BCH 100: Biochemistry and the Role of Water
1/67
Earn XP
Description and Tags
These flashcards cover key concepts and definitions related to biochemistry, specifically focusing on the properties and roles of water in biochemical processes.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
Biochemistry
The study of chemical processes related to living organisms.
Universal Solvent
Water's ability to dissolve and interact with a wide variety of substances.
Polarity
The property of a molecule having a positive charge on one side and a negative charge on the other, as seen in water.
Hydrogen Bond
An electrostatic interaction between the partial negative charge of an oxygen atom and the partially positively charged hydrogen of another water molecule.
Hydrogen bonds readily form between an electronegative atom (the hydrogen acceptor) and a hydrogen atom covalently bonded to another electronegative atom (the hydrogen donor)
• In the donor, the hydrogen must be attached to a very electronegative atom – e.g. N, O, or F
Directionality: strongest when the acceptor atom is in line with the covelant bond betweent he donor atom and H
Hydrophilic
Describes compounds that dissolve easily in water; generally charged or polar compounds.
Hydrophobic
Nonpolar molecules that do not dissolve in water; also described as water-fearing.
Amphipathic
Molecules that contain regions that are both polar (or charged) and nonpolar.
Hydrophobic Effect
The tendency of nonpolar regions to cluster together in water, maximizing interactions among polar regions and with the solvent
Bond Energy
The average amount of energy required to break a bond.
Formation: When a bond forms, energy is released.
Breakage: When a bond breaks, energy is absorbed
Cohesion
The attraction between molecules of the same substance, such as water molecules sticking to each other.
Adhesion
The attraction between molecules of different substances, allowing water to stick to other surfaces.
Hydrolysis
A cleavage process accompanied by the addition of water.
Condensation
A reaction where the elements of water are eliminated as a product.
Micelle
A thermodynamically stable structure of amphipathic compounds in water.
Noncovalent Interactions
Interactions among biomolecules that are much weaker than covalent bonds, including hydrogen bonds and van der Waals interactions.
Weak Interactions
Interactions that are continually forming and breaking, but when accumulated, can significantly stabilize structures like DNA and proteins.
Ex:
Hydorgen Bonds
Ionic Interactions
Hydrophobic interations
van der Waals interactions
Importance of Water
Universal Solvent:
– dissolve and interact with a wide variety of substances
• Chemical Reactions:
– It participates in many biochemical reactions
• Temperature Regulation:
– high specific heat capacity* helps maintain a stable temperature in organisms
*requires lot of energy to change the temperature
• Transportation:
– primary component of blood and other bodily fluids
– facilitates transport of nutrients, hormones, and waste products
• Structural:
– It contributes to the 3D structure of proteins and nucleic acids
• Catalysis:
– Water often acts as a reactant or a product in enzymatic reactions
Properties of Water
Water is a polar molecule
– Polar covalent bonds
• Unequal sharing of electrons b/w O and H
• Creates a charge across the bond (dipole)
– Hydrogen bonding !!!
– Universal solvent
high specific heat
melting and boiling point;
excellent solvent - dissolves
polar and charged
(hydrophilic) molecules
Water Can Be Essential to Protein Function
H2O molecules are often found to be bound so tightly to biomolecules that they are part of the crystal structure
• For many proteins, associated water can have a specific function:
✓ example – cytochrome f – five H2O molecules provide path for protons to move through the membrase
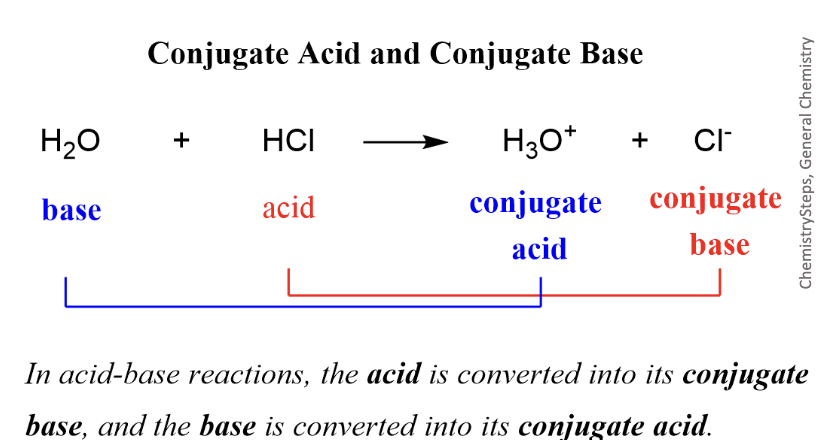
Brønsted-Lowry definition
Acids are proton donors and bases are proton acceptors
Lewis definition
Acids are electron pair acceptors and bases are electron pair donors
Conjugates
Species that differ by only one hydrogen
Strong vs Weak Acids
Compounds have different tendency to ionize
• Strong acids and bases dissociate completely in solution
• Weak acids and bases only partially dissociate.
Henderson-Hasselbalch Equation
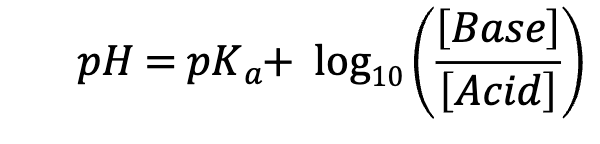
pH = pKa so [Base] = [Acid]
pH < pKa so [Base] < [Acid]
pH > pKa so [Base] > [Acid]
Titration Curve
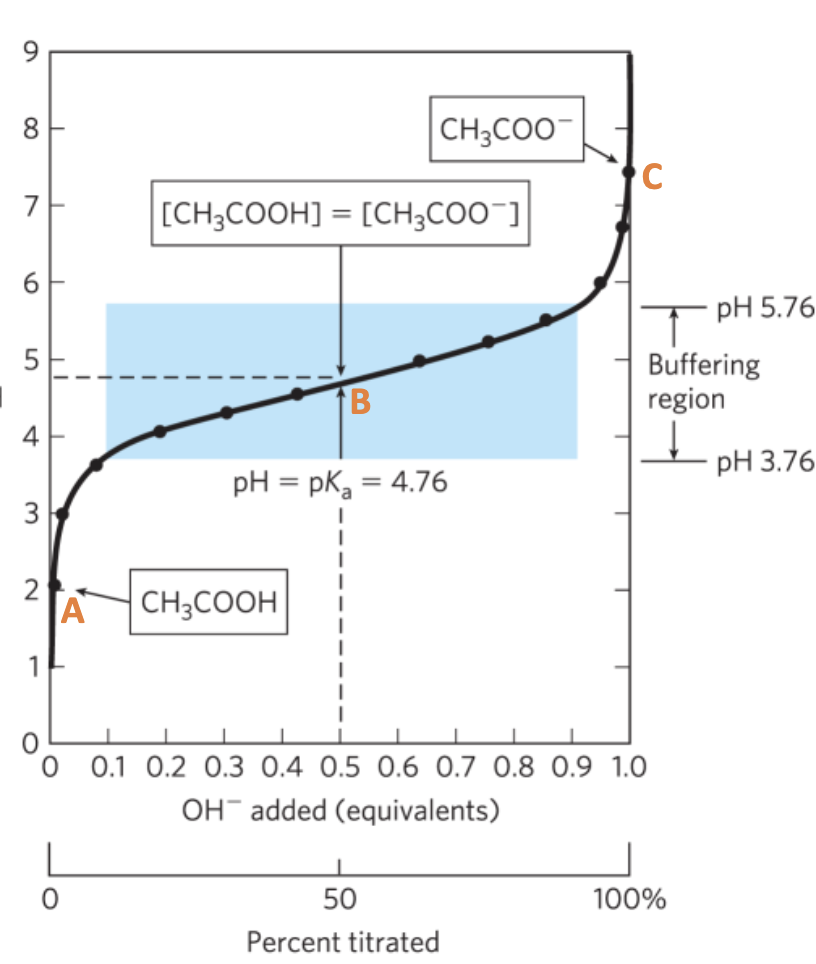
A. Initial Point:
Mainly HA, with some conjugate base (A-) depending on the acid dissociation
B. ½ equivalence point:
[HA] = [A-]
pH = pKa ← HH Equation
C. Equivalence point
all conjugate base A-
pH>pKa
Buffers
prevent extreme changes in pH upon addition of a small amount of base or acid.
Weak acid or base, and its conjugate, can have buffering property
• The buffering region is surrounding the pKa approx. +/- 1 pH, where relatively big changes in OH- addition do lead to mild changes in the pH
Buffer Capacity
The ability of a buffer to resist changes in pH.
Proportional to the [weak acid] and [conjugate base] (or vice versa)
• The greater the concentration - [HA] and [A-], the greater the buffer
capacity (the more effective the buffer!)
• i.e. 100 mM Tris-HCl buffer has a greater capacity than a 50 mM Tris-
HCl buffer
• The best buffering capacity is when pH = pKa
Buffers in Biological Systems
The phosphate buffer system acts in the cytoplasm of
all cells:
H2PO4− ⇌ H+ + HPO42−
The pH of blood is buffered by the conjugate acid-base
pair of carbonic acid and bicarbonate (H2CO3/HCO3–).
called the bicarbonate buffer system
hypoventilation increases blood CO2 (retention) and lowers blood pH
• hyperventilation raises blood pH the body has that can sense pH and, through the brain stem, can quickly adjust breathing to maintain pH –response within mins
acidosis = pH of blood plasma below the normal value of 7.4 common in people with severe, uncontrolled diabetes.
alkalosis = pH of blood plasma above the normal value of 7.4
Structure of α-Amino Acids
α-Amino Acids
• Amino group (-NH3+)
• Carboxylic acid group (-COO-)
• α-carbon is called the carbon attached to a key functional group, commonly carbonyl in o-chem; here the amino group is attached to the α-C, so α-amino acids
zwitterionic form
at physiological pH (~7), the amine is protonated (NH3+) and the carboxyl group is deprotonated (COO-)
D- configuration
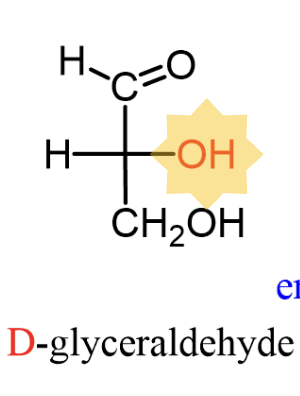
bottom OH is on right side
L-configuration
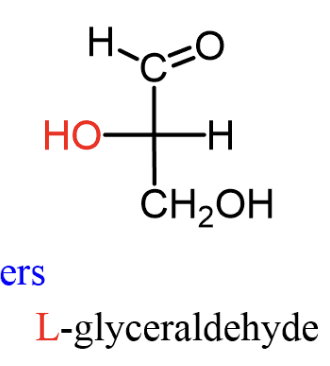
bottom OH is on left side
Chirality of Amino Acids
Amino acids are naturally L-configuration
Chirality of Carbohydrates
Carbohydrates are naturally D- configuration
Amino Acids Can Act as Acids or Bases
the alpha- amino group and the alpha-carboxyl group can be proton donor/acceptors => act as Lowry acids/bases
7 of the 20 amino acids have readily ionizable side chains. They are able to form ionic bonds, and to donate or accept protons (called acid–base catalysis) to facilitate reactions.
Proteins are made of
amino acids
Proteins are synthesized as linear polymers of amino acids in the ribosome
• The sequence of the amino acids in the protein chain is determined by the sequence of the corresponding gene (DNA); protein sequences involves mRNA intermediate
DNA -> RNA -> protein
polypeptide chains
linear polymers of amino acids in the ribosome
What determines the folding of proteins
multiple factors, importantly, weak interactions
– hydrogen bonds, hydrophobic effect, etc.
Primary Protein Structure
Polypeptide chain
Secondary Protein Structure
alpha-helix
beta-(pleated) sheet
Secondary (2°) structure refers to local areas of repeating 3D structure
• Held together with hydrogen bonds in the backbone between the amide nitrogens (N-H) and the carbonyl (C=O) carbons
Tertiary Protein Structure
The final folded version of 1 chain
tertiary (3°) structure refers to the spatial arrangement of the secondary structural elements in the polypeptide chain
important interactions:
• Noncovalent, weak interactions
• Hydrogen bonding
• α-carbonyls and α-amides
• Rs that can hydrogen bond (which?)
• Hydrophobic – nonpolar side chains
• Electrostatic – attraction or repulsion of charged Rs
• prosthetic groups (such as heme or metal
ions) – additional compounds embedded
into the proteins
• disulfide bridges – covalent bonds between Cysteines
Quaternary Protein Structure
Assembly of several chains (each is called subunit)
Quaternary (4°) structure refers to the spatial arrangement of multiple polypeptide chains to form multi-subunit complexes; Not all proteins have quaternary structure!
• primarily held together by non-covalent bonds, S-S bonds can participate
globular proteins = folded into a spherical or globular shape
fibrous proteins = arranged in long strands or sheets
Peptide bonds
chains of amino acids joined by amide bonds
These bonds are called PEPTIDE BONDS
Peptide bonds connect the α-carboxyl group of one amino acid to the α-amino
group of the next amino acid.
• Peptide bonds are covalent bonds formed through condensation and broken
through hydrolysis; this reaction is catalyzed in the ribosome by the rRNA
Rotation around Bonds in a Polypeptide Backbone
Because the bond between the carbonyl carbon and the (the peptide bond) has a partial double bond character (resonance), the atom positions in the amide plane are basically fixed
The alpha-Helix is a Common Protein Secondary Structure
simplest arrangement, maximum number of H- bonds
– R groups protrude out from the
backbone
– each helical turn = 3.6 residues,∼5.4 Å
– H bonds are nearly parallel to the helix axis
– C=O to N-H four residues away
– often with distinct hydrophilic and hydrophobic faces
– Entire helix has a dipole
– Most are
Held together with hydrogen bonds in the backbone between the amide nitrogens (N-H) and the carbonyl (C=O) carbons
alpha-helix disruption
Several things can disrupt α-helices
• Proline: rigid cyclic structure ⟶ “helix breaker”
• Prevents rotation
• α-amino group not available for
hydrogen bonding
• Strong electrostatic repulsion between R groups with the same charges, i.e., Lys and Arg (+) or Asp and Glu (-)
• Steric hinderance of bulky R groups, Like Ile, Thr
Overall:
• charge, size, and shape of R chains candestabilize
• formation of ion pairs and hydrophobic effect can stabilize
Beta Pleated Sheets
Formed between adjacent parts of a polypeptide chain
• These are very extended compared to the compact helix
• side chains alternate sides of the sheet
• stabilized by interchain H bonds (C=O and N-H)
– Perpendicular to the direction of the sheet.
Beta Sheet can be ___ or ___
Antiparallel or Parallel
antiparallel = opposite orientation
– more common
– N→C, C →N, N →C
parallel = same orientation
– All N→C
Super-secondary Structures
Also called Motifs: combo of secondary structures
Common examples:
• Leucine Zipper:
2 α-helices that bind to DNA
Has Leucine every 7 residues
• Helix-Turn-Helix:
2 α-helices separated by a turn
Insulin: example of processing and cross-chain bridges
Insulin is a complex peptide hormone held together by disulfide bonds.
• It is synthesized in the ER as precursor protein with disulfide bonds
• Then the middle portion is cleaved, and two remaining chains – A and B –
remain linked thanks to some of these bonds
Domains
domain = part of a polypeptide chain that is independently stable or could undergo movements as a single entity
• domains may appear as distinct or be difficult to discern
• small proteins usually have only one domain
• Larger proteins often contain two or more distinct “domains” of compact folded structure
• A domain frequently possesses some defined function (e.g., D N A recognition, oligomerization, cofactor binding, etc.)
Intrinsically disordered regions
part of the polypeptide chain that does not fold into a secondary structure
• Provide flexibility; can act as flexible scaffolds; target for modifications, etc.
Determining Protein Structures
Structural biology = study of the 3D structures of biomolecules (proteins, nucleic acids, lipids,
carbohydrates) to understand function; uses biochemical, biophysical, and computational
methods
• X-ray crystallography: X-rays diffract from a protein crystal; Diffraction patterns are mathematically
reconstructed into a 3D structure. It provides high-resolution atomic detail. Limitations: requires crystallization;
not the same environment as in the cell; less suitable for flexible or disordered proteins; no information about
the movements; data quality depends on crystal order
• NMR spectroscopy: Provides insight into protein dynamics and flexibility; Limitation: best for small
proteins
• Modern advances:
– Cryo-EM enables structure determination without crystallization
– Computational prediction (e.g., AlphaFold) complements experimental methods
Protein Denaturation
Loss of the structure (2°, 3°, 4°, or a combo) that gives a protein its biological activity; the loss of structure leads to loss of function
• Denaturation (unfolding) can happen due to:
• Heat
• Large changes in pH
- alters charges on side chains, -COO- to -COOH or –NH3+ to –NH2
• Detergents (soaps!) disrupt hydrophobic interactions
• Urea or guanidine decrease hydrophobic effect
• Reducing agents such as β-mercaptoethanol reduces
disulfide bonds in proteins
Is denaturation reversible?
In principle, denaturing does NOT affect primary structure
• Anfinsen hypothesis (in the 1950s) – the entire information required for a protein to fold into its correct three- dimensional structure is encoded solely within its primary amino acid sequence
Intrinsically disordered proteins (IDPs)
do not have a defined structure under physiological conditions until they interact with other molecules.
• Metamorphic proteins exist in an ensemble of structures of approximately equal energies that are in equilibrium.
Protein Misfolding and Aggregation Are Associated with
Some Neurological Diseases
Prion diseases
Some infectious neurological diseases are caused by infectious proteins called prions. Prions exist in two states
• one α-helix-rich (PrPC)
• β-sheet-rich (PrPSC)
• PrPSC forms aggregates that disrupt cell
function.
• Template-Driven Misfolding – the “SC” form
binds and converts the native form into SC
Human PrP protein – mutations can cause
different inherited prion diseases e.g.
Creutzfeldt-Jakob Disease (CJD)
Fibrous Proteins are Specialized for a Structural Function
overall linear shape: do not fold back on themselves
• give strength and/or flexibility to structures
• simple repeating element of secondary structure
• H2O insoluble due to high concentrations of hydrophobic residues
The Structure of α-Keratin in Hair
α-keratin helix is a right-handed α helix
• two strands of α-keratin, oriented in parallel, wrap about each other to form a supertwisted coiled coil
– supertwisted helical path is left-handed
The Structure of Collagen
Each polypeptide chain = left-handed helix, not an α-
helix
• Single helix: procollagen
• Single helix not formed by H-bonds, but Pro steric repulsion
• 3 procollagens ⟶ 1 tropocollagen
• Three strands held together by H bonding
• Every 3rd amino acid is Gly
• Repeating sequences are X-Pro-Gly and X-Hyp-Gly
• X = any amino acid
• Hyp = hydroxyproline <- modified Proline (a PTM); helps
H boinding
Collagen Deficiencies
Osteogenesis imperfecta
• brittle bone disease
• Genetic – caused by mutation, where Gly→ another amino acid.
• Scurvy
• Occurs when vitamin C is deficient; (GULO gene, which codes for the enzyme needed to make vitamin C, was lost in primate ancestors around 60-70 MYA)
• Vit C is required for the synthesis of Hyp, which is important for collagen stability
• The unstable collagen causes a range of symptoms
• The Scourge of the Seas: “In 1499, Vasco da Gama lost 116 of his crew of 170; In 1520, Magellan lost 208 out of 230; ... all mainly to scurvy” (from Wikipedia) Researcher estimated over 2 million sailors died of scurvy over the centuries…
Globular Proteins
Proteins that are folded to a spherical (Glob) shape
• Shape based on direction and 2° structure folding
• Usually have many α-helices and β-sheets
but can also have just α-helices or β-sheets only, depending on the protein
Porins
Found in outer mitochondrial membrane (eukaryotes)
• Outer membrane (prokaryotes)
• β-barrel (no α-helices)
• Antiparallel β-sheet with 16-18 strands in a cylinder
Exterior has hydrophobic residues
• Interact with lipid core of membrane
• Interior has hydrophilic residues
• Water, ions, and small molecules can pass