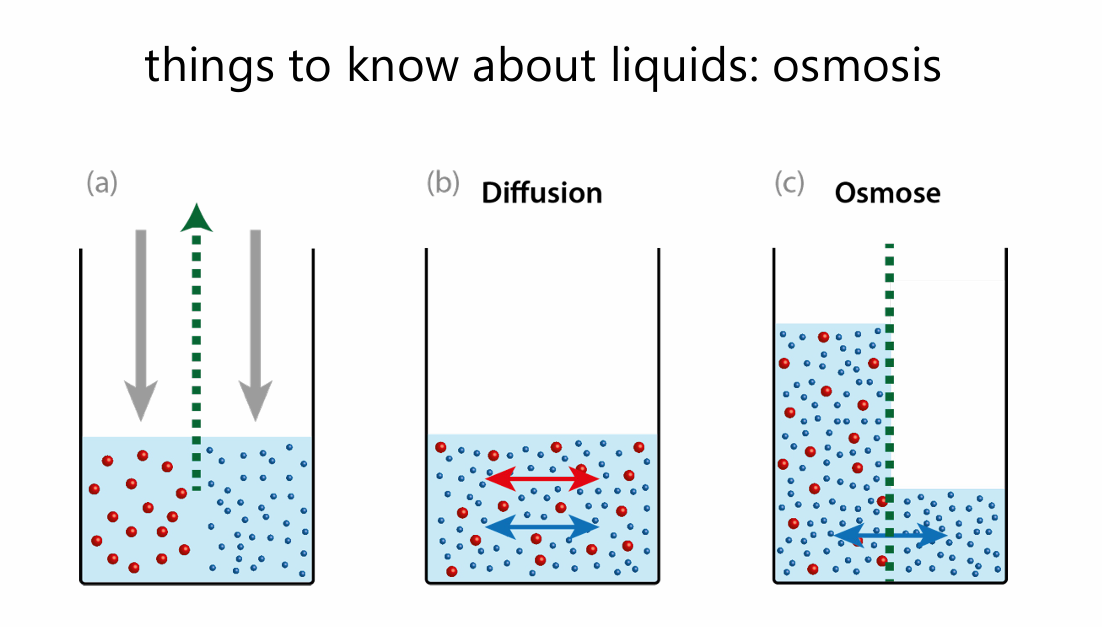
Osmosis – Osmosis is the movement of solvent particles (usually water) across a semipermeable membrane from a dilute solution to a concentrated solution. The solvent dilutes the concentrated solution until concentration is equalized on both sides of the membrane.
Diffusion – Diffusion is the movement of solvent and solute particles from an area of higher concentration to lower concentration. At equilibrium, the net effect is a homogeneous concentration throughout the medium.
what is the differnce between a saturated and an unsaturated solution?
In an unsaturated solution part of the solute remains undissolved
what is the product of sedimentation?
precipitate
when is equilibrium?
When the net change of the products and reactants is zero, the reaction has reached equilibrium
reaction rate
change in amount of substance overime
amount of products formed or reactants used
the law of mass action
K
concentration at equilibirum not starting conditions
the larger K the___
more the reaction lies on the products side
avogadros number
6.022 × 10²³
activity
Applies whenever different phases are in contact with one another:
• a solid and a liquid
• two not miscible liquids (oil/water)
• etc.
The substance in contact with the phase in question always (!) has an activity of a(substance) = 1. (no unit!)
when does saturation occur?
when IP bigger or the same as Ksp
what do we use for Ksp?
pKsp = -lgKsp
Insoluble ionic compounds
CO3²- Carbonates
PO4³- Phosphates
S²- Sulfide
OH^- Hydroxide