Lecture 10: coordination compounds
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
coordination compounds aka complexes
a central metal atom is surrounded by nonmetal atoms or groups of atoms, called ligands
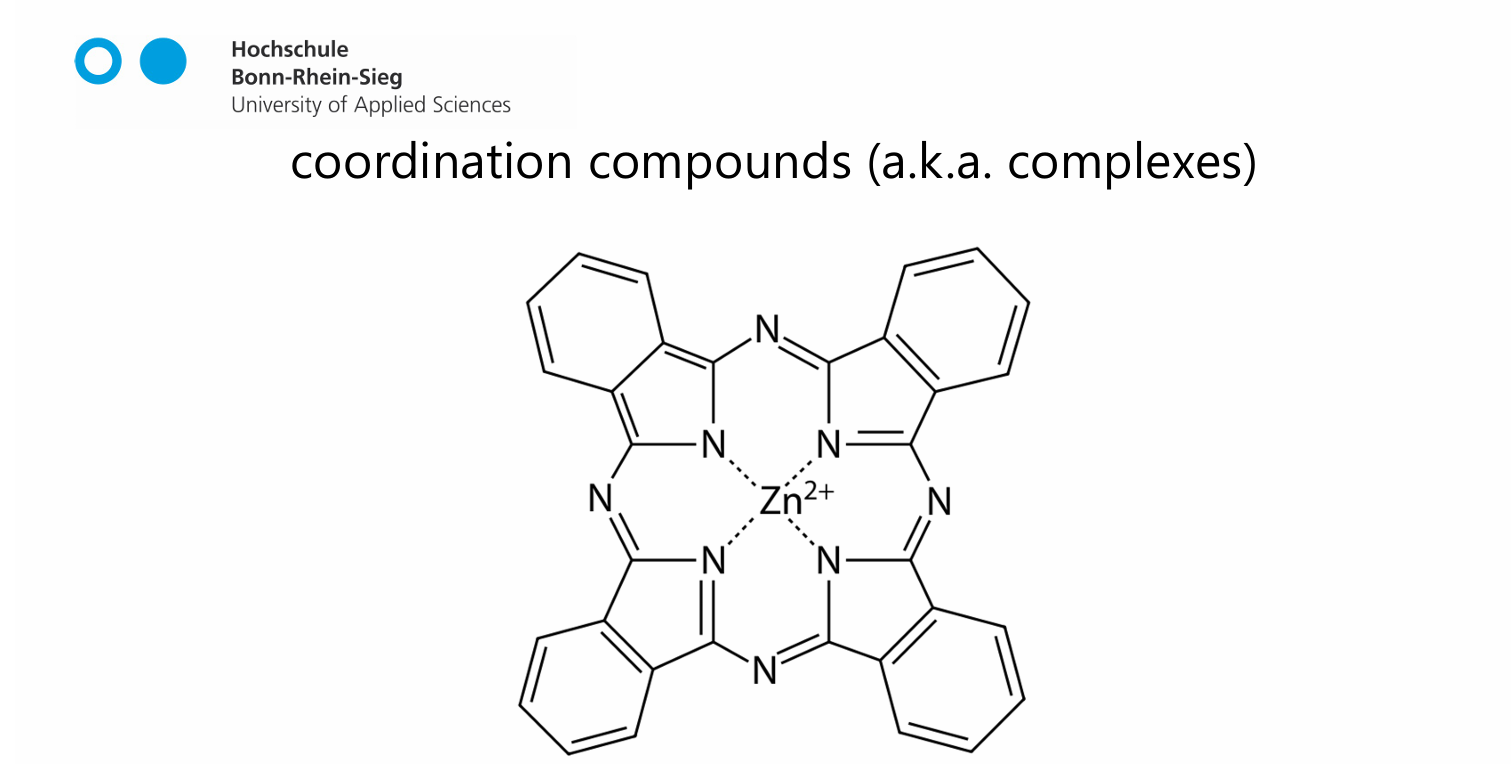
complex ions
the combination of a transition metal cation with a ligand molecule or ion
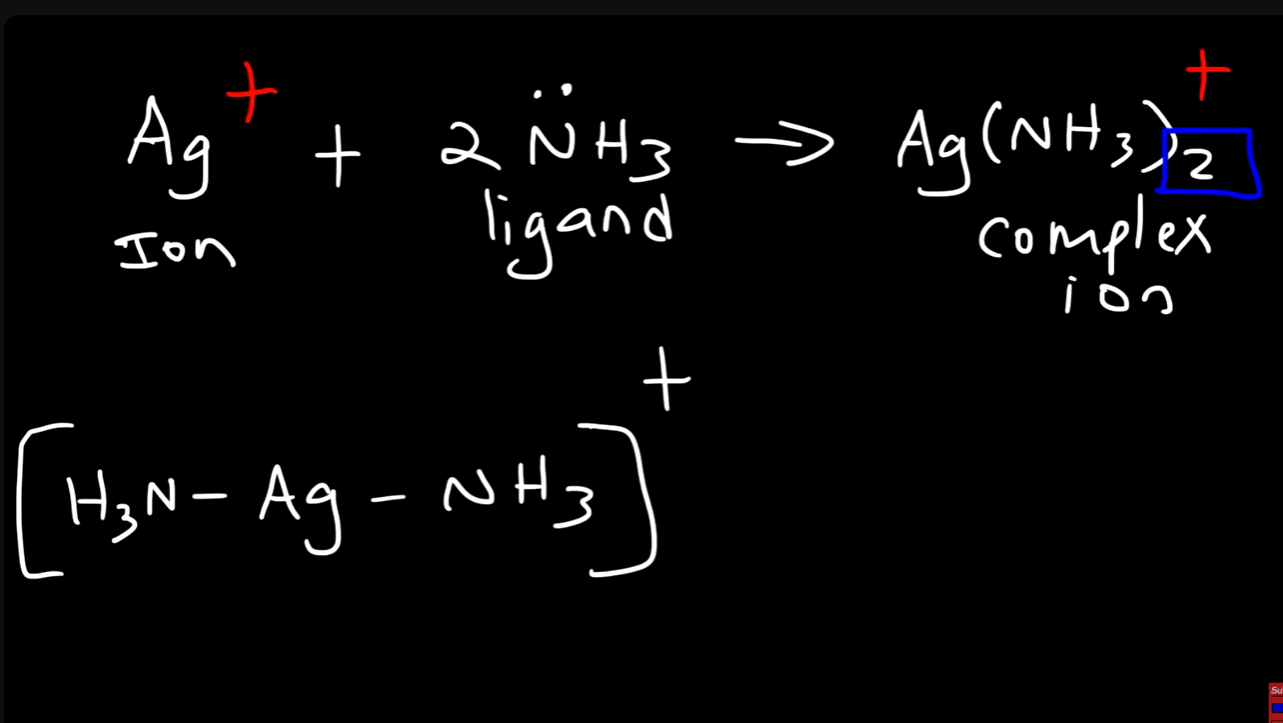
Lewis acid and base
acid: electron acceptor
base: electron donor
reaction of ammonia with water
NH3 + H2O → NH4+ + OH-
NH₃ donates its electron pair → Lewis base
H⁺ accepts it → Lewis acid NH3+H+→NH4+
The new N–H bond is a:
coordinate covalent bond
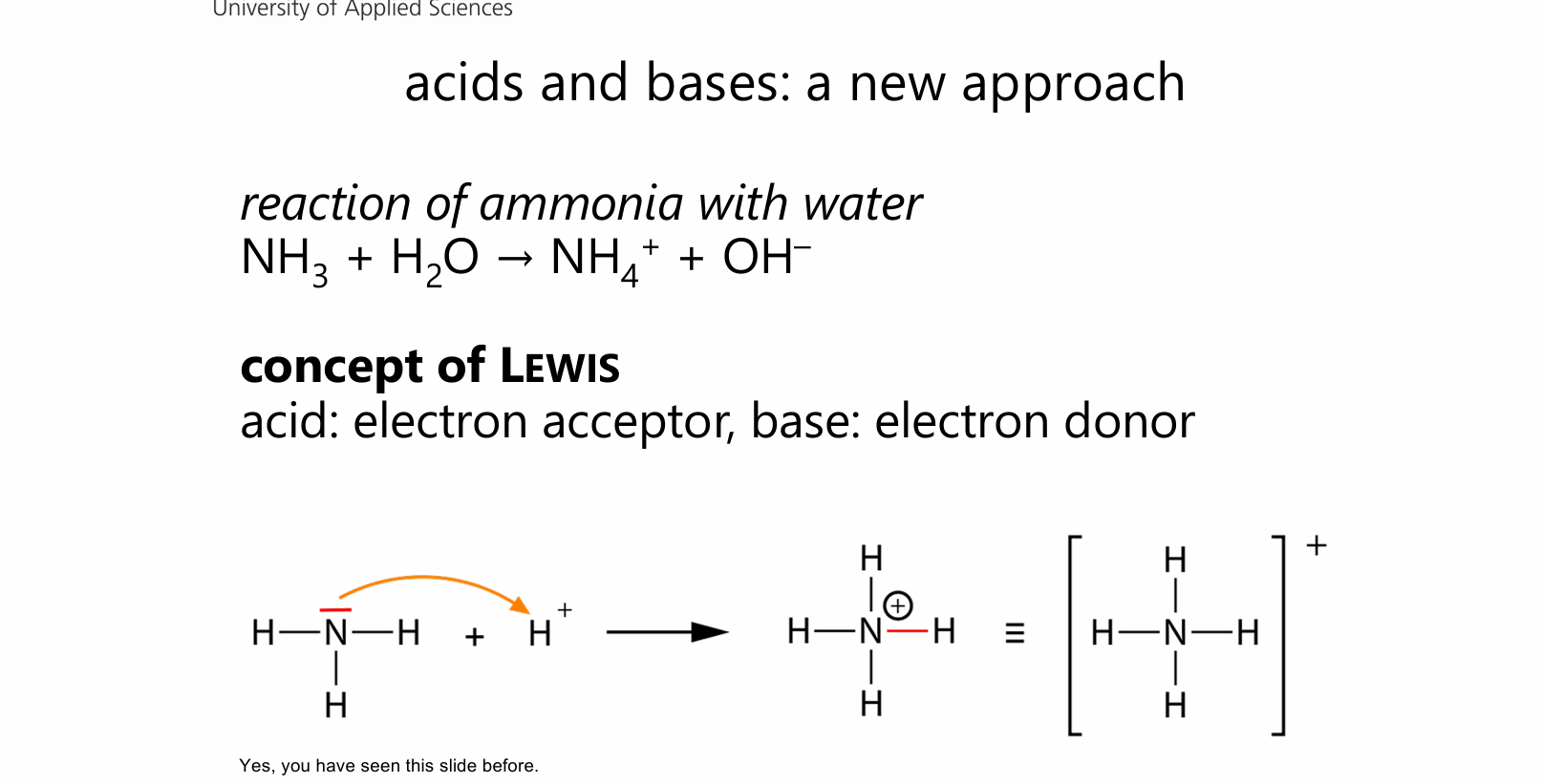
the coordinate covalent bond
a covalent bond, in which the two electrons forming the bond derive from the same atom.
(unlike e.g. H• + •H → H–H, H2)
Once the bond is formed, it cannot be distinguished from a «conventional» covalent bond
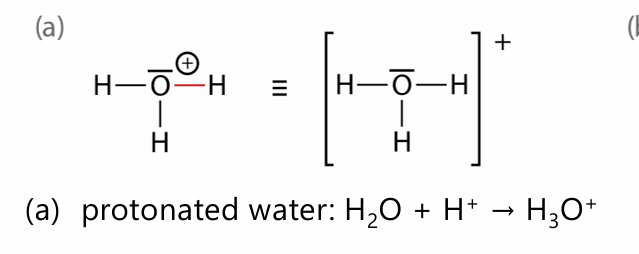
protonated water
The atom, molecule or ion donating the electron pair is called the ____, often abbreviated _____
ligand, L
The atom or ion accepting the electron pair is called the __________
The atom or ion accepting the electron pair is called the coordination centre
Since the coordination centre in a complex is in almost all cases a metal atom or ion, it is generally abbreviated ___
Thus L| -) M -)
M
[M-L]

An atom, molecule, or ion can only function as a ligand (and as a LEWIS base) if it has at least one ______
lone pair

NC: All anionic complexes (i.e. complex anions) get the ending
-ate
Central particle of anions is named based on the origin of the element's symbol:
with iron:
copper:
tin :
Mercury:
Ag:
iron: ferrate (Fe-ferrum)
copper: cuprate (Cu-cuprum)
tin; stannate (Sn-stannum)
Exception: HG= hydragyrum, but an anionic complex containing mercury is called mercurate.
Silver: argentate
Naming anionic complexes
1.1 determine charge
1) counter cation
2) number of ligands (greek numerical prefixes)
3) name of ligand
4) name of central particle ending with -ate
5) overall charge of the complex ion
Ex: [Cu(OH)4]2-
Tetrahydroxidocuprate(2-) ion
K3[Fe(CN)6]
Potassium hexacyanidoferrate(3-)
Na3[Ag(S2O3)2]
Sodium dithiosulfatoargentate(3-) ion
naming cationic complexes
1.1 determine charge
1) number of ligands
2) name of the ligands
3)name of central particle
4) charge of the complex ion
5) counter ion
[Cr(H₂O)₆]³⁺
Hexaaquachromium(3+)
[Co(NH₃)₆]³⁺
Hexaaminecobalt(3+)
[Pt(NH₃)₂Cl₂]NO₃)
diammine-dichloroplatinum(+1) nitrate
[Co(NH₃)₄Cl₂]Br
terraammine-dichlorocobalt(+1) bromide
polydentate ligands
Polydentate ligands are molecules or ions that can bind to a central metal atom or ion through multiple donor atoms
A polydentate ligand binds to a metal at multiple points at the same time
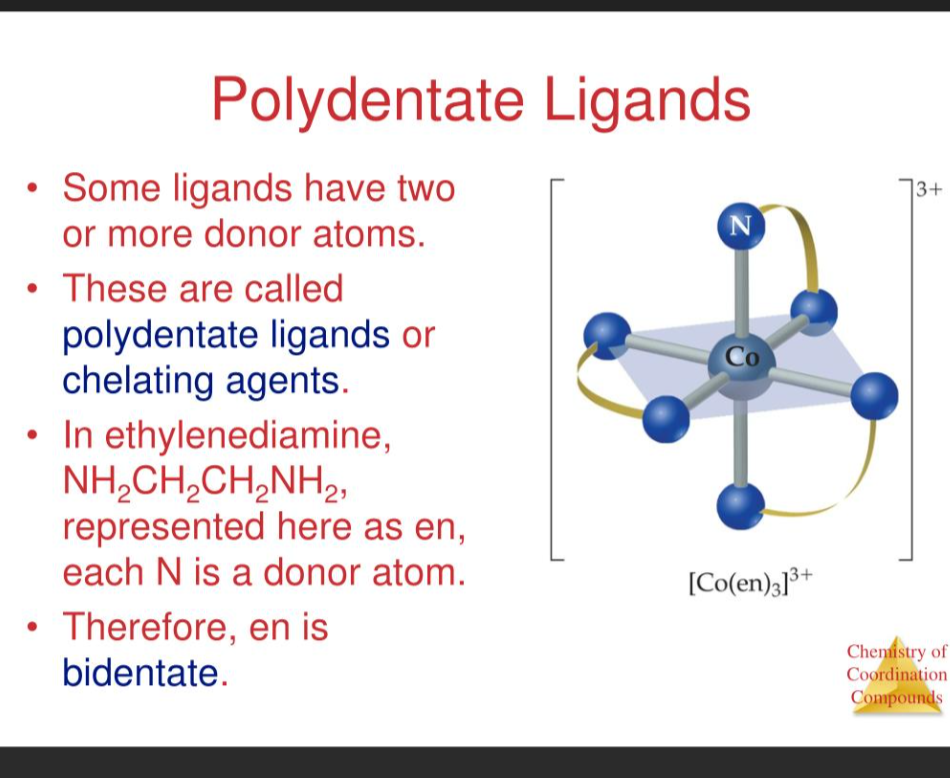
chelate complexes
complexes that contain polydentate ligands are called chelate complexes.
Chelate complexes are coordination compounds where a multidentate ligand binds to a central atom through multiple coordination sites
what do chelate complexes have to do with entropy and statistics?
Statistics (probability argument)
A mono-dentate ligand binds with one point → easy to detach.
A polydentate ligand binds at multiple points → if one bond breaks, the rest are still attached.
=)So it’s more likely to stay bound → more stable.
Entropy
[M(L)6] +3 x→[M(x)3]+6L
=) More particles = higher entropy DeltaS = negative DeltaG = reaction is favored = stabler products
M(x)3 is more stable it has lower energy than M(L)6
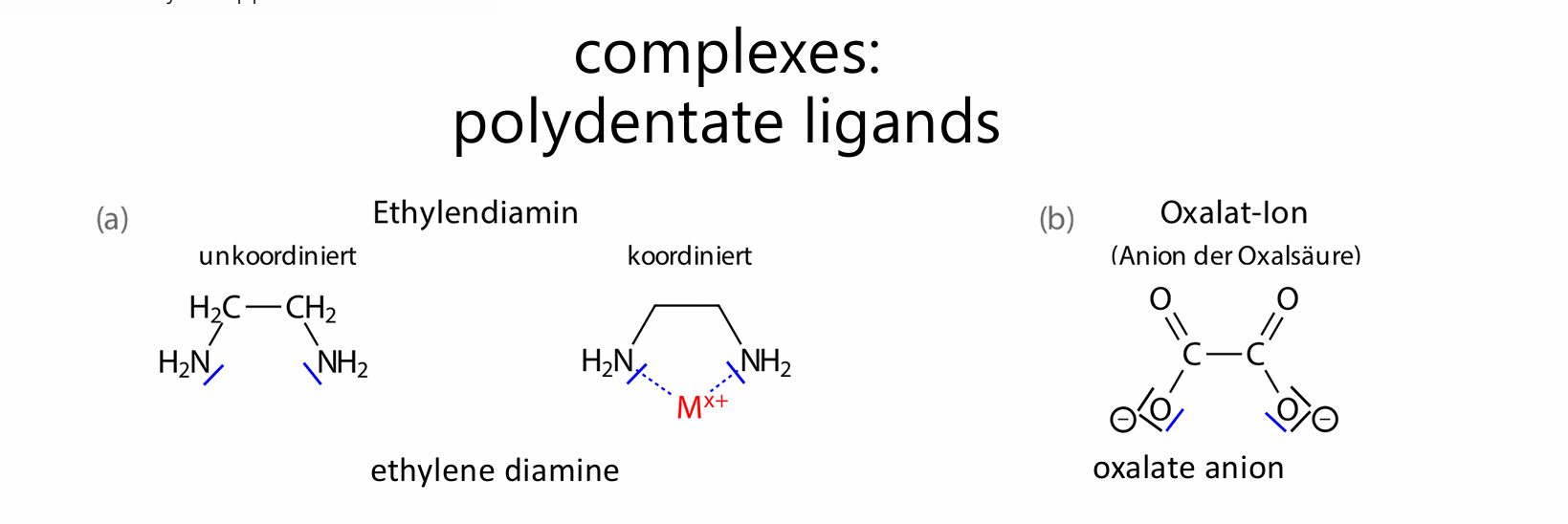
uncoordinated
The molecule is free: H₂N–CH₂–CH₂–NH₂
The two nitrogen atoms each have a lone pair (blue marks)
coordinated
The same molecule now binds to a metal
Both nitrogen atoms attach to the metal
It forms a ring (chelate ring)
en → binds through 2 N atoms
oxalate → binds through 2 O atoms
“Dentate” only applies when a ligand is _____
“Dentate” only applies when a ligand is coordinated
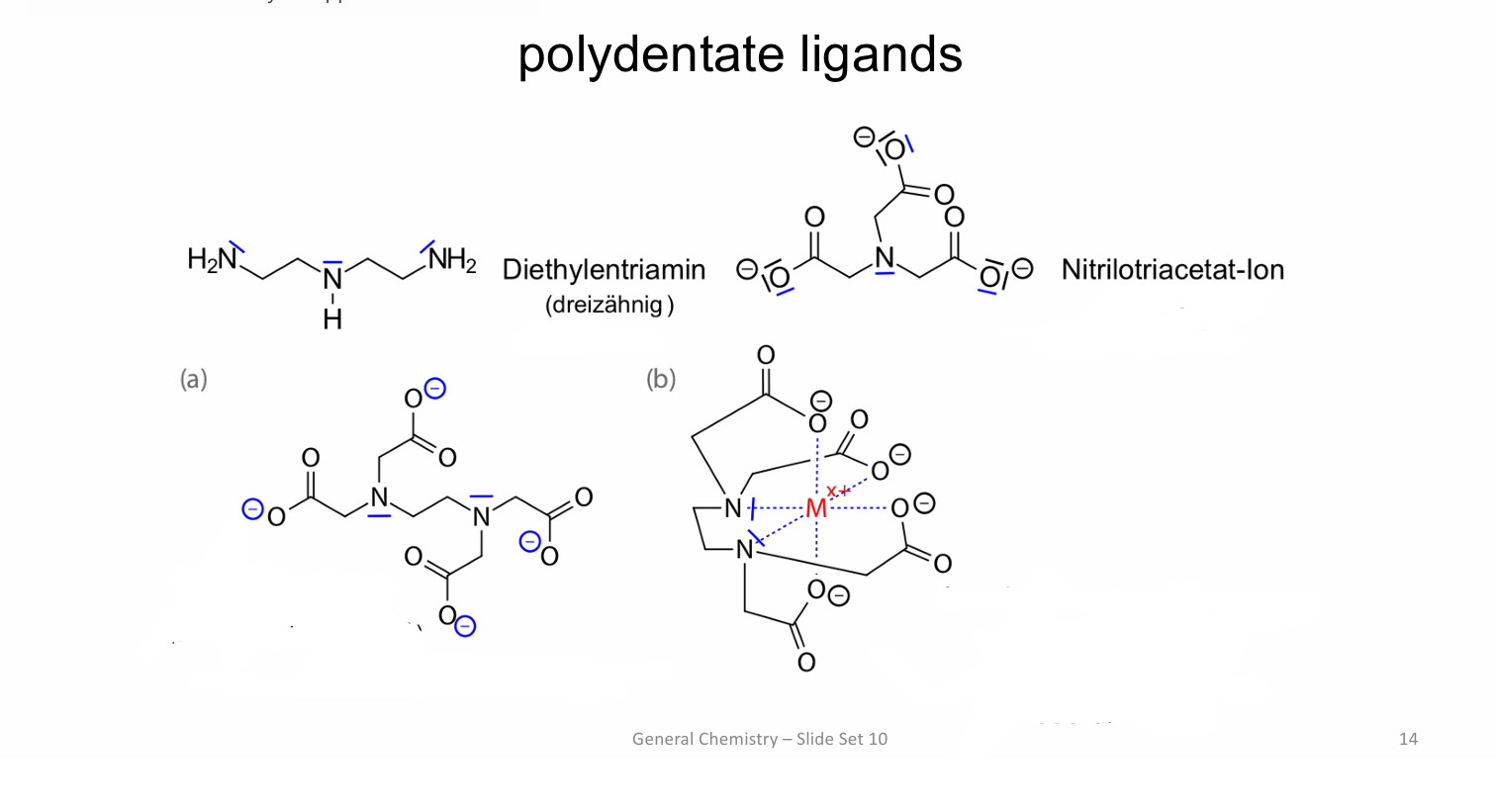
name the spatial arrangement
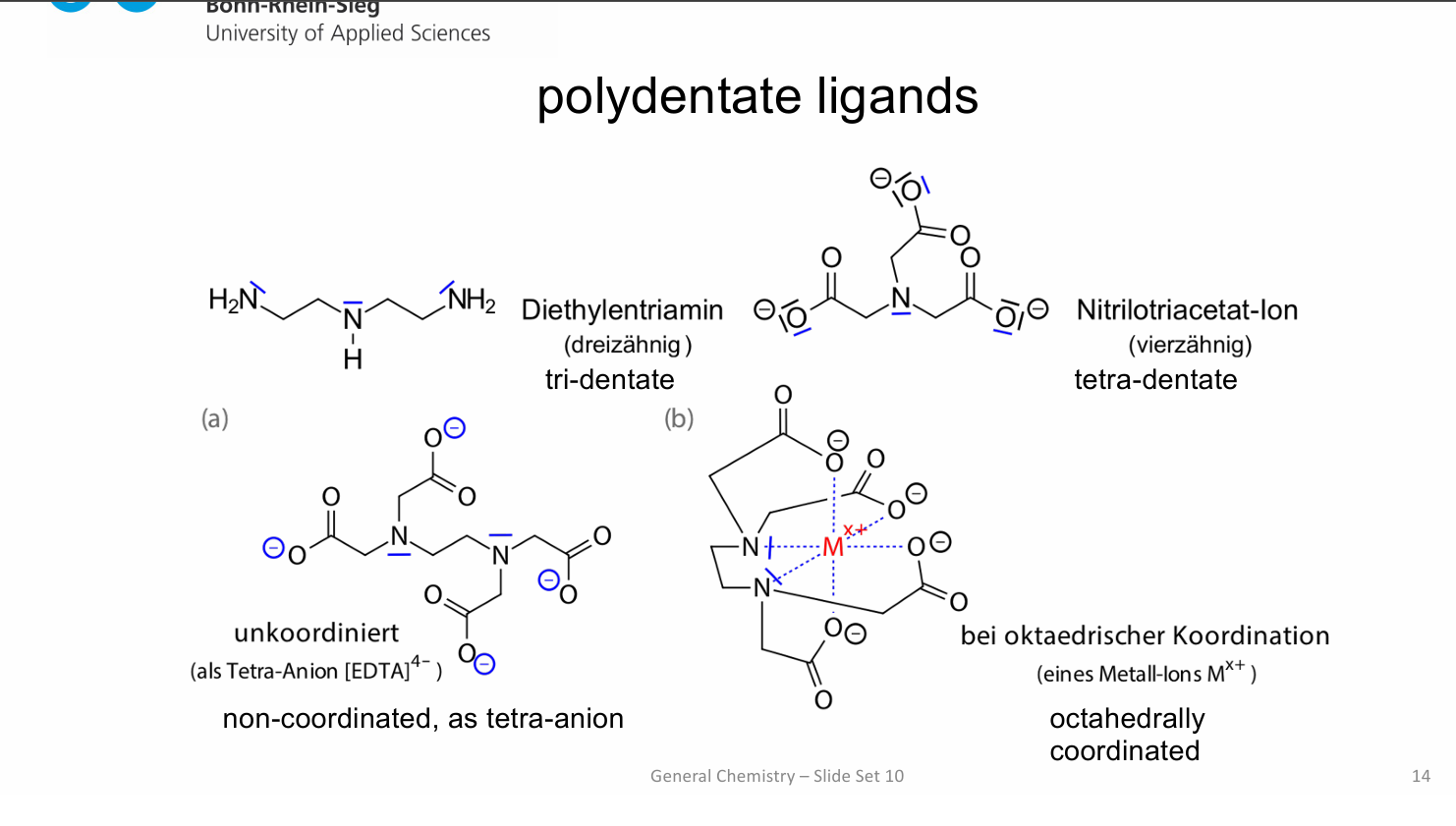
differnce between octahedrally coordinated and octa-dentate
Octahedral = geometry (6 positions)
Octadentate = One ligand binding through 8 atoms
nomenclature for polylingands like en
1) modified greek prefixes:
di- becomes bis-
tri- becomes tris-
tetra- becomes tetrakis- etc.
(meaning “two times, three times, four times, respectively)–
2) The name of the ligand is then set in parenthesis: (name)
Ex:[Cu(en)2]2+
bis(ethylendiamine)copper(2+) cation
Upon adding chloride anions (Cl-) to an aqueous solution of silver ions (Ag+), solid silver chloride (AgCl) precipitates due to the low solubility product (10-10 mol2/L2).
If now an aqueous solution of ammonia (NH3(aq)) is added, the precipitate will dissolve again
why?
since the silver ions (in solution!) react with ammonia to form the complex cation [Ag(NH3)2]+ (diammine silver(I)-ion), thus lowering the amount of silver ions present in the solution
→ the precipitate slowly dissolves. The compound [Ag(NH3)2]Cl has a much higher solubility product
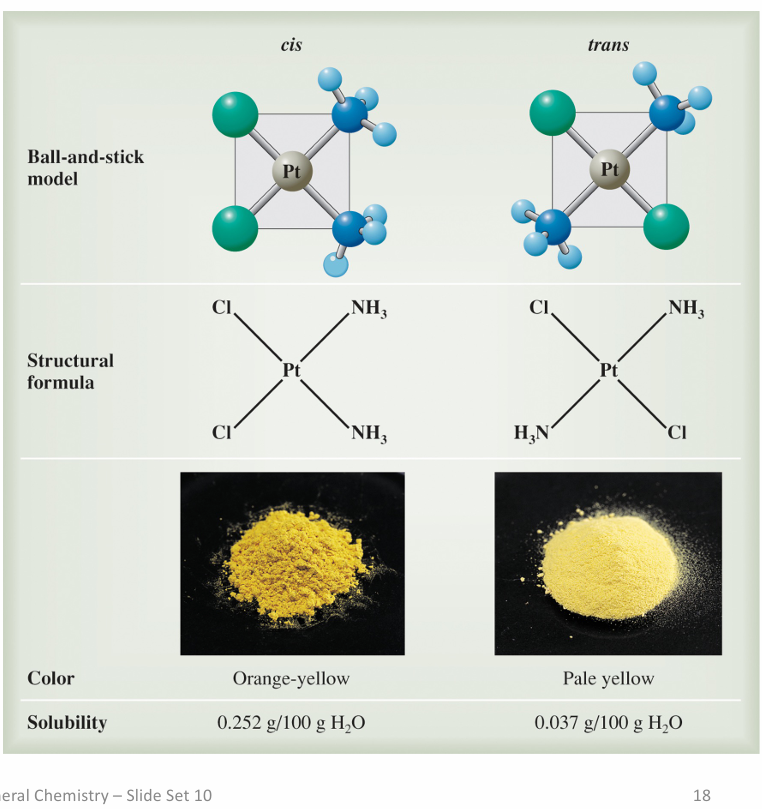
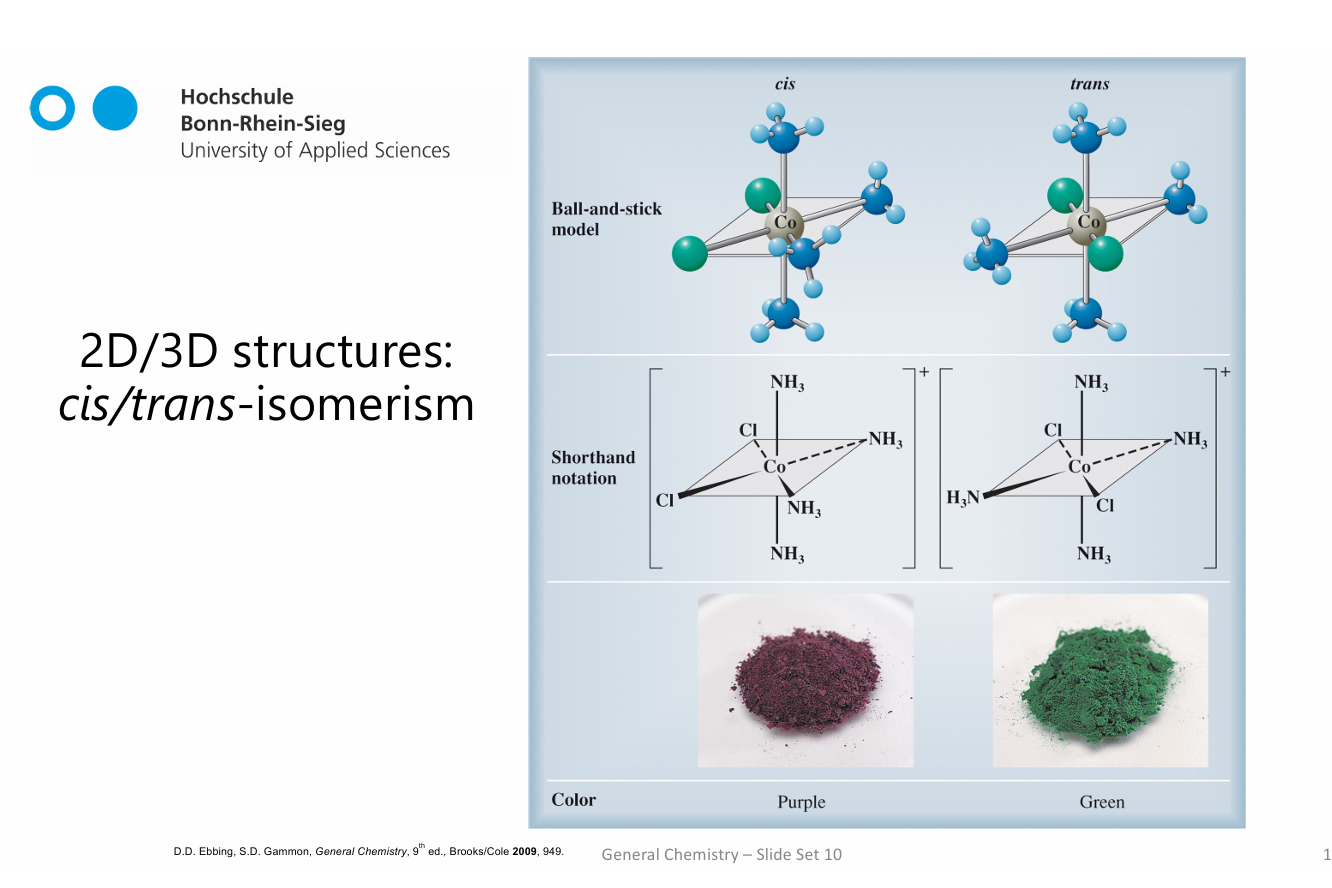
cis-trans-isomerism
which has higher solubility?
Cis isomers are usually more soluble because they are more polar, since their dipols dont cancel out
Polar molecules interact better with polar solvents (like water)
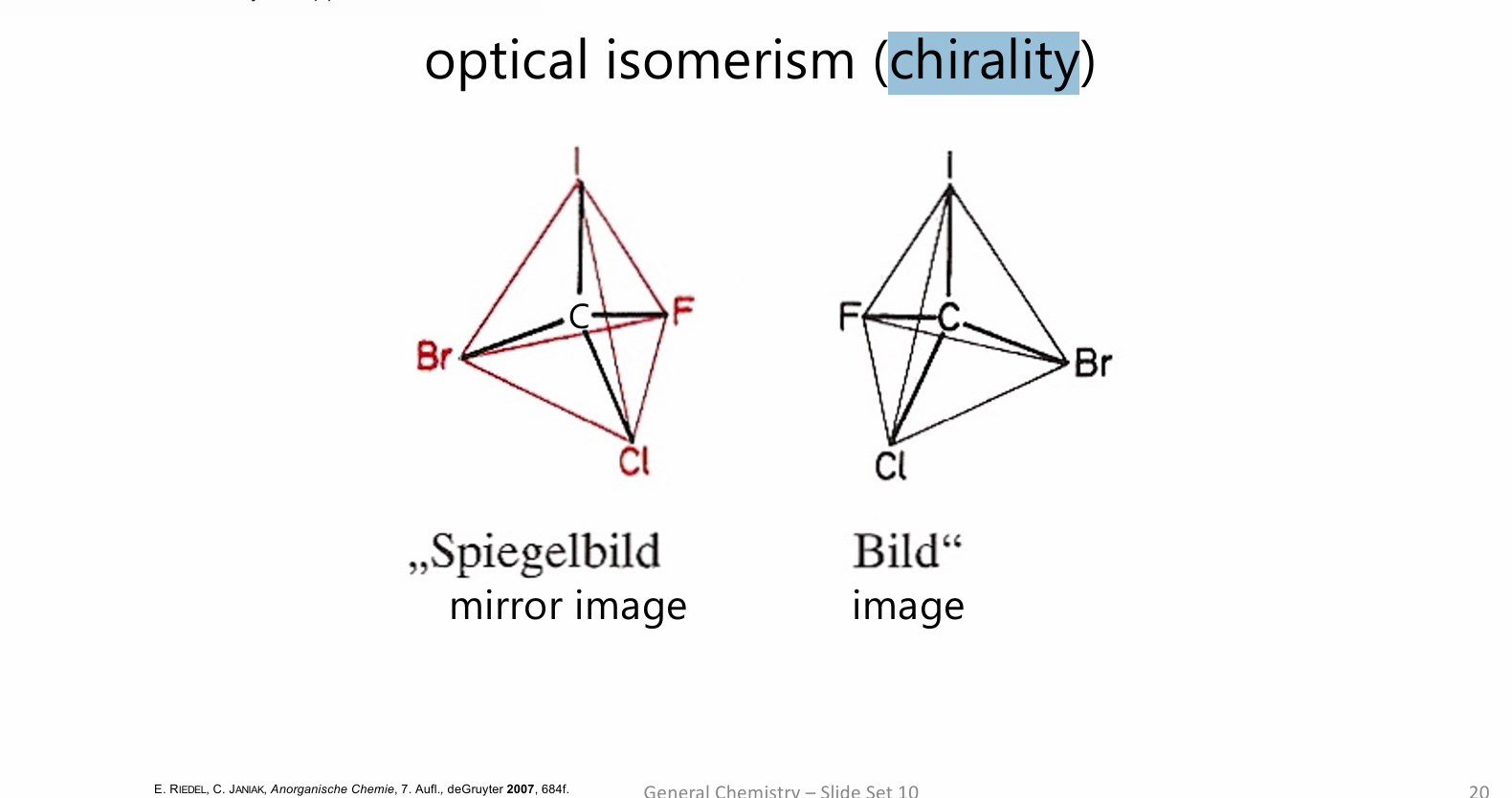
chirality
Chirality is the property of an object not being identical to its mirror image
chirality needs four different groups
or lingands
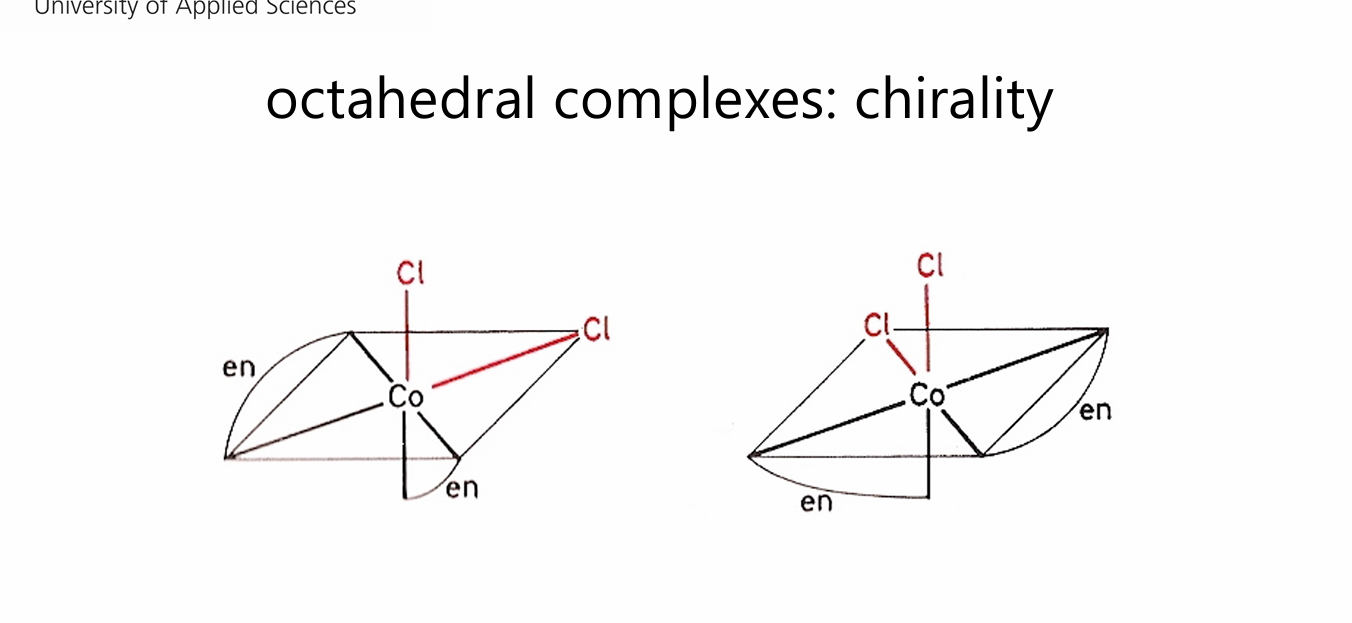
complexes formed with ethylenediamines (en) in an octahedral geometry are always chiral.
Cis arrangement:
Trans arrangement:
what is chiral what is achiral?
Cis arrangement: No inversion center → can form non-superimposable mirror images → chiral.
Trans arrangement: Inversion center exists → mirror image is superimposable → achiral.
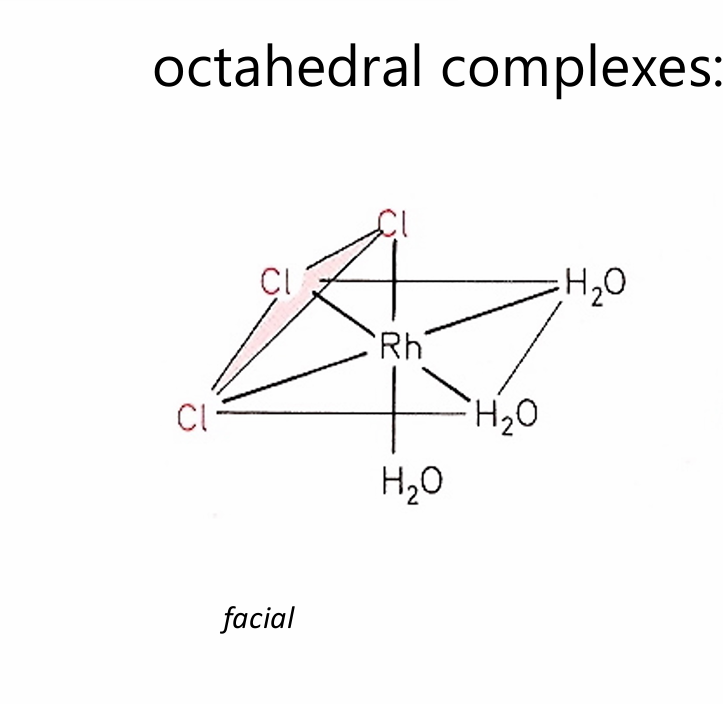
facial (fac)
The three identical ligands occupy one face of the octahedron.
All three A ligands form a triangle on one side of the octahedron.
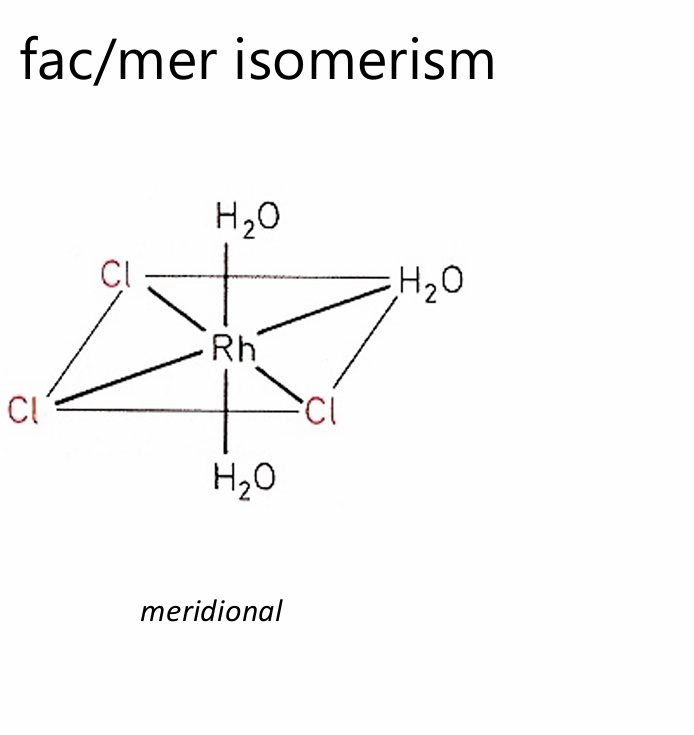
meridional (mer)
The three identical ligands occupy a plane passing through the metal center.
One ligand is opposite another, and the third is perpendicular
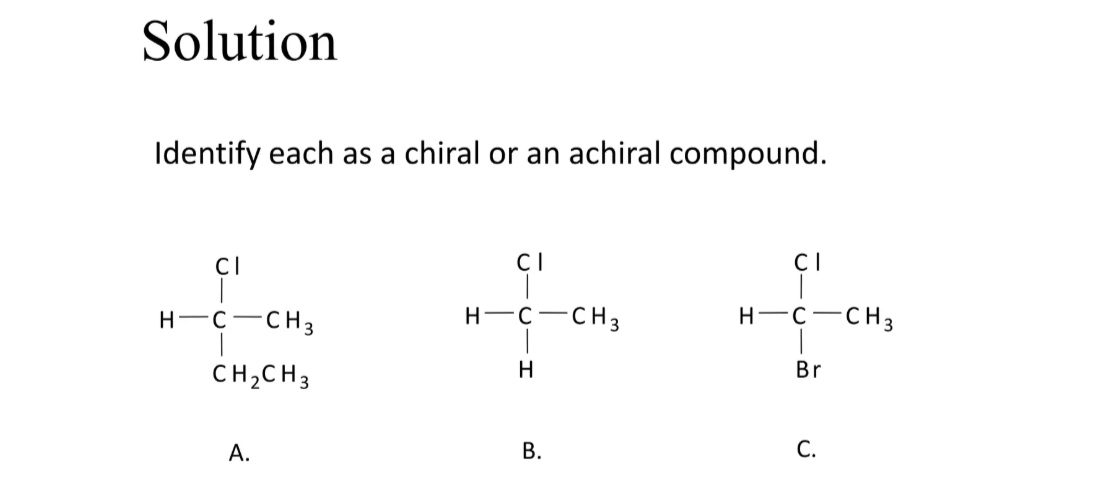
chiral, achiral, chiral
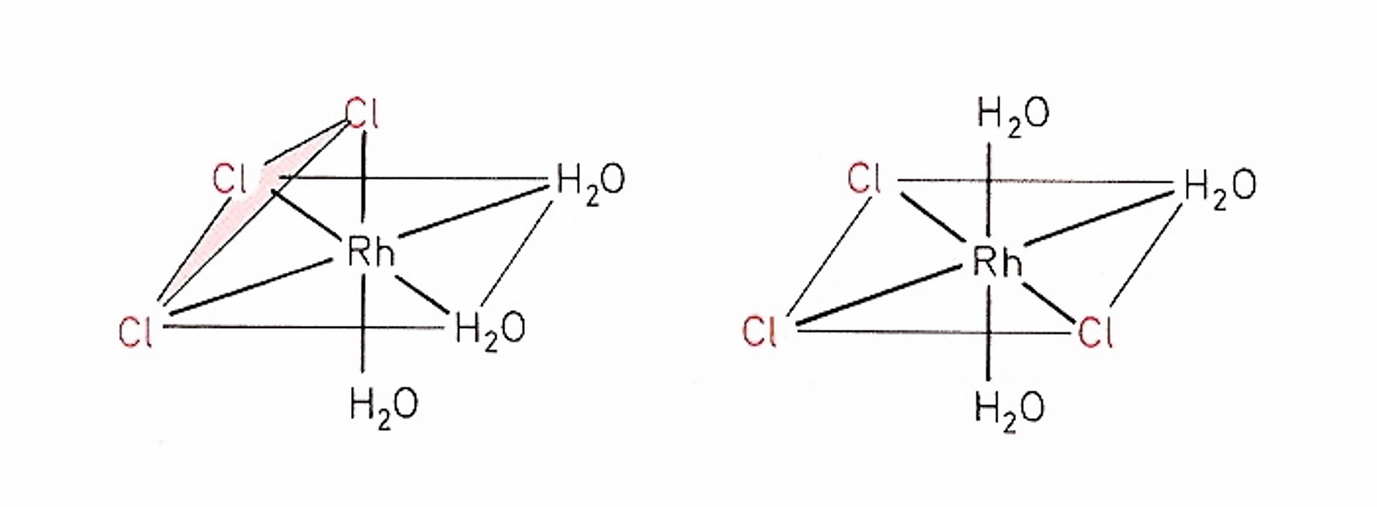
chiral or achiral
meridional is achiral
facial is also achiral