Topic 2 - States of Matter & Mixtures
1/86
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
density of solid
high
density of liquid
medium
density of gas
low
arrangement of particles in a solid
regular lattice
arrangement of particles in a liquid
randomly arranged
arrangement of particles in a gas
randomly arranged
movement of particles in a solid
vibrate around a fixed position
movement of particles in a liquid
move around and past each other
movement of particles in a gas
move rapidly and randomly in all directions
ranking of states of matter from lowest energy to highest per particle
solid
liquid
gas
What happens during a change of state from solid to liquid and liquid to gas
When the melting/boiling point is reached, then energy is used to break the attractive forces between the particles instead of increasing the mean particles’ energy
melting
solid to liquid
boiling
liquid to gas(from below surface as well as on surface)
freezing
liquid to a solid
evaporation
liquid to a gas(at surface only)
condensation
gas to a liquid
sublimation
solid to a gas
deposition
gas to solid
What type of change is a change of state of matter?
physical change
Explain the change in arrangement from solid to liquid
The regular lattice arrangement of a solid is broken and now there is a random arrangement. However, the particles are still touching due to existing but weaker forces of attraction
Explain the change in energy from solid to liquid
The mean energy per particle increases due to the solid being heated up
Explain the change in particle movement from solid to liquid
the particles go from vibrating in their fixed positions to being able to move past each other
Explain the change in particle arrangement from liquid to gas
The particles go from being able to touch each other to being completely separate from each other, this is due to the attractive forces between the particles being completely broken
Explain the change in particle movement from liquid to gas
the particles go from moving past each other to flying around the container in a rapid and random motion
Explain the change in particle energy from liquid to gas
the mean energy has increased per particle, thus the energy is at its highest in a gas
How does condensation work?
When a gas is cooled its particles lose energy and when they bump into each other, they lack energy to bounce away again, instead grouping together to form a liquid
When does evaporation only happen?
Evaporation occurs only at the surface of liquids where high energy particles can escape from the liquids surface at low temperatures, below the boiling point of the liquid
What 2 factors affect how quickly a liquid can evaporate?
The larger the surface area and the warmer the liquid/surface, the more quickly a liquid can evaporate
Does evaporation occur at one specific temperature?
No, evaporation occurs over a range of temperatures, but heating will speed up the process as particles need energy to escape from the surface
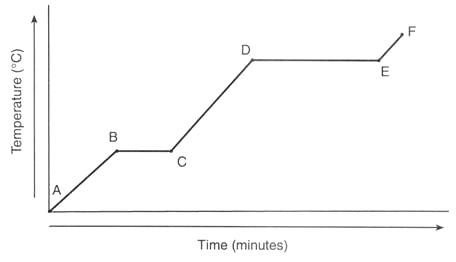
label this diagram
A - solid
B - transitioning from solid to liquid
C - liquid
D - transitioning from liquid to gas
E - gas
F - increasing the temperature and speed of the particles in a gas
How to work out substance state of matter given temperature, bp and mp
mp > temp> bp - liquid
temp <= mp - solid
temp >= bp - gas
Why do u suppose condensing and freezing plateau?
Energy is being absorbed to create attractive forces between the particles
pure substance
consists of a single element/compound and contains no other substances
mixture
2 or more elements that are physically but not chemically mixed together.
The chemical properties of the substances in a mixture remain unchanged
What’s the difference between mps and bps in pure substances vs mixtures?
mixtures have a range of melting and boiling points as they consist of different substances that tend to lower the mp and broaden the mp range
pure substances melt and boil at sharp and specific temperatures
How do you test the purity of drugs?
Slowly heat up a small amount of the sample which makes it easier to observe the exact melting point. This is then compared to data tables and the closer the measured value is to the actual mp/bp then the purer the sample is
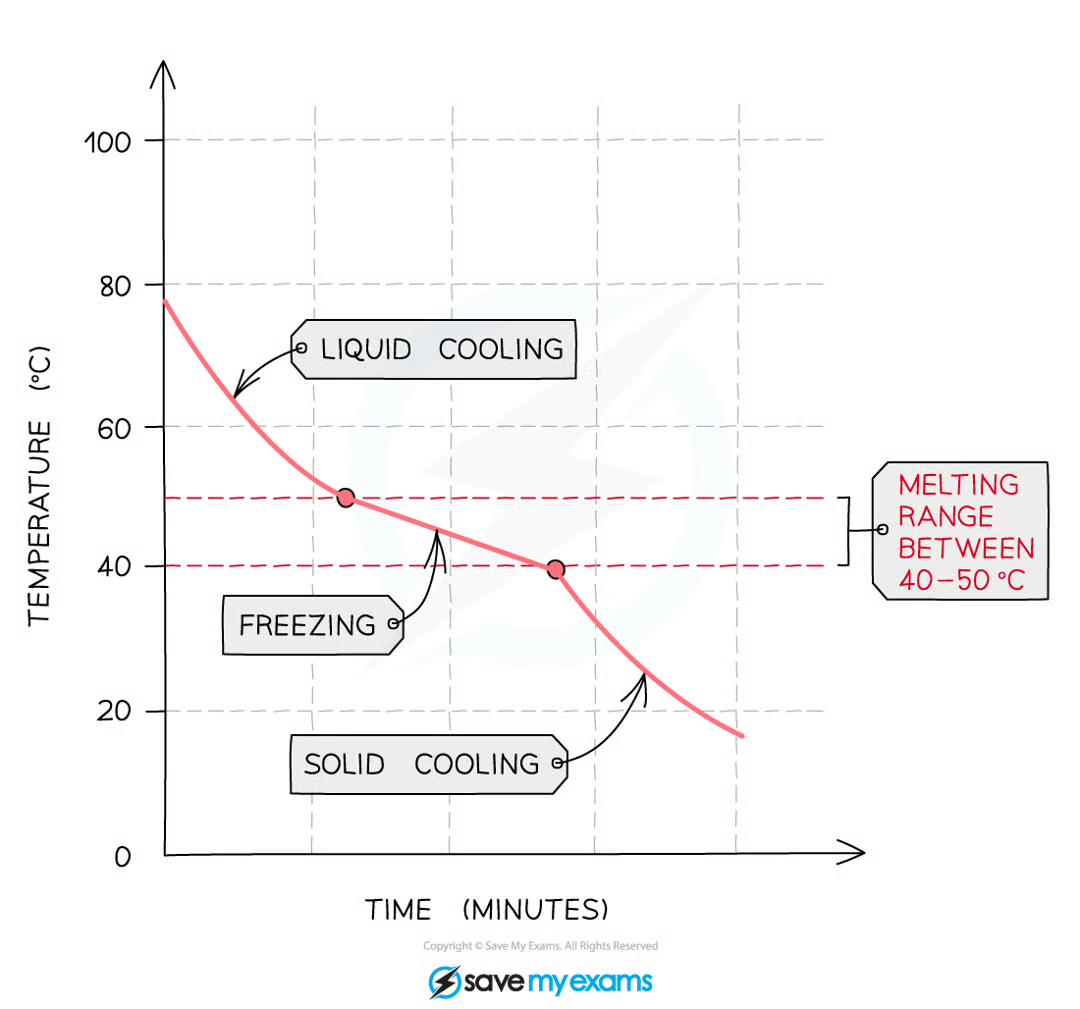
What does an impure substance look like on a cooling curve
An impure sample of the compound would produce a gradual decrease in temperature as it freezes as shown in the graph below
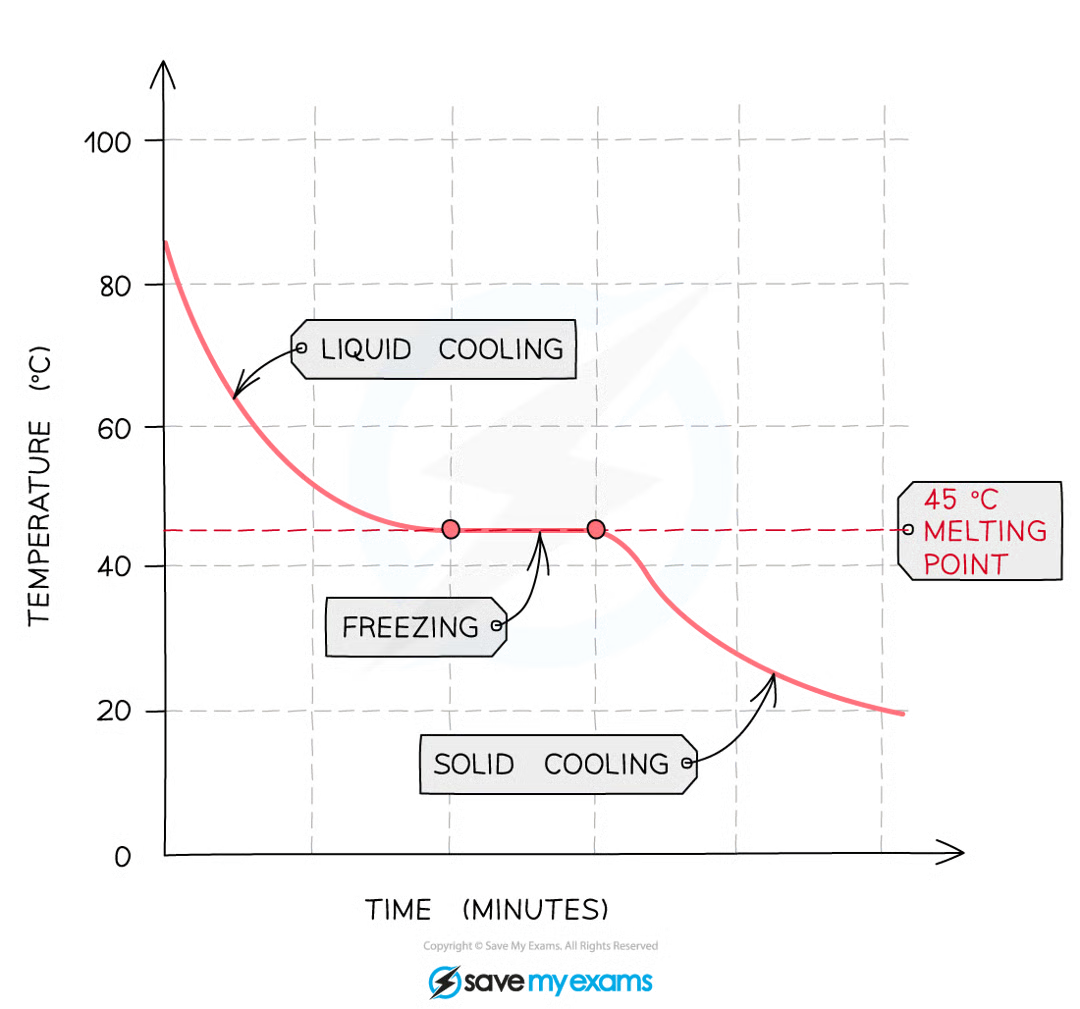
What does a pure substance look like on a cooling curve
The horizontal part of the graph shows that the compound has a sharp melting point, so the compound is pure
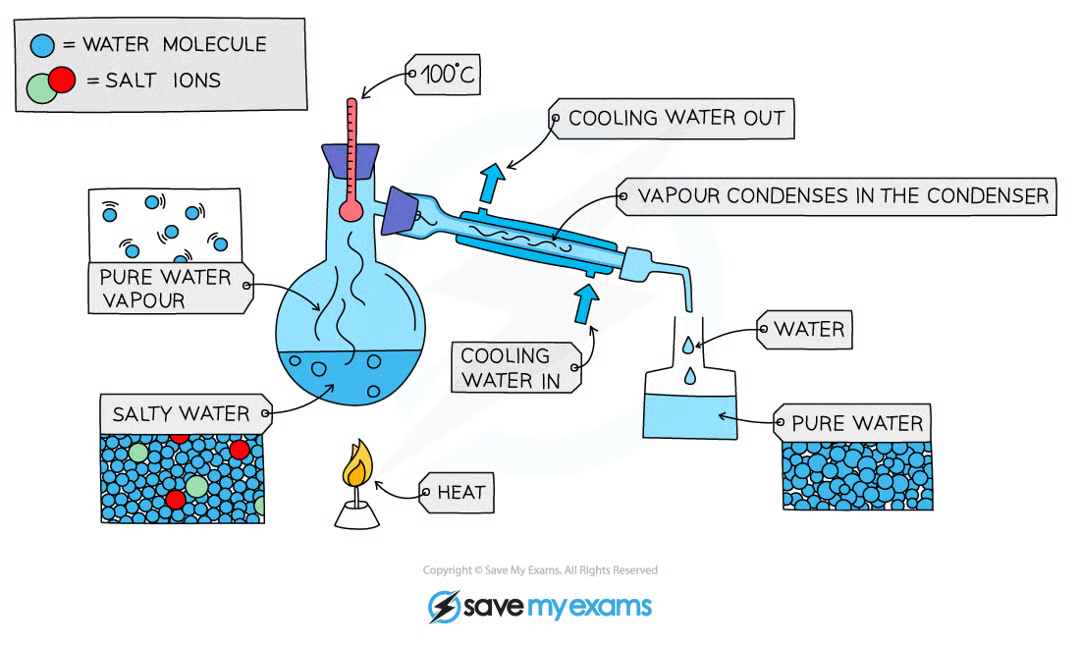
What is simple distillation used for?
To separate a liquid and soluble solid from a solution
Why does simple distillation work?
Because there is a difference in boiling points
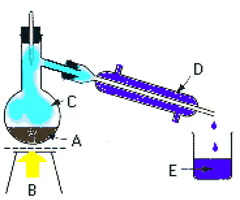
Label the apparatus for distillation
A - round bottom flask
B - a heat source, usually a Bunsen Burner
C - evaporated solvent
D - condenser with condensing jackets(water going out at the top and water coming in at the bottom)
E - distillate
Describe the stages of simple distillation
Stage 1 - Set up equipment.
Stage 2 - Place and heat the mixture in a distillation flask.
Stage 3 - The liquid will boil to become a gas.
Stage 4 - As the solvent vapourises, it rises up and leaves the solute behind
Stage 5 - The vapourised solvent enters the condenser
Stage 6 - The cold water around the condenser will make the steam condense back into liquid form.
Stage 7 - Collect the distillate
miscible
Describes two or more liquids that are completely soluble in each other
What is fractional distillation used for?
two or more liquids that are miscible with one another (e.g., ethanol and water from a mixture of the two)
What is a fractionating column?
A vertical column used in fractional distillation to separate liquids based on their boiling points.
How does a fractionating column work?
The lower melting point liquid rises to the top while the higher melting point liquid sinks, due to multiple condensers at different levels cooling the vapors at different rates.
What is simple distillation weak in that fractional distillation excels in?
Separating components of mixtures with similar boiling points
anti-bumping granules
Added to the round bottomed flask during distillation to spread the heat
Uses of fractional distillation in real life
separate crude oil
make alcoholic drinks such as vodka and whisky, hence the name a brewing distillery
separate elements of air
What is filtration used for?
to separate an undissolved solid from a mixtures of the solid and a liquid/ solution
How does filtration work?
A piece of filter paper is placed in a filter funnel above a beaker
A mixture of insoluble solid and liquid is poured into the filter funnel
The filter paper will only allow small liquid particles to pass through as filtrate
Solid particles are too large to pass through the filter paper so will stay behind as a residue

filtrate
the liquid that passes through the filter paper in filtration
residue
Solid left in the filter paper after filtration
solubility
a measure of how much solute can dissolve in a given solvent at a given temperature.
Why might we crush the solute to increase solubility?
When a substance dissolves, its particles are broken apart by the solvent particles, if we crush the solute then the solvent has less work to do
solubility factors
surface area, agitation(moving the solute to improve contact with the solvent particles), temperature, pressure
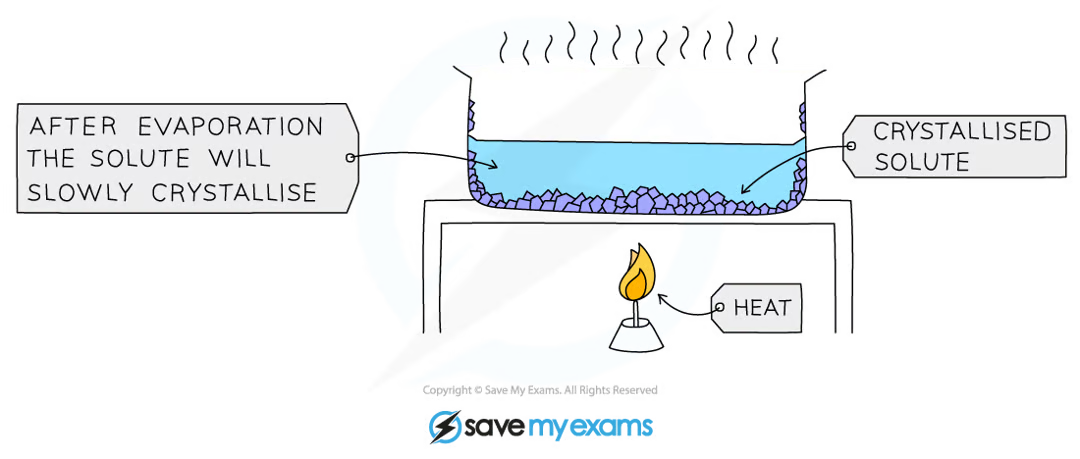
What is crystallisation used for?
Used to separate a dissolved solid from a solution, when the solid is much more soluble in hot solvent than in cold (e.g., copper sulphate from a solution of copper (II) sulphate in water)
How are solute crystals obtained via crystallisation?
The solution is heated, allowing the solvent to evaporate, leaving a saturated solution behind
Test if the solution is saturated by dipping a clean, dry, cold glass rod into the solution
If the solution is saturated, crystals will form on the glass rod
The saturated solution is allowed to cool slowly
Crystals begin to grow as solids will come out of solution due to decreasing solubility
The crystals are collected by filtering the solution, they are washed with cold distilled water to remove impurities and are then allowed to dry
What is paper chromatography used for?
to separate substances that have different solubilities in a given solvent (e.g., different coloured inks that have been mixed to make black ink)
What is a pencil line used for in paper chromatography?
To place spots of the sample on it
Why is a pencil line used on chromatography paper?
Pencil is used for this as ink would run into the chromatogram along with the samples
Why does the pencil line need to sit above the solvent level?
so the samples don’t wash into the solvent container
How does the solvent travel up the paper?
Via capillary action, basically it moves up even with gravity acting on it
Why will the substances spread apart in chromatography?
Different substances have different solubilities so will travel at different rates, causing the substances to spread apart
Which substances spread the furtthest?
Those substances with higher solubility will travel further than the others
What would pure substances look like on a chromatogram?
Pure substances will produce only one spot on the chromatogram
What would mixtures and impure substances look like on a chromatogram?
If the substance is a mixture, it will separate on the paper to show all the different components as separate spots
An impure substance therefore will produce a chromatogram with more than one spot
What will 2 identical substances look like on a chromatogram?
Both chromatograms produced will be identical
What are Rf (retention values) used for?
To identify the components of mixtures by comparing the Rf values of unknown substances in chromatography to substances with known Rfs to see if they match in Rf value
Rf value equation
distance travelled by substance ÷ distance travelled by solvent
What is the scale of Rf values?
0-1
What can be said about a substance with an Rf value close to 1?
the closer it is to 1, the more soluble is that component in the solvent
CP: Investigating Inks - separating the inks
Ink consists of a solvent which has different dyes dissolved in it
The investigation must thus include analysis of both the solvent and the dyes used
The solvent must first be separated which can be done by simple distillation
Solvents tend to have low boiling points than the dyes which tend to be more viscous so it will evaporate first.
Add anti-bumping granules and heat gently so as not to go past the boiling point
Record the temperature of boiling point
CP: Investigating Inks - paper chromatography materials
A 250 cm3 beaker
A wooden spill
A rectangle of chromatography paper
Four known food colourings labelled A–D
An unknown mixture of food colourings labelled U
Five glass capillary tubes
Paper clip
Ruler & pencil
CP: Investigating Inks - Method of chromatography
Use a ruler to draw a horizontal pencil line 2 cm from the end of the chromatography paper
Use a different capillary tube to put a tiny spot of each colouring A, B, C and D on the line
Use the fifth tube to put a small spot of the unknown mixture U on the line
Make sure each spot is no more than 2-3 mm in diameter and label each spot in pencil
Pour water into the beaker to a depth of no more than 1 cm and clip the top of the chromatography paper to the wooden spill. The top end is the furthest from the spots
Carefully rest the wooden spill on the top edge of the beaker. The bottom edge of the paper
should dip into the solvent
Allow the solvent to travel undisturbed at least three quarters of the way up the paper
Remove the paper and draw another pencil line on the dry part of the paper as close to the wet edge as possible. This is called the solvent front line
Measure the distance in mm between the two pencil lines. This is the distance travelled by the water solvent
For each of food colour A, B, C and D measure the distance in mm from the start line to the middle of the spot
The Rf values of food colours A, B, C and D should be compared to that for the unknown sample as well as a visual comparison being made
In the UK, where is drinking water sourced from?
rivers
lakes
underground aquifers
potable water
water that is safe to drink
What happens to water in screening and sedimentation?
Water first passes through a sieve to remove large debris
In sedimentation, larger insoluble particles sink to the bottom
In sedimentation, why might chemicals such as iron sulfate or aluminium sulfate be added?
To help smaller particles clump together and settle
What happens in water filtration?
Water is filtered through beds of sand and gravel to remove remaining small solid particles
What happens in water chlorination?
Chlorine is added to kill harmful bacteria and other microorganisms that can’t be removed by filtration.
This helps prevent diseases such as cholera or typhoid
How do you make sea water potable?
Since it contains mainly salts, simple distillation can be used to separate the water and the salts
Why is making seawater potable expensive?
Lots of energy is required to heat the large volumes of water to 100 degrees Celsius
What must happen to the wastewater produced in making seawater potable?
It must be disposed of correctly as the wastewater is extremely toxic due to the very high concentration of salts.
deionised water
water that has had metallic ions such as calcium or copper removed
How does water deionisation work?
Deionisation uses specifically designed ion-exchange resins that remove ions by exchange with hydrogen and hydroxide ions in water, which then recombine to form water molecules
Why is deionised water used in chemical investigations
Deionised water is used as the ions could react with the substances under analysis and would give the experiment a false result