Addition Polymer ( hydration of ethene )
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
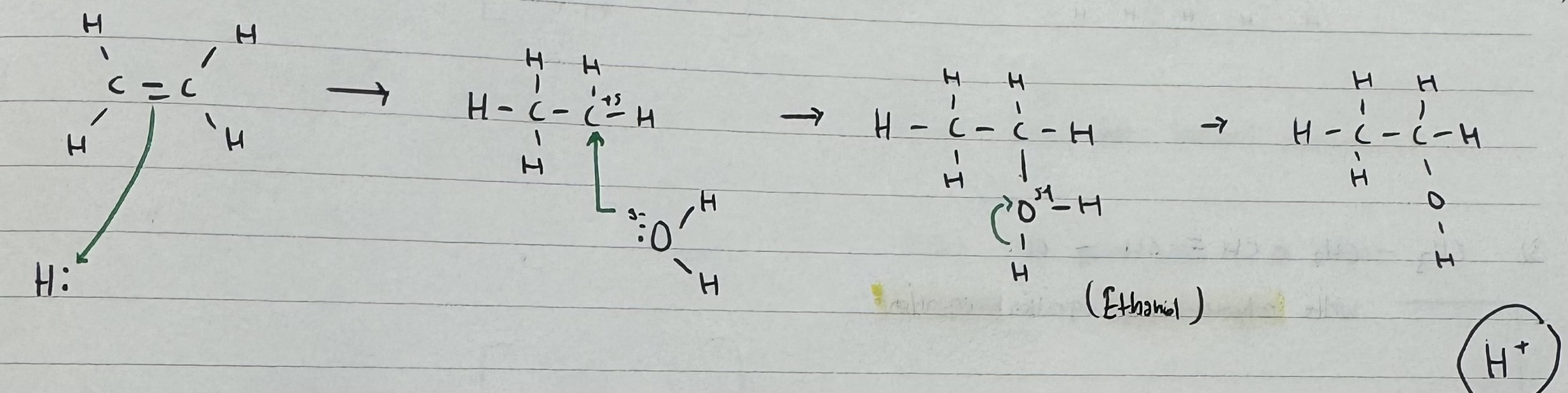
What is the product when Alkene reacts with steam ?
Alkene + steam → Alcohol
What catalyst is used in this reaction ?
Using H3 PO4
H + + H2 PO4 —> H3 PO4
What are the conditions for this reaction ?
H3 PO4 Catalyst
Temp : 30 *C
Pressure : 60 atm
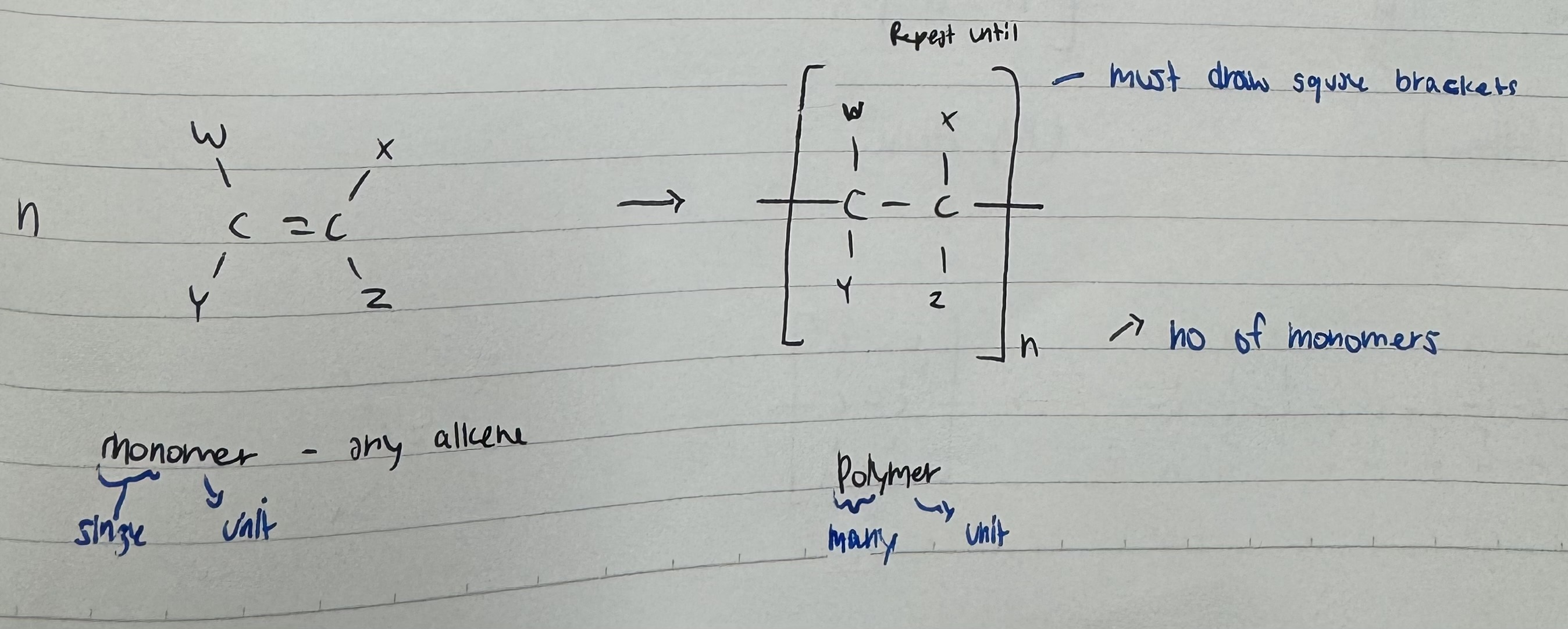
Addition Polymers
Polymers :
BIG molecules , produced from tiny monomers
Made from monomers with Alkenes
We polymers are polymerase , the C = C double bind will open up and form a big chain of C - C
What are the conditions for High Density Polyethene ?
Temp : 60 *c
Pressure : 1 atm
Zeigier Natta Catayst
What does high density polyethene produces ?
Polymers with little branching
Chains pack together well - so high density and higher MPT
Uses in milk bottles and buckets
What are the conditions for low density polyethene ?
Temp : 200 *c Pressure
Pressure : 2000 atm
Free radical substitution
What does low density polyethene produces ?
Large chain - branched polymers
Chains don’t pack well - flexible / low density
Use : plastic bags / cable insulation
Recycling / Mechanical
Separate different types of plastics
Wash the plastic
Melted / remoulded
Recycling / Feedstock
Heat plastic to break polymer bonds
Produces monomers
What is plasticizsers ?
Small molecules set in between polymer chains
Allow chain to side easier
Make polymers more flexible