Chem 14A Midterm 3
1/98
Earn XP
Description and Tags
Waves & electromagnetic radiation, Bohr model of atom, orbitals, electron configuration, quantum numbers, periodic trends, effective nuclear charge, structure, and bonding.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
What is the acronym for electromagnetic radiation?
GXU VIBGYOR IMR
What does GXU VIBGYOR IMR stand for?
Gamma rays, x rays, ultraviolet, violet, indigo, blue, green, yellow, orange, red, infrared, microwaves, radio waves
How many electrons can be housed in the S orbital?
2
What orbital can only house 2 electrons?
S
How many electrons can the p orbitals house?
6
Which orbital can house up to 6 electrons?
P
How many electrons can the d orbitals house?
10
Which orbital can house up to 10 electrons?
D
How many electrons can the F orbital house?
14
Which orbital can house up to 14 electrons?
F
What shape is the S orbital?
Spherical
Which orbital assumes a spherical shape?
S
What shape are the P orbitals?
Bi-lobal
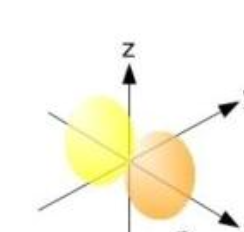
What is this shape and what orbitals assume it?
Bi-lobal, P
What shape are the D orbitals?
Four fold symmetry and dumbbell with donut
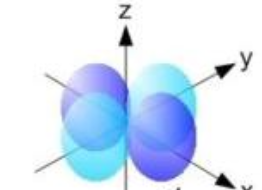
What is this shape and what orbitals assume it?
Four-fold symmetry and dumbbell with donut, D
What shape are the F orbitals?
Multi-lobal
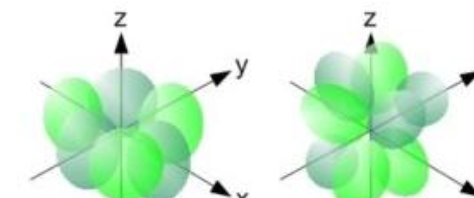
What is this shape and what orbital assumes it?
Multi-lobal, F
What are the four quantum numbers?
n, l, ml, ms
How is the length of a wave measured?
Peak-to-peak distance
What is an antinode?
The node at the peak of the wave
What is a node?
Where the wave crosses over the axis
What is a wave amplitude
Measurement between the wave peak/valley and the axis
What happens when amplitudes add?
Constructive interference (amplitude increases)
What happens when amplitudes cancel?
Destructive interference
What is constructive interference?
When waves add
What is destructive interference?
When waves cancel
What are the units of energy?
Joules (J)
What is a photon?
The amount of energy in one wavelength
What are the units for wavelength?
nanometers (nm)
What are the units for energy per mole?
KJ/mol
What is the unit of frequency?
wave events per second (s^-1)
What is the series that corresponds to electron jumps from nh to n=1?
Lyman series - UV
What is the series that corresponds to electron jumps from nh to n=2?
Balmer series - visible light
What is the series that corresponds to electron jumps from nh to n=3?
Paschen series - infrared
What is the series that corresponds to electron jumps from nh to n=4?
Bracket series
How many orientations does the S orbital have?
1
What orbital has 1 orientation?
S
How many orientations does the P orbital have?
3
Which orbital has 3 orientations?
P
How many orientations does the D orbital have?
5
What orbital has 5 orientations?
D
How many orientations does the F orbital have?
7
Which orbital has 7 orientations?
F
What is the equation for the max number of electrons an energy level (principle quantum number) can hold?
2n²
What is 2n²
The equation for the max electrons an energy level (principle quantum number) can hold
How do you calculate the number of orbitals in a subshell?
2L+1
What are shielding electrons?
All electrons except valence
What is Zeff and what is the equation?
Effective nuclear charge. Protons-shielding electrons
What is the periodic trend for atomic radius?
increases going down, decreases going right
When an anion is formed, what happens to the atomic radius?
It increases
When a cation forms, what happens to the atomic radius?
it decreases
What is the periodic trend for electronegativity?
Decreases going down, increases going right
What does a higher zeff value indicate in terms of electronegavitity?
Higher zeff, more electronegative
What is the periodic trend for metallic character?
Increases going down, decreases going right
What does it mean to have higher metallic character?
Less affinity for electrons
What is the periodic trend for ionization energy?
Decreases going down, increases going right
What does it mean to have a high ionization energy?
A lot of energy is required to remove an electron.
What is the periodic trend for electron affinity?
Decreases going down, increases going right
What periodic trends are opposite that of electronegativity?
Atomic radius and metallic character
What periodic trends are the same as electronegativity?
Ionization energy and electron affinity
How do you calculate whether there’s a dipole moment and how strong it is?
Find delta EN. If it’s greater than zero, there is a dipole. The larger the value, the stronger the dipole toward the more electronegative atom
What bond exists when the delta EN is 0-0.4?
Nonpolar covalent
What bond exists when delta EN is 0.4-1.8?
Polar covalent
What bond exists when delta EN is >1.8?
ionic
What is meant by a covalent bond?
Electrons are shared between atoms.
What type of bond is present when atoms are sharing electrons?
Covalent
How do you calculate formal charge?
[# valence electrons] - [1/2(# bonding electrons)+(# lone electrons)]
How many electrons is H allowed to carry?
2
How many electrons is C allowed to carry?
8
How many electrons is N allowed to carry?
8
How many electrons is O allowed to carry?
8
How many electrons is F allowed to carry?
8
Which elements follow the octet rule?
C N O F
How many electrons is Cl allowed to carry?
more than 8
How many electrons is As allowed to carry?
more than 8
How do you calculate electron geometry?
LP + BG
How do you calculate molecular geometry?
EG, BG
What molecular shape results from MG of 2,2? how does it look? What is the bond angle?
linear, 180

What molecular shape results from an MG of 3,2? How does it look? What is the bond angle?
Bent, <120

What molecular shape results from an MG of 4,2? How does it look? What is the bond angle?
Bent, <120

What shape results from an MG of 3,3? How does it look? What is the bond angle?
Trigonal planar, 120
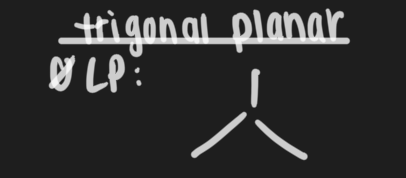
What shape results from an MG of 4,3? How does it look? What is the bond angle?
Trigonal pyramidal, bond angle <120

What does paramagnetic mean?
There are unpaired electrons
What does diamagnetic mean?
All electrons are paired
What atoms most commonly have H-bond donors and acceptors?
N, O, and F
What is the full procedure for molecule examples?
Valence calculation, lewis structure, initial formal charge, modified lewis structure, final formal charge, resonance, bond angle, EG + MG, delta EN, vectors, hybridization, orbital diagrams
What bonds are strongest? Which are weakest?
Single bonds or less than single bonds are weakest, triple bonds and above are strongest
What does bond order signify about strength?
Higher bond order means stronger bond
How do you calculate quantum number L from N?
n-L
For energy level one, what is the orbital shape, orientation(s), orbital name(s), and max electrons?
L=0, 1, s-orbital, 2 max electrons
For energy level 2, what is the orbital shape, orientation(s), orbital name(s), and max electrons?
L=0, one, s-orbital, 2 max electrons
L=1, 3, p-orbitals, 6 max electrons
total of 8 max electrons, 4 orbital orientations
For energy level 3, what is the orbital shape, orientation(s), orbital name(s), and max electrons?
L=0, one, s-orbital, max 2 electrons
L=1, 3, p-orbitals, max 6 electrons
L=2, 5, d-orbitals, max 10 electrons
Total of 18 max electrons and 9 orientations
For energy level 4, what is the orbital shape, orientation(s), orbital name(s), and max electrons?
L=0, 1, s-orbital, 2 max electrons
L=1, 3, p-orbitals, 6 max electrons
L=2, 5, d-orbitals, 10 max electrons
L=3, 7, f-orbitals, 14 max electrons
total of 32 max electrons and 16 orientations
What does isoelectronic mean?
electron configuration matches that of another element - or they are isoelectronic with that element
What is the rule for electron removal upon ionization?
Remove from highest energy orbital first, which depends on the principle quantum energy numbers. You add up all the N values for a certain orbital to determine this. e.g. 4s is n=4 and 3d is n=3
What is the periodic trend for Zeff?
Increases to the right and stays consistent downward
What shells/orbitals are valence electrons contained in?
The highest principle quantum number shell
When is it appropriate for the central atom to not have a formal charge of zero? How should charges be distributed within a molecule to be most stable?
When the total charge doesn’t add up properly. negative charges should go to the most electronegative atoms, so the central atom may not be set up to have a charge of 0.