Master Guide to Organic Chemistry Reactions: Starting Material to Product, Reagents, Mechanisms, etc.
1/301
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
302 Terms
Hydrohalogenation (Outcome)
Adds H and X (Br, Cl, I)
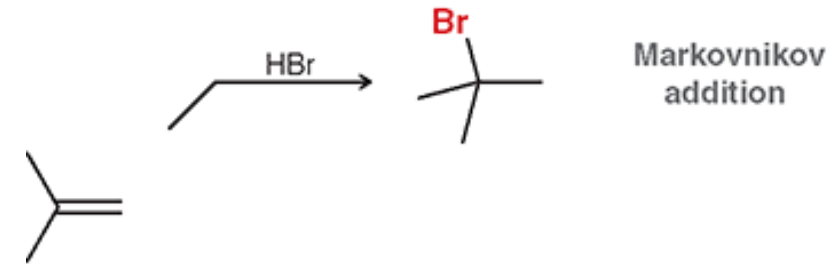
Hydrohalogenation (Reagents)
HBr, HCl, HI ←General form of HX
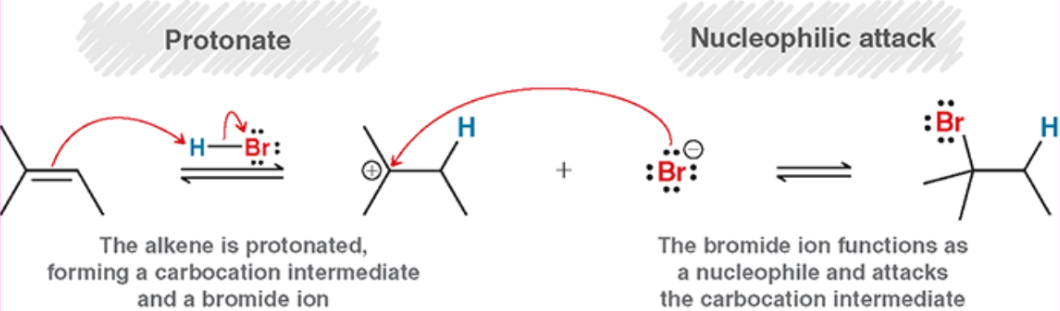
Hydrohalogenation (Mechanism)
Radical Addition of HBr (Outcome)
Anti-Mark addition of X to the less substituted side and H to the more substituted side
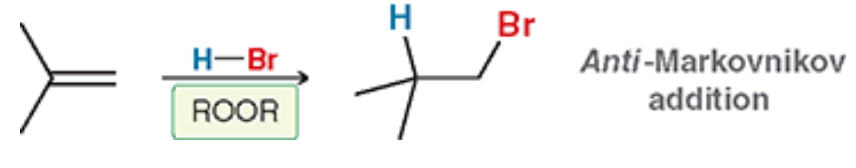
Radical Addition of HBr (Reagents)
This specific reagent. It utilizes any Peroxide (ROOR)
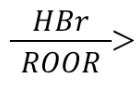
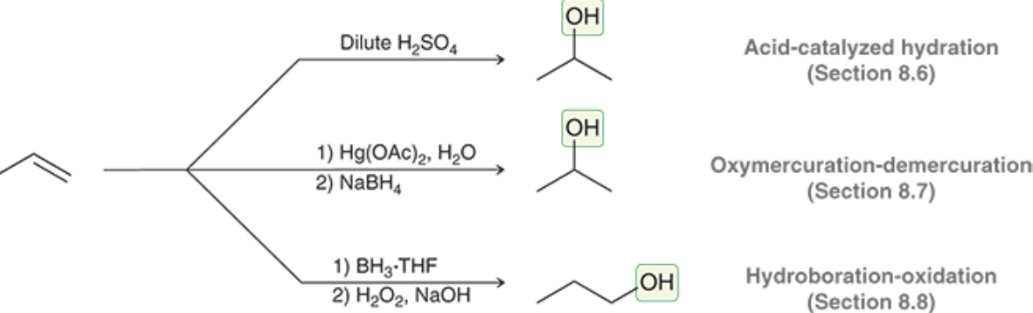
Acid-Catalyzed Hydration (Outcome)
Markovnikov addition of an OH (more sub side) and H (less sub side)
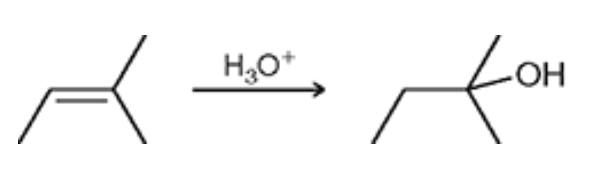
Acid-Catalyzed Hydration (Reagents)
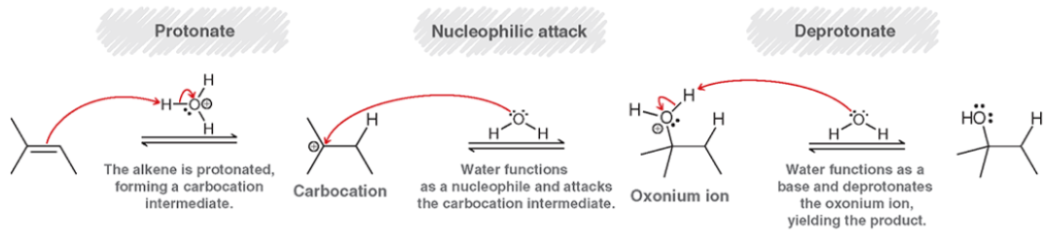
Acid-Catalyzed Hydration (Mechanism)
Watch out for Carbocation rearrangements!
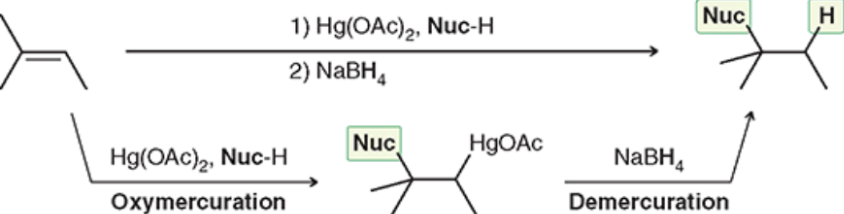
Oxymercuration-Demercuration (Outcome)
Markovnikov Addition of Nuc: and H
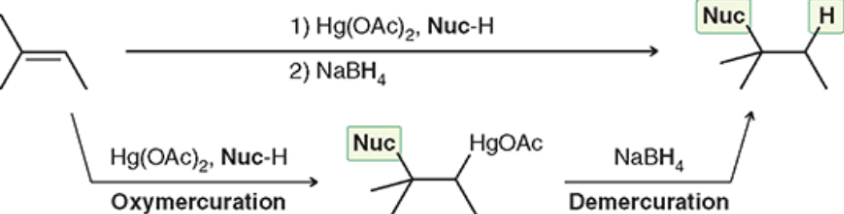
Oxymercuration-Demercuration (Reagents)
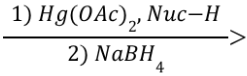
Oxymercuration-Demercuration (Mechanism)
No carbocation rearrangements possible (makes it superior to acid-catalyzed hydration)
Hydroboration-Oxidation (Outcome)
Anti-Markovnikov addition of OH and H
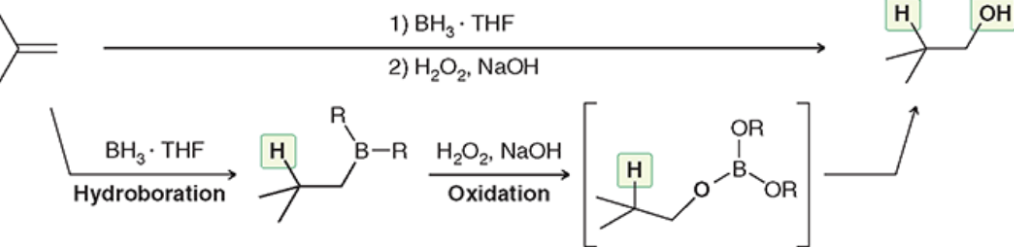
Hydroboration-Oxidation (Reagents)
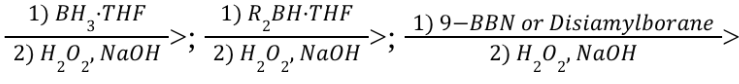
Hydroboration-Oxidation (Mechanism)
Anti-Mark addition + syn addition (chiral center = both on wedge/dash + enant.)
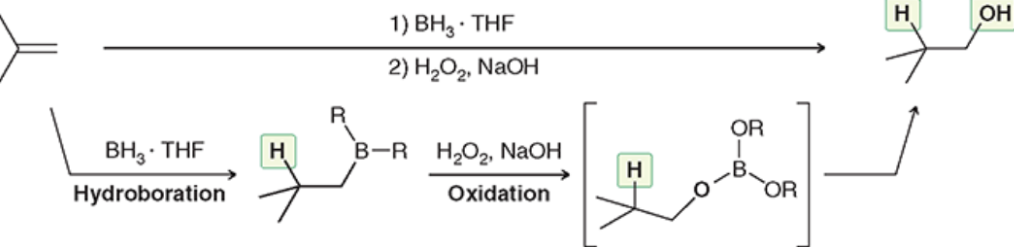
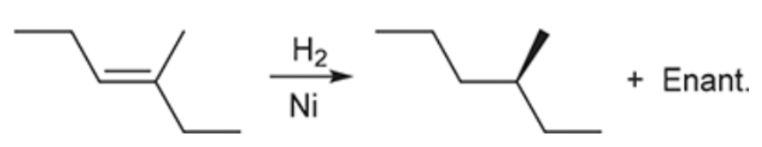
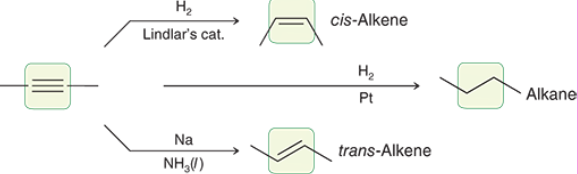
Catalytic Hydrogenation (Outcome)
Syn addition of 2 Hydrogens
Simply turns an alkene into an alkane
Catalytic Hydrogenation (Reagents)
Bromination/Chlorination (Outcome)
Addition of either Br or Cl across the alkene; Stereochemistry depends on the configuration of the starting alkene
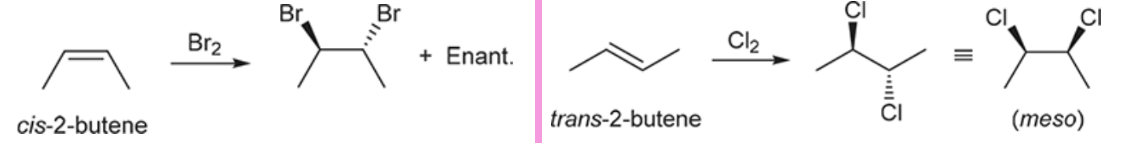
Bromination/Chlorination (Reagents)
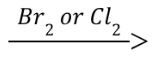
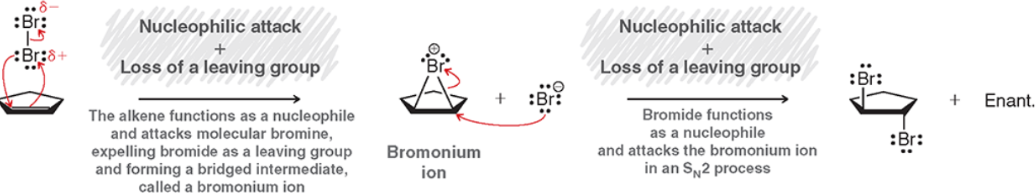
Bromination/Chlorination (Mechanism)
Halohydrin Formation (Outcome)
Addition of X and OH (OH on more substituted carbon)
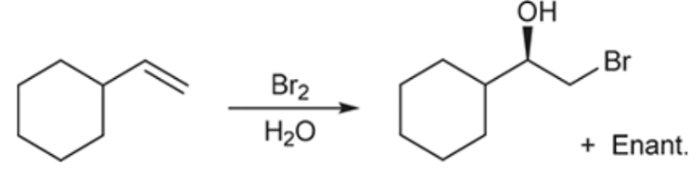
Halohydrin Formation (Reagents)
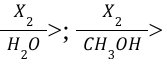
Halohydrin Formation (Mechanism)
Anti addition (one on wedge, other on dash + enant.)
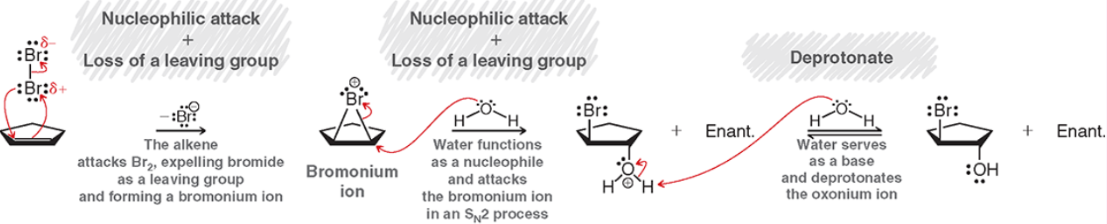
Syn-Dihydroxylation (Outcome)
Syn addition of OH’s
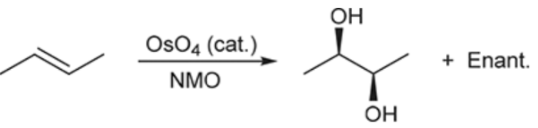
Syn-Dihydroxylation (Reagents)
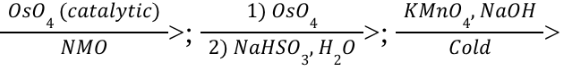
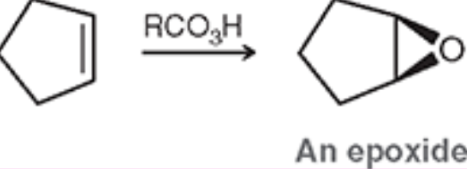
Epoxidation (Outcome + Reagents)
Anti-Dihydroxylation (Outcome)
Anti-addition of OH’s
Goes through an epoxide intermediate
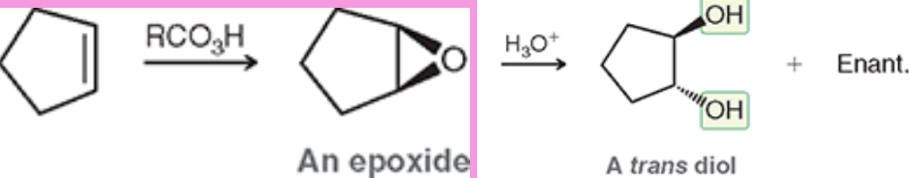
Anti-Dihydroxylation (Reagents)
mCPBA is a type of RCO3H
Ozonolysis (Outcome)
C=C cut into C=O fragments
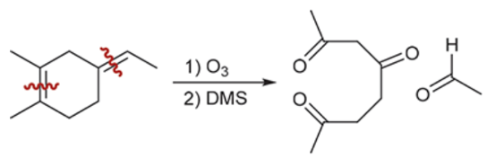
Ozonolysis (Reagents)
Alkyne Elimination (Outcome)
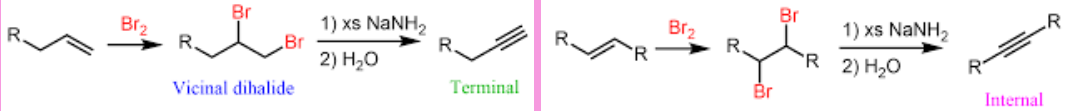
Alkyne Elimination (Reagent)
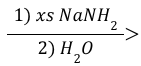
Alkyne Acid-Catalyzed Hydration (Outcome)
It will try to make a ketone.
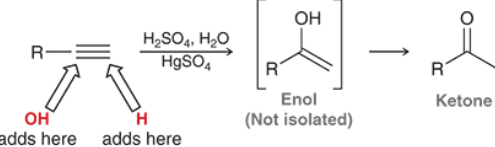
Alkyne Acid-Catalyzed Hydration (Reagents)
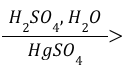
Alkyne Hydroboration-Oxidation (Outcome)
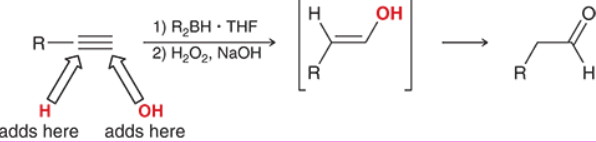
Alkyne Hydroboration-Oxudation (Reagents)
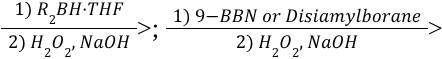
Alkyne Halogentation (Outcome)
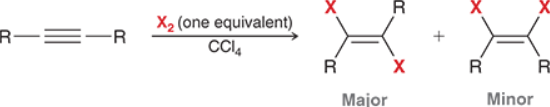
Alkyne Halogenation (Reagents)
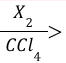
Alkyne Ozonolysis (Outcome)
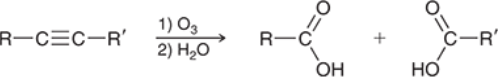
Alkyne Alkylation (Outcome)
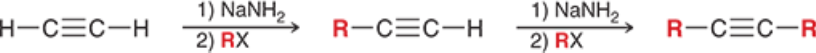
Alkyne Alkylation (Reagents)
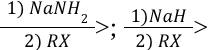
Alkyne Dissolving Metal Reduction (Outcome)
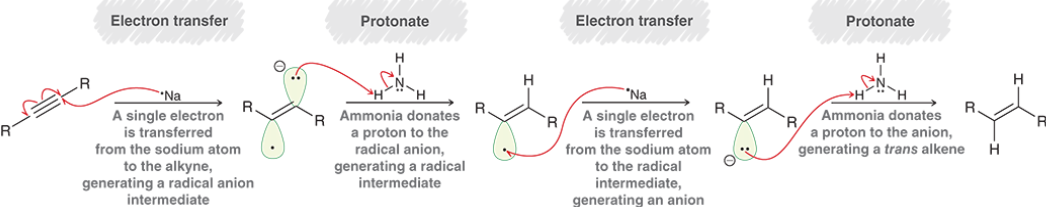
Alkyne Dissolving Metal Reduction (Reagents)
Alkyne Catalytic Hydrogenation (Outcome)
Which Alkene Addition Reactions generate Alcohols?
Acid-Catalyzed Hydration
Oxymercuration-demercuration
Hydroboration-Oxidation
Hydride Reduction of Carbonyl (Outcome)
Changes Carbonyl to secondary (2o) alcohol
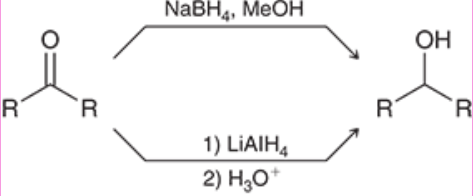
Hydride Reduction of Carbonyl (Reagents)
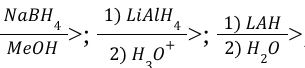
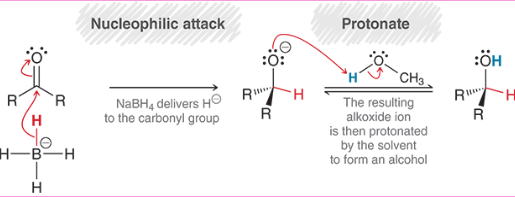
Hydride Reduction of Carbonyl (Mechanism)
Hydride Reduction of Carboxylic Acids (Outcome)

Hydride Reduction of Carboxylic Acids (Reagents)
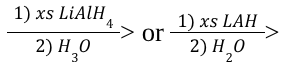
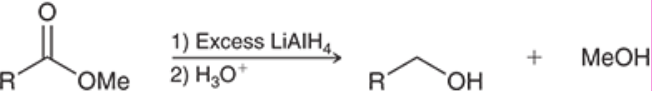
Hydride Reduction of Esters (Outcome)
The OMe can be subbed out for any R group bonded to that oxygen
Hydride Reduction of Esters (Reagents)
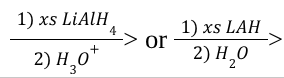
Hydride Reduction of Esters (Mechanism)
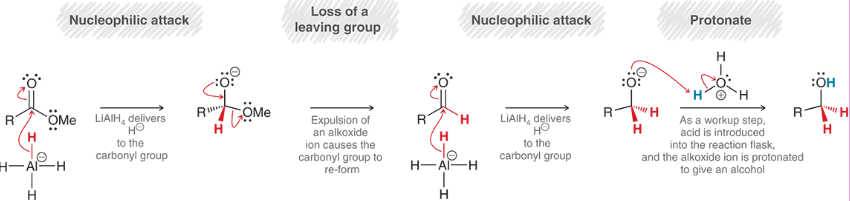
How do NaBH4 and LAH/LiAlH4 compare when reducing carbonyls?
NaBH4 is weaker than LAH/LiAlH4, so it can only reduce carbonyl groups to alcohols, whereas LAH/LiAlH4 can also reduce carboxylic acids and esters
Carbonyl Grignard Reagent Reduction (Outcome)
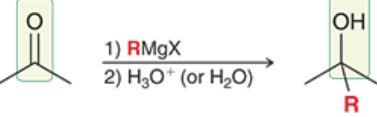
Carbonyl Grignard Reagent Reduction (Reagents)
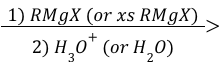
Carbonyl Grignard Reagent Reduction (Mechanism)
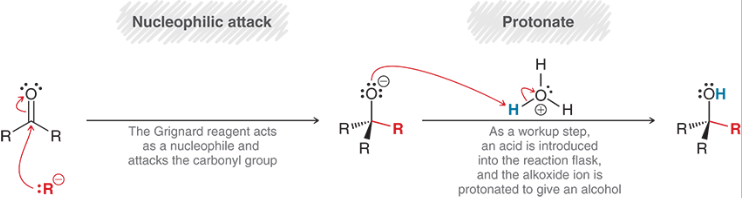
Ester Grignard Reagent Reduction (Outcome)
It requires excess of the Grignard reagent to add both R groups
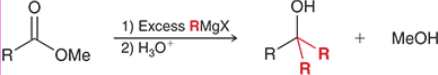
Ester Grignard Reagent Reduction (Mechanism)
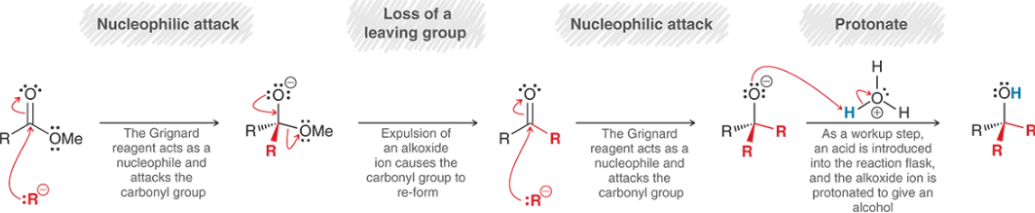
Protection of Alcohol
You MUST protect the alcohol because if added to a Grignard, it will deprotonate the alcohol, so you must sub out the hydrogen prior to the Grignard reaction.

Deprotection of Alcohol
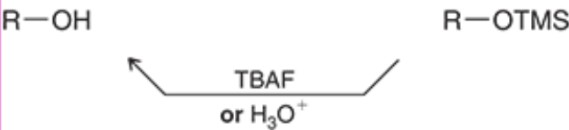
SN2 Reactions with Alcohols (Outcome)

SN2 Reactions with Alcohols (Reagents)
py and Et3N are just the solvents
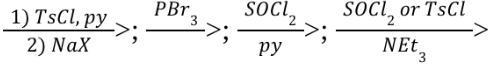
Primary 1o Alcohol Oxidation (Outcome)
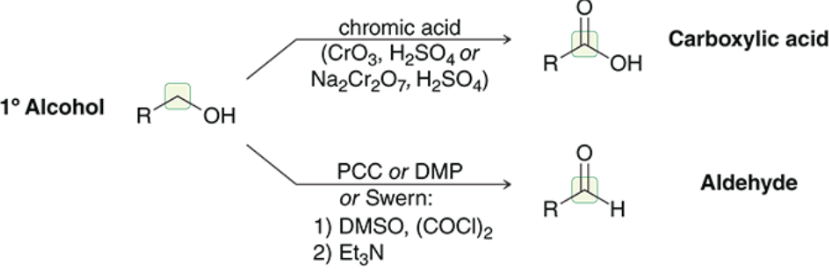
Primary 1o Alcohol Oxidation (Reagents)

Secondary 2o Alcohol Oxidation (Outcome)
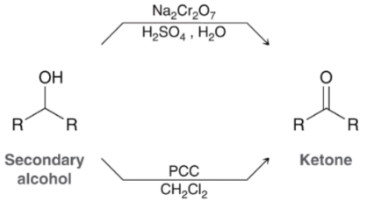
Secondary 2o Alcohol Oxidation (Reagents)

What happens when you try to oxidize a tertiary alcohol?
Trick question, nothing happens and there is no reaction
Williamson Ether Synthesis (Outcome)
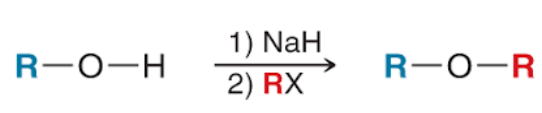
Williamson Ether Synthesis (Reagents)
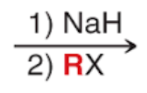
Williamson Ether Synthesis (Mechanism)
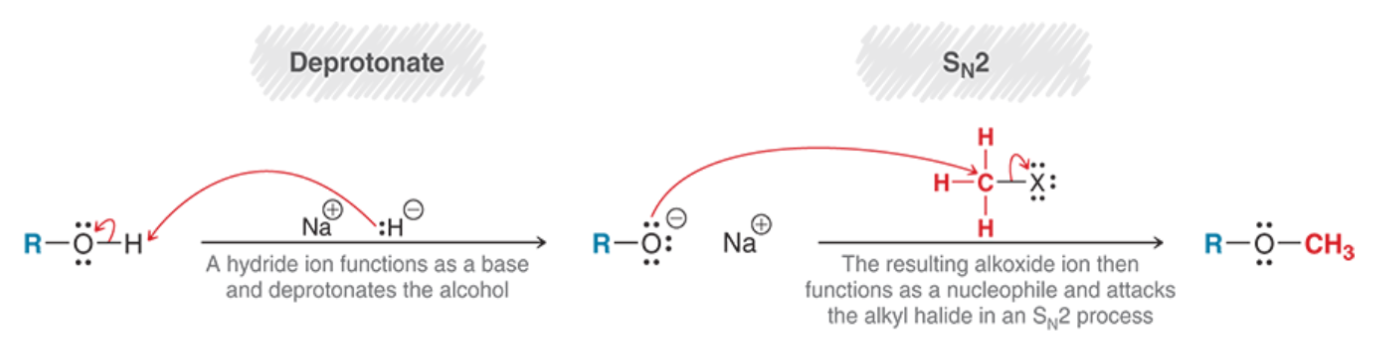
Ring-Opening of Epoxides w/ Strong Nucleophiles (Outcomes + Reagents)
Pay attention to the order of the side that opens and is protonated
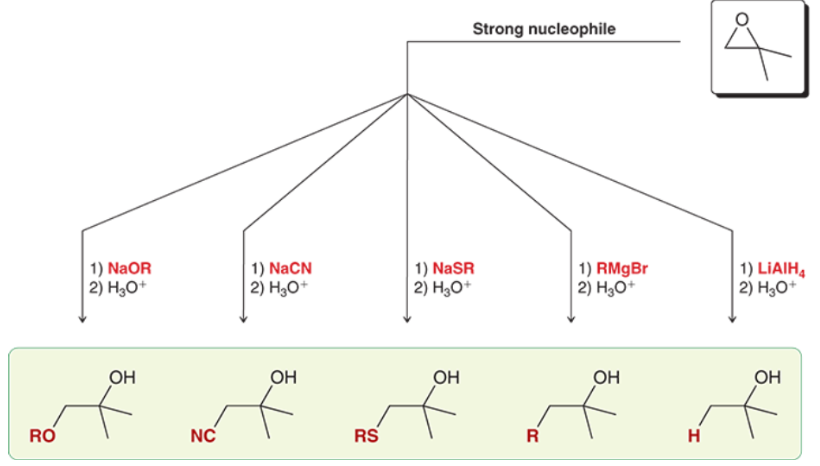
Ring-Opening of Epoxides w/ Acid-Catalyzing Reagents (Outcomes + Reagents)
What are the important characteristics of the Ring-Opening Reactions of Epoxides to be watchful for?
Epoxide ring-opening is SN2, and so the least hindered carbon is more reactive towards a strong nucleophile, so the nucleophile will go on the less substituted/more open carbon
As with SN2, inversion of configuration occurs at the carbon the nucleophile is attacking, while the O in the epoxide (that the H from H3O+ or H2O attaches to) remains the same
For acidic conditions, the X, OH, or OR nucleophile attacks the less substituted carbon if a 1o and/or 2o are present, while a 3o carbon makes the nucleophile attack said 3o carbon, and inversion of configuration still occurs at the carbon the X, OH, or OR is attacking
Diels-Alder Reaction (Outcome)
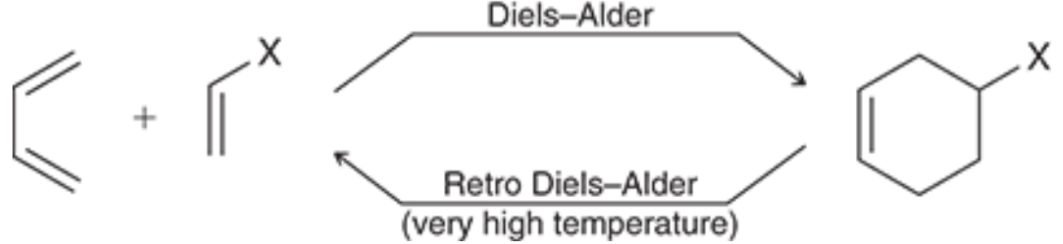
Diels-Alder Reaction (Mechanism)
The partial negative charge of the diene attacks the partial positive charge of the dienophile, and the other arrows conform around that to create your product
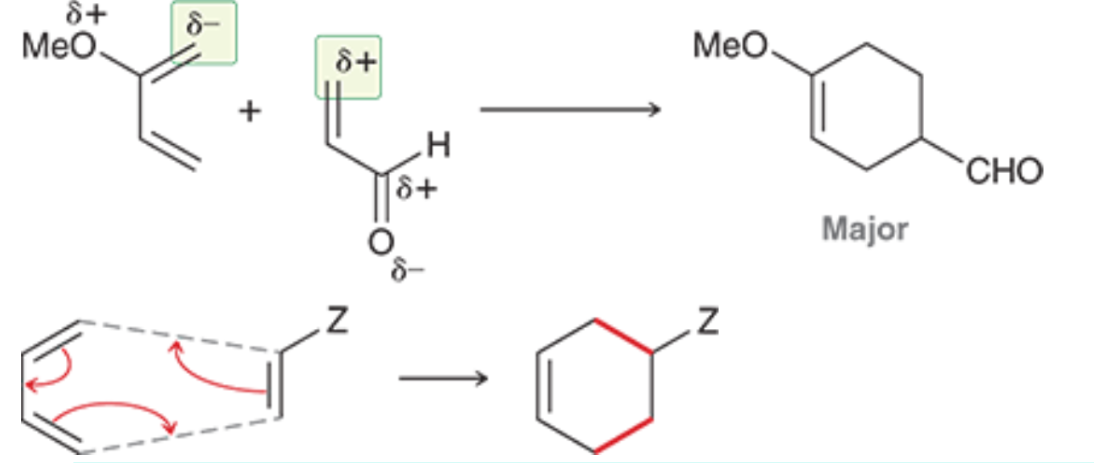
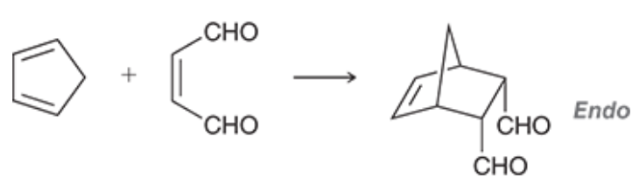
What is the Endo Rule and how does it pertain to Diels-Alder reactions?
The Endo Rule states that when a bridge is created by a Diels-Alder reaction, electron-withdrawing groups (EWGs) prefer to be under (endo) the created bridge
Electrophilic Addition (Mechanism + Outcome)
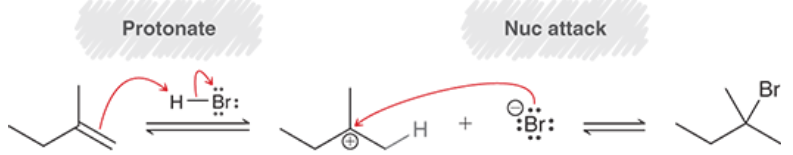
Electrophilic Addition of dienes w/ HBr
The kinetic (low temp) product is the one that is created by the most stable carbocation
The thermodynamic (high temp) product is the one that features the more substituted alkene
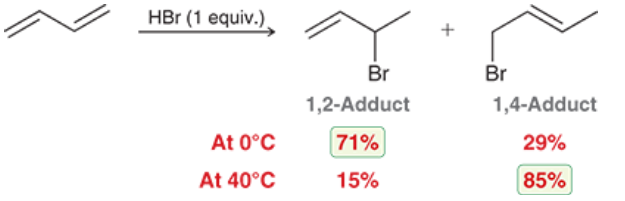
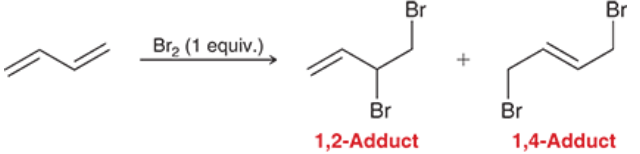
Electrophilic Addition of dienes w/ Br2
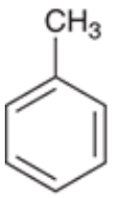
Toluene
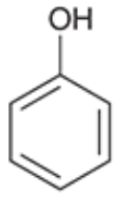
Phenol
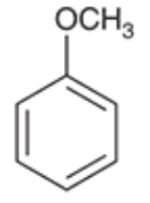
Anisole (Methoxybenzene)
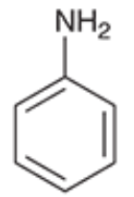
Aniline
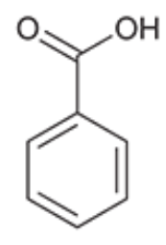
Benzoic Acid
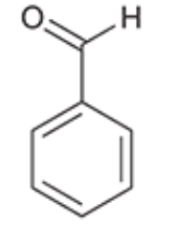
Benzaldehyde
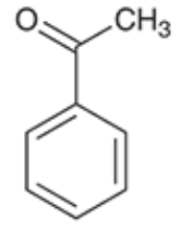
Acetophenone
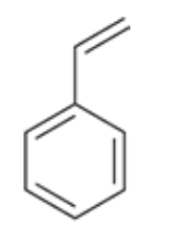
Styrene
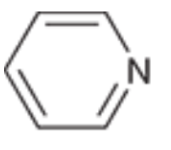
Pyridine
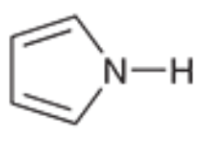
Pyrrole
What are the differences between the ortho-, meta-, and para- positions on disubstituted rings?
Ortho- is adajacent
Meta- is one apart
Para- is opposite
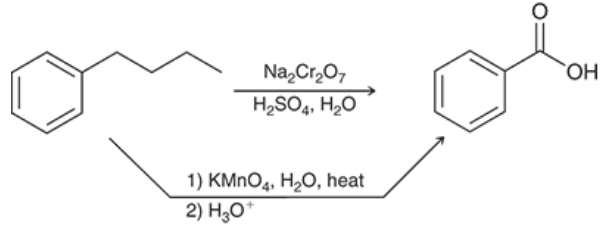
Oxidation at the Benzylic position (Outcome + Reagents)
Benzylic position is just any carbon atom attached directly to, but not on, a benzene ring)
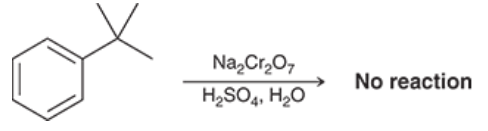
What is the one exception to the oxidation reaction at the benzylic position?
It can be done by any alkyl group except for tert-butyl
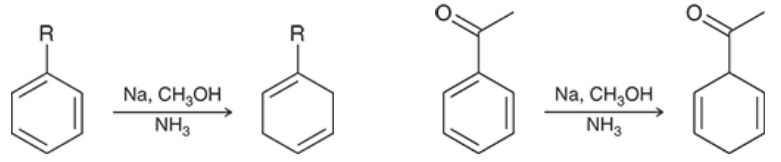
Birch Reduction (Outcome)
If the group at the benzylic position is an EWG, then the carbon on the ring itself will be reduced and lose that double bond. If the group there is an EDG, it will NOT be reduced and will be an alkene double bond on the left or right of that group
Birch Reduction (Reagents)
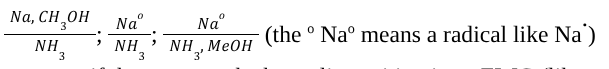
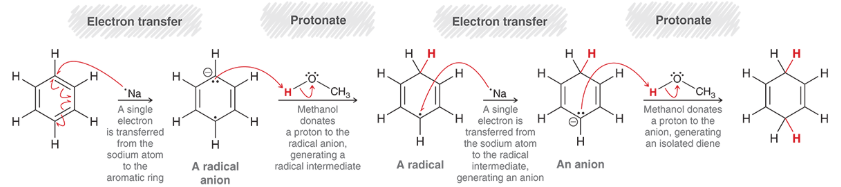
Birch Reduction (Mechanism)
EDGs are what type of directors?
Ortho-para directors (and also activators)
EWGs are what type of directors?
Meta directors (and deactivators)
Why are halogens a special case in directing?
Because while they are deactivators, they are ortho-/para-directing instead of meta-directing
What does it mean to be an activator versus deactivator?
Activators push electron density into an aromatic ring and thus speed up reactions, while deactivators pull electron density away from the ring and thus slow it down
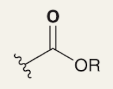
EWG + Moderate Deactivator (Meta-Directing)