Chapter 13 - Lipids
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
storage of energy
insulation from environment
water repellant
buoyancy control in marine animals
membrane structure
cofactors for enzymes
signaling molecules
pigments
antioxidants
9 common functions of lipids
they are reduced compounds, so they have lots of available energy and they are hydrophobic, so they are good at packing
why are lipids good for storage of energy
they have low thermal conductivity, high heat capacity (can absorb heat), and mechanical protection by absorbing shocks
how are lipids able to provide insulation from the environment
vitamin K (blood clot formation) and coenzyme Q (ATP synthesis in the mitochondria)
2 cofactors for enzymes that are lipids
paracrine hormones (act locally), steroid hormones (act body-wide), growth hormones, and vitamins A and D (hormone precursors)
4 signaling molecules that are lipids
vitamin E
which vitamin is a lipid and antioxidant
structure and function
what are lipids classified based on
lipids that do not contain fatty acids vs lipids that do contain fatty acids (complex lipids)
how are lipids classified based on structure
storage lipids vs membrane lipids
how are complex lipids classified based on function
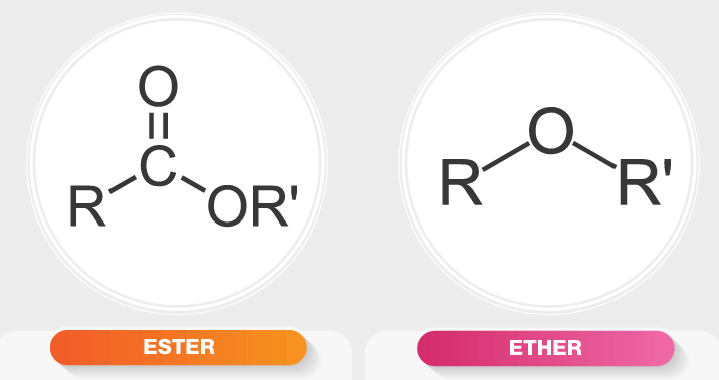
draw ester vs ether
carboxylic acids with hydrocarbon chains containing between 4 to 36 carbons
what are fatty acids (composition wise)
most have an even number of carbons and are unbranched
2 characteristics of most natural fatty acids
saturated have no double bonds, monounsaturated have 1 double bond, and polyunsaturated have more than 1 double bond
saturated vs monounsaturated vs polyunsaturated fatty acids
essential nutrients (humans need them but cannot synthesize them)
what are omega-3 fatty acids considered
ALA, DHA, and EPA
3 examples of omega-3 fatty acids
solubility decreases
how does solubility change as fatty acid length increases
melting point decreases
how does melting point change as fatty acid length decreases
melting point decreases
how does melting point of fatty change when double bonds increases
cis-configuration which kinks the chain
what configuration are the double bonds in natural unsaturated fatty acid commonly in
saturated fatty acids pack in a fairly orderly way while unsaturated cis fatty acids pack less orderly due to the kink
saturated vs unsaturated cis fatty acid packing
unsaturated cis fatty acids because it takes less thermal energy to disrupt their disordered packing
do saturated fatty acids or unsaturated cis fatty acids have lower melting point
formed by partial hydrogenation of unsaturated fatty acids and change from liquid to solid form
how are trans fatty acids formed
trans fatty acids can pack more regularly and have higher melting points
trans fatty acid packing and melting point compared to cis fatty acids
cardiovascular disease
what does the consumption of trans fatty acids increase risk of
triacylglycerols
what form are majority of fatty acids in biological systems found in
liquids ones are called oils and solid ones are called fats
what is a liquid vs solid triacylglycerol called
triacylglycerols
what is the primary storage form of lipids
triacylglycerols are less soluble in water due to the lack of charged carboxylate group
solubility of triacylglycerols compared to fatty acids
triacyclglycerols are less dense than water (float in water)
density of triacylglycerols compared to water
fatty acids carry more energy per carbon because they are more reduced and they carry less water per gram because they are nonpolar
advantage of fats over polysaccharides
glucose and glycogen are for short-term energy needs, quick delivery
fats are for long-term energy needs, good storage, slow delivery
energy needs fatty acids vs polysaccharides provide
esters of long-chain saturated and unsaturated fatty acids with long-chain alcohols
what are waxes (composition)
they are insoluble and have high melting points
characteristics of waxes
storage of metabolic fuel in plankton
protection and pliability for hair and skin in vertebrates
waterproofing of feathers in birds
protection from evaporation in tropical plants and ivy
used by people in lotions, ointments, and polishes
5 functions of waxes
modifying a different backbone
changing the fatty acids
modifying the head groups
how can structural lipids in membranes be diversified
the properties of head groups
what determines the surface properties of membranes
different organisms and different tissues
what is composed of different membrane lipid head group compositions
glycerophospholipids
primary constituents of cell membranes
two fatty acids form ester linkages with the first and second hydroxyl groups of L-glycerol-3-phosphate
how is glycerophospholipid formed
at physiological pH
when is head group of glycerophospholipid charged
glycerol with highly polar phosphate group, saturates fatty acid, unsaturated fatty acid connected to C2, and head group alcohol
general structure of glycerophospholipids
phosphatidyl-”name of head-group alcohol”
how is type of glycerophospholipid named
phosphatidic acid
what is the parent compound glycerophospholipid
phosphatidylcholine
glycerophospholipid found in eukaryotic cell membranes
vinyl ether analog of phosphatidylethanolamine → vertebrate heart tissue, some protozoa and anaerobic bacteria
what is plasmalogen and where is it found
aliphatic ether analog of phosphatidylcholine
what is platelets-activating factor