Chapter 1: Atoms, Elements, Compounds and Chemical Equations
1.1-Atoms
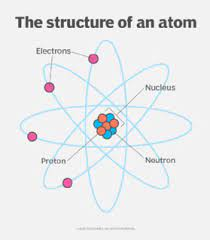
Atoms contain protons, neutrons and electrons
Atoms have a radius of about 10 nanometres(1x10(-9)). There are a few different modern models of the atom.
The nucleus
- It’s in the middle of the atom
It contains protons and neutrons
- The nucleus has a radius of around 1x10(-14) which is around 1/10,000 the radius of an atom
- It has a positive charge because of the protons
- Almost the whole mass of the atom is concentrated in the nucleus
The electrons
- Move around the nucleus in electron shells
- They’re negatively charges and tiny, but they cover a lot of space
- Electrons have virtually no mass
Particles, relative mass and charge
- Proton:1 and +1
- Neutron:1 and 0
- Electron:Very small and -1
Number of protons equals number of electrons
- Atoms are neutral-they have no charge overall(unlike ions).
- This is because they have the same number of protons as electrons
- The charge on the electrons is the same size as the charge on the protons, but opposite-so the charges cancel out
- In an ion, the number of protons doesn’t equal the number of electrons. * This means it has an overall charge. For example, an ion with a 2- charge, has two more electrons than protons
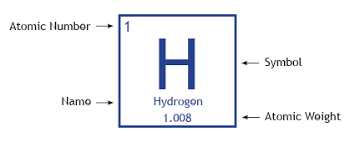
Atomic Numbers and Mass Number describe an Atom
- The nuclear symbol of an atom tells you its atomic(proton) number and mass number
- The atomic number tells you how many protons there are
- The mass number tells you the total number of protons and neutrons in the atom
- To get the number of neutrons, just subtract the atomic number from the mass number
1.2-Elements
An element is a substance made up of atoms that all have the same number of protons in their nucleus
Elements consist of atoms with the same atomic number
- Atoms can have different numbers of protons, neutrons and electrons.
- It’s the number of protons in the nucleus that decides what type of atom it is * For example, an atom with one proton in its nucleus is hydrogen and an atom with two protons is helium. * There are about 100 different elements
- So all the atoms of a particular element have the same number of protons and different elements have atoms with different numbers of protons
Atoms can be represented by symbols
- Atoms of each element can be represented by a one or two letter symbol-it’s a type of shorthand that saves you the bother of having to write the full name of the element. * e.g. C=carbon, O-oxygen, Mg=magnesium * or e.g. Na=sodium, Fe=iron, Pb-lead
\
Isotopes are the same except for extra neutrons
- Isotopes are different forms of the same element, which have the same number of protons but a different number of neutrons
- So isotopes have the same atomic number but different mass numbers
- A very popular example of a pair of isotopes are carbon 12 and carbon 13
- Carbon 12: * 6 protons * 6 electrons * 6 neutrons
- Carbon 13 * 6 protons * 6 electrons * 7 neutrons
- Because many elements can exist as a number of different isotopes, relative atomic mass is used instead of mass number when referring to the element as a whole. * This is an average mass taking into account the different masses and abundances of all the isotopes that make up the element
- You can use a formula to work out the relative atomic mass: * Relative atomic mass = sum of(isotopes abundance x isotope mass number) / sum of abundance of all the isotopes
\
1.3-Compounds
Atoms can join together to make compounds
- When elements react, atoms combine with other atoms to form compounds * Compounds: substances formed form two or more elements * The atoms of each are in fixed properties throughout the compound and they’re held together by chemical bounds
- Making bonds involves atoms giving away, taking or sharing electrons.
- Only the electrons are involved-the nuclei of the atoms aren’t affected at all when a bond is made
- It’s usually difficult to separate the original element of a compound out again * A chemical reaction is needed to do this
- A compound which is formed from a metal and a non-metal consists of ions. * The metal atoms lose electrons to form positive ions and the non-metal atoms gain electrons to form negative ions. * The opposite charges(positive and negative) of the ions mean that they’re strongly attracted to each other. * This is called ionic bonding. * Examples of compounds which are bonded ionically include sodium chloride, magnesium oxide and calcium oxide
- A compound formed from non-metals consists of molecules.
- Each other shares an electron with another atom-this is called covalent bonding. * Examples of compounds that are bonded covalently include hydrogen chloride gas. carbon monoxide and water
- The properties of a compound are usually totally different from the properties of the original elements. * For example, if iron and sulfur react, the compound formed is a dull grey solid lump, and doesn’t behave anything like either iron or sulfur
A formula shows what atoms are in a compound
- Just as elements can be represented by symbols, compounds can be represented by formulas.
- The formulas are made up of elemental symbols in the same proportions that the elements can be found in the compound
1.4-Chemical Equations
Chemical changes are shown using chemical equations
- One way to show a chemical reaction is to write a word equation.
- It’s not as quick as using chemical symbols and you can’t tell straight away what’s happened to each of the atoms, but it’s dead easy. * Here’s an example-you’re told that methane burns in oxygen giving carbon dioxide and water * Methane + Oxygen - Carbon Dioxide + Water * The molecules on the left-hand side of the equation are called reactants(because they react with each other) * The molecules on the right-hand side are called the products(because they’ve been produced from the reactants
Symbol equations show the atoms on both sides
- Chemical changes can be shown in a kind of shorthand using symbol equations.
- Symbol equations just show the symbols or formulas of the reactants and products * Magnesium + Oxygen - Magnesium Oxide * 2Mg + 02 - 2Mg0
Symbol equations need to be balanced
- There must always be the same number of atoms on both sides-they can’t just disappear
- You balance the equation by putting numbers in front of the formulas where needed. * Take this equation for reacting sulfuric acid with sodium hydroxide: * H2S04 + Na0H - Na2S04 + H20
- The formulas are all correct but the numbers of some atoms don’t match up on both sides
- You can’t change formulas like H2SO4 to H2SO5. You can only put numbers in front of them.
- The more you practise, the quicker you get, but all you do is this: * Find an element that doesn’t balance and pencil in a number to try and sort it out * See where it gets you. It may create another imbalance, but if so, pencil in another number and see where that gets you * Carry on chasing unbalanced elements and it’ll sort itself out pretty quickly
\