5/6) Phosphorus, metals, and salt
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
details on phosphorus, and as a contaminant
essential nutrient, often the limiting factor, excess P leads to eutrophication
particulate phosphorus sticks to particles, not particularly useful for microorganisms
what are potential sources of phosphorus?
urbanization
effluents from wastewater treatment plants
agricultural runoff
upstream sources
where is phosphorus more of an issue, surface or groundwater?
surface water
sediments can contain phosphate attached to sediments, but phosphorus measured in river is low
what does phosphorus easily attach to?
iron and aluminum oxides and hydroxides
how is phosphorus released from iron oxides
in reducing conditions, iron gets reduced, becoming soluble and iron oxide dissolves
any phosphate that was stuck to it has nothing to attach to anymore, it gets released
true or false: phosphorus contamination can be buffered by wetland
true: more organic life present, so more phosphorus use, it will be less intense of a contaminant
details on manganese in alberta groundwater including MAC and sources
MAC = 0.12 mg/L
aesthetic objective = <0.02 mg/L
reducing component, likes to hang out in water
rocks from marine shales, weathering, deposit, places where pyrite oxidation happens
will exceed MAC in post-oxic zone
details on iron in alberta groundwater
no MAC
aesthetic objective = <0.3 mg/L
taste of water affected
oxidized Fe3+ is usually in solids as Fe(III)
reduced Fe2+ is usually dissolved in water
where will iron exceed AO?
will exceed in post oxic zone, it gets reduced
will not exceed in oxic zone, iron would immediately precipitate
will not in sulfidic zone, would precipitate as iron sulphate immediately
details on acid groundwater
oxidation of pyrite
one of the most strongly acid-producing processes in nature
lowering of groundwater table may expose and oxidize pyrite
if pyrite remains reduced in subsurface, then this wouldn’t happen
what are consequences of acid groundwater
acid water seeps out, metals can be too high, or just the effects of acid
pH<3
releases high amounts of Fe 3+ and SO42-
if pH is less than 4, Fe3+ becomes soluble
other heavy metals soluble at low pH such as Zn and Cu
as pH increases to >4, Cu adsorbs and concentrations decrease rapidly. zinc is the same but at a higher pH
what happens to pyrite oxidation in a closed system?
which system would result in lower pH values?
it stops because you can’t resupply pyrite to continue the reaction
open system results in lower pH values because you can replenish the reactants and continue the reaction
what’s important about arsenic in groundwater
oxidation of arsenopyrite liberates arsenic
shallow groundwater may have significantly elevated concs of arsenic
definition of heavy metals and metalloids, examples, extra details
heavy metal: any metallic chemical element that has a relatively high density (>5 g/cm3) and is toxic or poisonous at low concentrations
metalloid: has both metal and nonmetal properties
ex: As, Cd, Cr, Hg, Pb. arsenic, cadmium, chromium, mercury, lead
many metal(loids) are redox-active, and thus are mobilized or immobilized via redox processes
where would you expect to see the highest trace metal concentrations in groundwater
heavy metal and trace elements can be found at any depth
concs are dependent on:
natural presence of metal in aquifer sediments
groundwater chemistry (oxic vs anoxic)
potential anthropogenic sources
what are common anthropogenic sources of arsenic?
insecticides, weed killers, wood presevatives
added to animal feed to prevent diseases
pigments/paint
manufacturing of electronic components
medicine
chemical warfare
what are sources of arsenic contamination in water?
natural/human activities
arsenic is an element that occurs naturally
volcanic activity, erosion of rocks and minerals, and forest fires
agricultural applications, mining and smelting
90% of arsenic used in industry is for wood preservative purposes
what is the MAC of arsenic
0.001 ppm / 10 ppb
Canada: as low as reasonably achievable (ALAR)
US: maximum contaminant level goal (MCLG) = 0ppm
before 2001 = 50 ppb
which form of arsenic is more mobile?
As3+ more mobile than As5+
As5+ is oxidized, it sorbs easily on iron hydroxides
As3+ is reduced, is more mobile
arsenic mobility is linked closely with solid Fe oxides and hydroxides
what zones will As exceed the MAC?
very few samples exceed
middle of post-oxic zone has highest As conc. about half the upper 50% are above the MAC
if there is lots of iron in GW, what form is it in?
it must be Fe2+ because GW is typically pH 6-8, meanings Fe is under reducing conditions
what form of iron causes staining?
what form of iron causes bas taste?
Fe3+ causes staining
at the surface, there is lots of oxygen, so Fe2+ will oxidize to Fe3+
Fe2+ causes bad taste
what are the problems with arsenic in groundwater in SE asia?
1). As concentrations are patchy, there are general trends but it’s difficult to predict where you might find high arsenic wells
2). weathering of himalayas, if that material has arsenopyrite, then the arsenopyrite is deposited in the floodplains
floodplains have lots of organic-rich sediments and fine-grained materials
^these conditions are suitable for slow moving water, reducing down the redox ladder
where are arsenic concentrations lowest? where is it highest?
lowest at the surface (near-surface GW and surface water) AND in the deepest wells
highest in mid-depth wells (from 10-50m)
explain the relationship between arsenic and iron in sediments
if there is lots of iron in water, you get lots of As
in oxidizing conditions nothing happens, but in reducing conditions, the As gets released
shallow wells: when iron oxides exist, there is enough oxidizing power so that As sticks to iron
middepth wells: more reducing environment and iron reduces, liberating the As from the compound and releasing into the water
explain the sequence of events for As-Fe relationship
first need a source of As, then As in the aquifer sorbs to iron oxides under aerobic conditions
when anaerobic conditions evolve, reductive dissolution of iron oxides may occur
Fe-As compound dissolves, liberating As from solid materials to the groundwater, turning it from As5+ to the more mobile As3+
degradable organic carbon is needed to fuel redox reactions
what conditions are needed for arsenic contamination to occur
arsenic rich materials eroded and weathered in a specific way
areas with active mountain belts and associated sedimentary and deltaic basins
what type of rocks are a major geologic source of arsenic?
slates and phyllites
what is the problem with private wells?
public wells are regularly tested for various contaminants including arsenic
private wells it’s up to the owner to make sure it’s okay to drink
usually private owners only test their water after something goes wrong (bad taste/smell/colour, or a spill nearby)
arsenic has no taste, smell, or colour, and can do a lot of damage in small concs
it also costs money to test water, and even more to remove contaminants
what are ways to remove arsenic from drinking water?
what are ways that don’t work to remove arsenic from water?
reverse osmosis and distillation
reverse osmosis is cheaper and takes less time and energy
however most home rev osmosis systems can’t reduce arsenic concs more than 15%
doesn’t work: water softeners, carbon filters, UV treatment
what is the MAC for fluoride?
what are potential health effects?
what is recommended conc for fluoride?
1.5 mg/L
more than that = mottled teeth, bone damage
0.7 ppm
less than recommended also unhealthy for teeth
what are geologic sources of fluoride?
apatite: want to get phosphate out of apatite, end up liberating fluoride
fluorite
cryolite: lower melting point of ores that we want to get aluminum out of
micas
amphiboles
sellaite
anthropogenic: apatite (fertilizer) and cryolite (aluminum factories) mostly
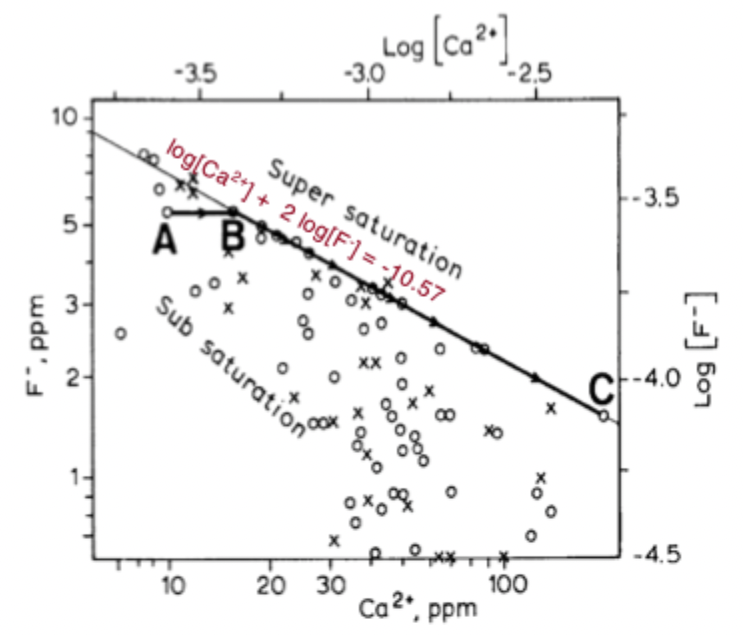
why do all the data points plot under the line in this figure? explain more about it
they are subsaturated with respect to fluorite
if you had so much Ca and F that water plots above, the mineral fluoride would precipitate out until you were back at the black line
on the line is in eqlm with respect to the mineral
at high Ca concs, you’ll get relatively low F concs, because if you had more F, the mineral would precipitate out above the line
what do water types have in common in alberta groundwater
dominated by sodium, don’t have much calcium
calcium bicarbonate water can exchange cations with clay minerals to get sodium back, sodium bicarbonate rich
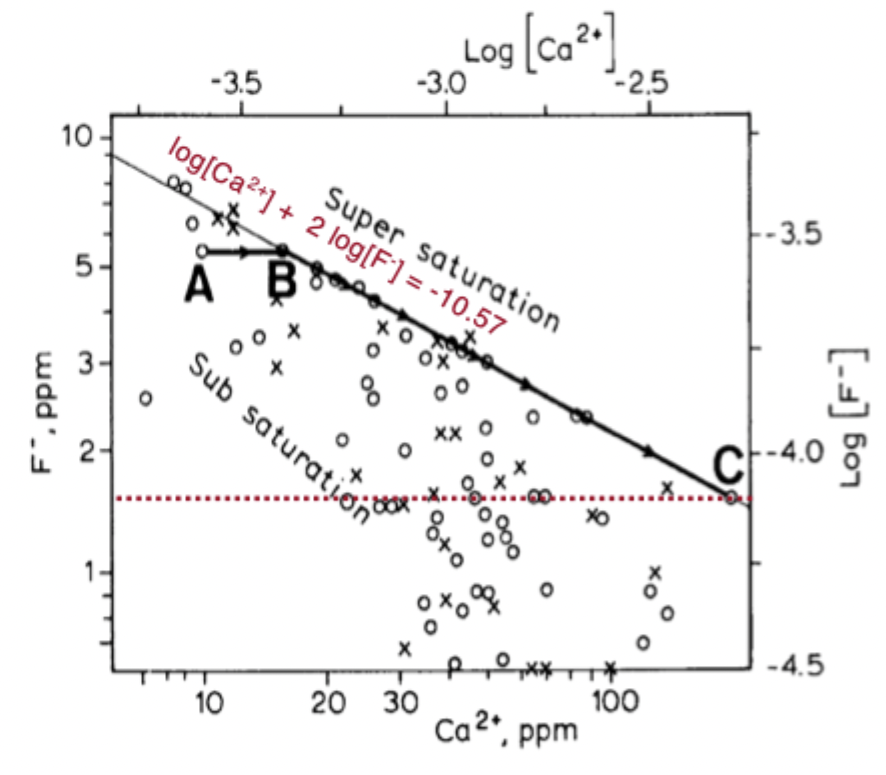
how can we remediate high fluoride in water?
adding Ca2+ (gypsum)
increasing Ca2+ puts you at a point on this graph where the eqlm line is lower, so fluorite will precipitate out and F decreases
gypsum is far more soluble than fluorite
details on chromium
naturally occurring in serpentinitesm
anthropogenically used for chrome plating, dyes, wood preservation
highly soluble at low pH
adsorbs to solids when pH is moderate-high
Cr3+ is more reducing, insoluble
Cr6+ is more mobile
chromate will adsorb to Fe/Al oxides in soils
MAC is 0.05 mg/L
De minimis = keep as low as you can even if its below MAC
details on cadmium
naturally found deep in subsurface in ores, released during volcanic activity
in solution occurs as Cd2+
solubility similar to iron, high at low pH, low at high pH
high adsorption potential in soils
MAC is 0.0007 mg/L
Cd bioaccumulates/magnifies
details on lead
common source is lead-acid batteries, historically in gasoline
occurs as Pb2+
MAC is 0.005mg/L
strong adsorption by soil at pH >5
what is the general rule for solubility of metals in water
high solubility at low pH
low solubility at neutral+ pH
high potential for adsorption at elevated pH values if there are sorbing materials available
extra details for sorption of metals in water
what metal is the first to adsorb and why?
pH values below 3 and they all remain in solution
Cr is the first to adsorb: high charge and small size. more affinity to negative compounds
order is charge related then size
what are sources of salts
naturally occurring, but are often anthropogenically enhanced like from overpumping aquifers near saltwater causing an intrusion
de-icing roads, saline spills
karstic environment: water on one side comes in, karstic surfaces allow the saline water to get to the surface where the evaporite is not on top of that side. salt is removed from evaporite formation
what are problems with salt contamination
crop damage (saline soils are generally unfit for plant growth)
weathering pipelines