Final review/reminders
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
Fill in the blanks in the table:
State | Density | Shape | Volume |
Gas | |||
Liquid | |||
Solid |
State | Density | Shape | Volume |
Gas | Low | Container | Container |
Liquid | High | Container | Definite |
Solid | High | Definite | Definite |
How to know which ion is bigger?
More shells + more protons = bigger ion
(same number of electrons → same number of shells, so look at how many protons)
Electronegativity values for C, N, H, O, F, B
C: 2.5
N: 3.0
H: 2.1
O: 3.5
F: 4.0
B: 2.0
Which state(s) of matter can be compressed?
ONLY gas
Sublimation
Solid → gas
Deposition
Gas → solid
Molecules with stronger intermolecular forces tend to have what kind of boiling points?
Higher boiling points
Which intermolecular force is stronger?
Adenine and Thymine with 2 H-bond donors and acceptors
Guanine and Cytosine with 3 H-bond acceptors and donors
Both had H-bonds so the forces are equivalent
Guanine and Cytosine with 3 H-bond acceptors and donors
Rank the types of intermolecular forces from weakest (1) to strongest (4)
Hydrogen bonding
3
Dipole-Dipole
2
Ion-Dipole
4
London Dispersion Forces
1
Hydrogen bond
an unusually strong dipole-dipole interaction
intermolecular force
occurs between an H atom in a polar bond (usually N-H, O-H, F-H), and an electronegative atom (usually O, N, F)
Is vaporization endothermic or exothermic?
Endothermic
Heat of vaporization
Enthalpy change needed to evaporate 1 mol of a liquid at its boiling point
Heat of vaporization equation
ΔH vapor = H vapor — H liquid
Is condensation exothermic or endothermic?
Exothermic
Condensation equation
ΔH vaporization = —ΔH condensation
Evaporation vs condensation
evaporation: liquid → gas
Condensation: gas → liquid
dynamic equilibrium
When the rates of evaporation and condensation are equal → leads to stable vapor pressure
Le Chatelier’s principle
When a system in dynamic equilibrium is disturbed, vapor pressure adjusts to minimize/counteract the disturbance
Boiling point
Temperature where vapor pressure is equal to external pressure (gas above liquid)
Normal boiling point
Temperature when a liquid boils, when the external pressure is 1 atm
Vapor pressure and temperature relationship
Directly proportional
Vapor pressure and intermolecular force relationship
inversely proportional
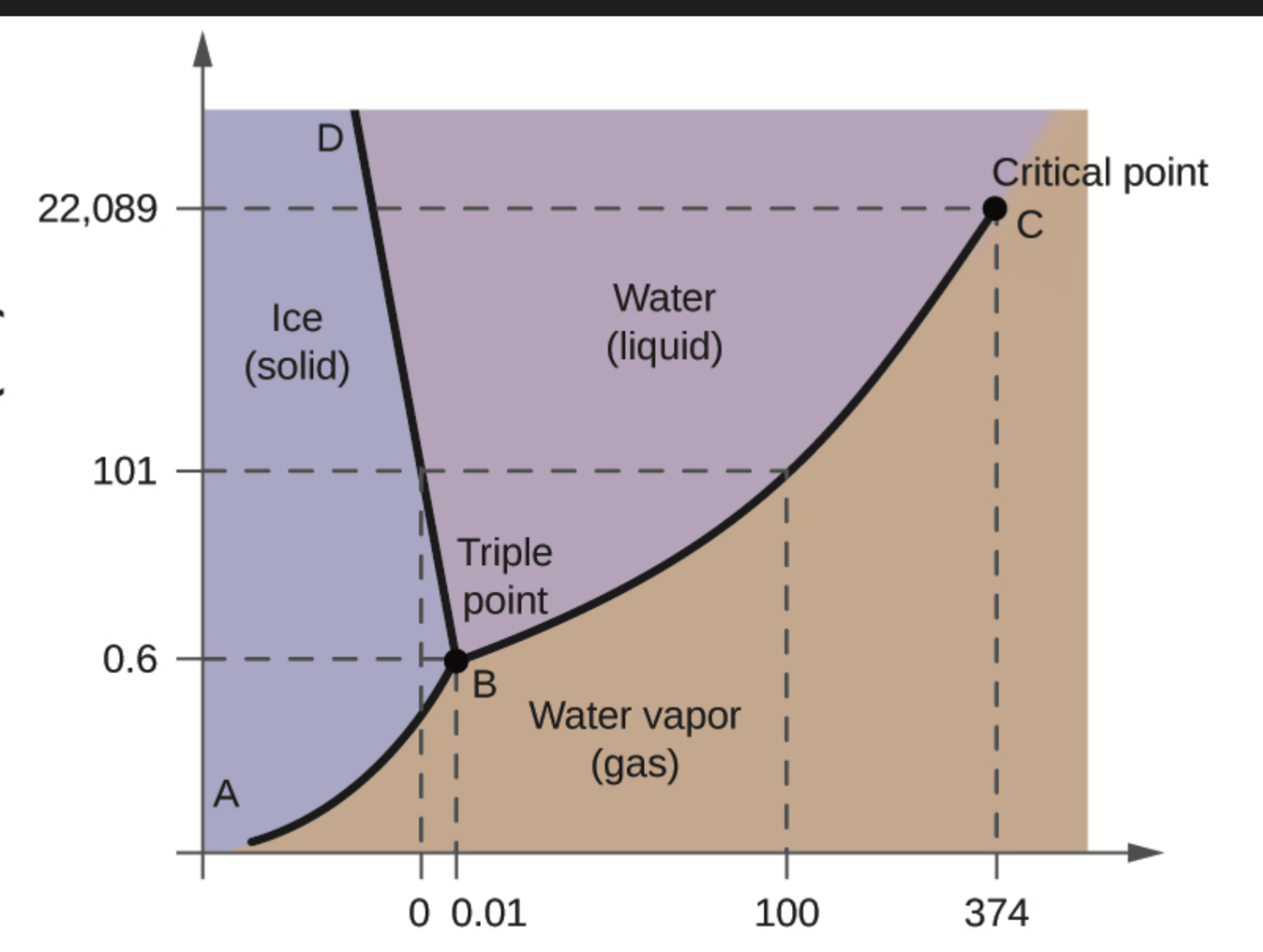
Critical point
At a certain temperature, the interface between a liquid and a gas disappears creating a supercritical fluid (not quite liquid, not quite gas)
Critical temperature + symbol
Tc
The temperature above which a gas cannot liquify
Critical pressure
Minimum pressure required to liquify a gas at its critical temperature
Heat of fusion
Enthalpy change needed to melt 1 mol of a solid at its freezing point
Heat of fusion equation
ΔH fusion (melting) = H liquid — H solid
Which one is bigger: heat of fusion or heat of vaporization?
Heat of vaporization
Molar heat of sublimation
enthalpy change needed to sublime (solid →gas) 1 mol of a solid
Molar heat of sublimation equation (Hess’s law)
ΔH sublimation = ΔH melting/fusion — ΔH vaporization
Vaporization
liquid → gas (aka boiling)
Pressure and density relationship
directly proportional
Intermolecular force and melting point and lowering point relationship
directly proportional
dipole-dipole strength and boiling point relationship
directly proportional
Is the molar mass of water high or low?
High
Which conditions must be true for gases to behave ideally?
Low pressure and High temperature
You begin with a 3.5L gas sample at 120ºC and 1.4atm. You increase the pressure to 4.7 atm and then cool the sample to 50ºC. What is the final volume of the sample?
Use combined gas law: PV/T = P2V2/T2
Match the following terms to their appropriate units
Molar Mass
Density
Molar Density
Molar Mass
grams/mols
Density
grams/mL
Molar Density
mols/L

Practice
Solve for molar mass
Molar mass = (d)(R)(T)/(P)
combined gas law:
PV/T = P2V2/T2
molar mass equation:
dRT/P
How to find limiting reactant?
Calculate n with ideal gas law. the smaller n is the limiting reactant, and the larger is the excess.

How to solve?
To find how many gases are left:
Find the limiting reactant
Calculate n with ideal gas law. the smaller n is the limiting reactant, and the larger is the excess.
Find the excess reactants (the one that is not limiting)
the product is also excess
left over: the product and excess reactant (ammonia and nitrogen)

How to solve?
The logic is that because there is not an equal amount of H2 and N2 available, you cannot equally use both of them to make 2NH3.
You need to find the limiting reactant, then set up a ratio using ONLY that reactant. The reason is because there is an excess reactant, you don’t need to worry about whether it will run out or not.
The limiting reactant is H2 (Calculate n with ideal gas law. the smaller n is the limiting reactant, and the larger is the excess.)
The excess is N2
3 mol H2/2 mol NH3 = 0.07 mol H2/?mol NH3 → then you solve it (you don’t include N2 in the ratio bc it is excess, so you don’t need to worry about it running out)

How to find mol fraction?
mol of molecule A/total mols
How to find partial pressure?
(n of substance A)(RT)/V

Solve
The logic of this problem is that because H2 is a limiting reactant, its mol fraction AFTER the reaction is 0 because it ran out. therefore, you compare the mol fraction of ammonia to nitrogen.

Solve
The logic of this problem is that equilibrium means the gases will mix, so you add the volumes together. Also, you will need to calculate a new number of mols for the excess reactant bc it is getting ‘eaten up”, bc when the gases mix the number of mols changes bc the reaction is taking place.
you do this by: 0.07 H2 (from calculation) /3 H2 (from equation) = 0.0245 mol N2 used
So, 0.22-0.0245 mol = 0.1962 mol N2 (the ACTUAL mol value you use)

kinetic molecular theory of gas
molecules have finite mass but no volume
molecules are neither attracted to nor repulsed by others
which conditions increase gas molecules attraction to each other?
low temp
high mass
high intermolecular forces
Van der waals equation
image
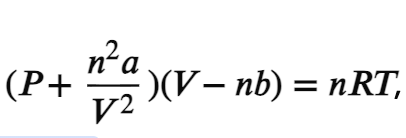
What is equal to 1J?
0.239 calories, 1kgm²/s²
Is density intensive or extensive?
Intensive
Heat
A way of exchanging energy that leads to a change in temperature
Thermal energy
The energy associated with temperature
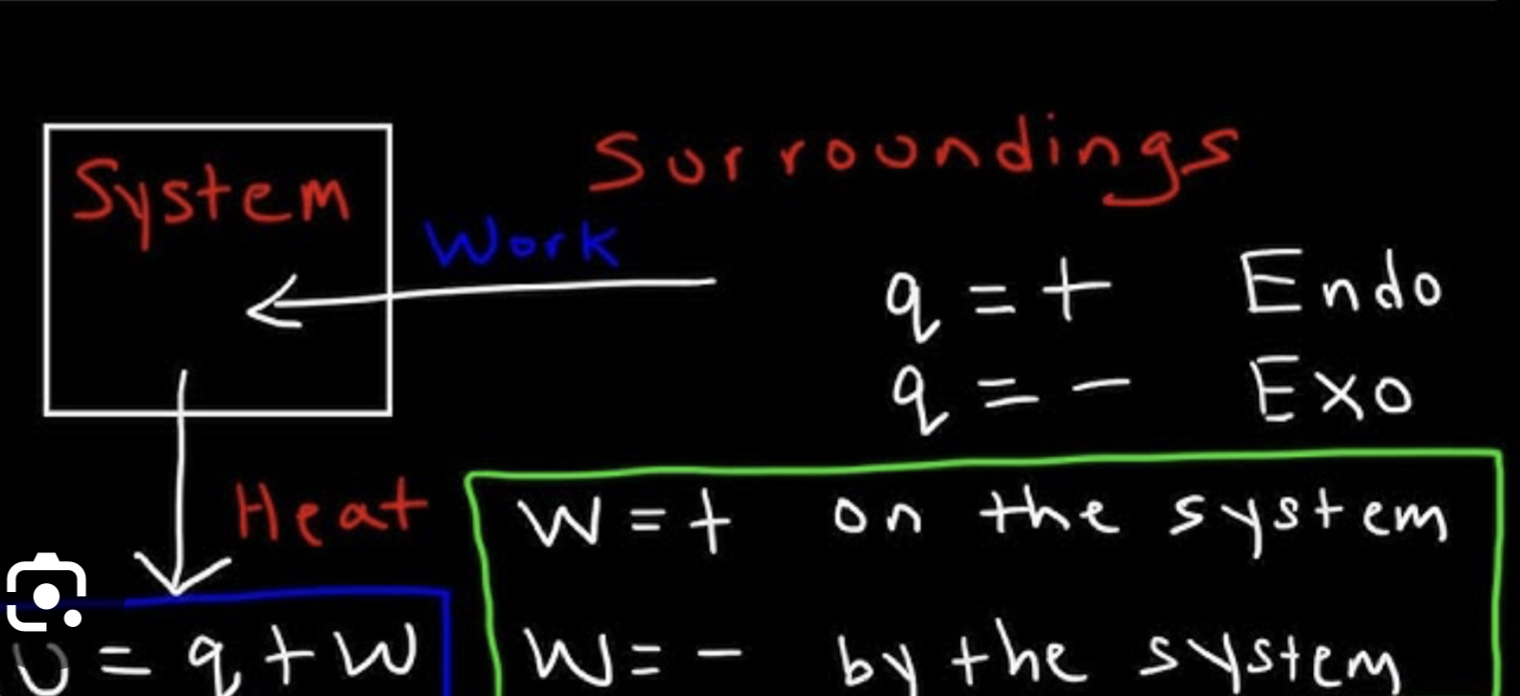
System does work ON surroundings →
Surroundings does work ON systems →
Answer
A 102g piece of silver with heat capacity 0.235 J/gºC absorbs 56.7 J of heat, if its initial temperature was 5.0ºC what is the temperature after absorbing this heat?
Reminder: if everything is the same unit, you don’t have to convert anything to liters or Kelvin (keep this in mind for specific heat capacity problems)
Answer: around 7.36
What is the sign for work in the following situations?
a. A balloon expands positive
b. Gas in a rigid container (constant volume) is warmed negative
c. A weight is placed on the top of a cylinder causing the volume to decrease negative
Trick: look at the equation sheet for work
“constant volume” = means the volume is 0
a → negative
b → zero
c → positive
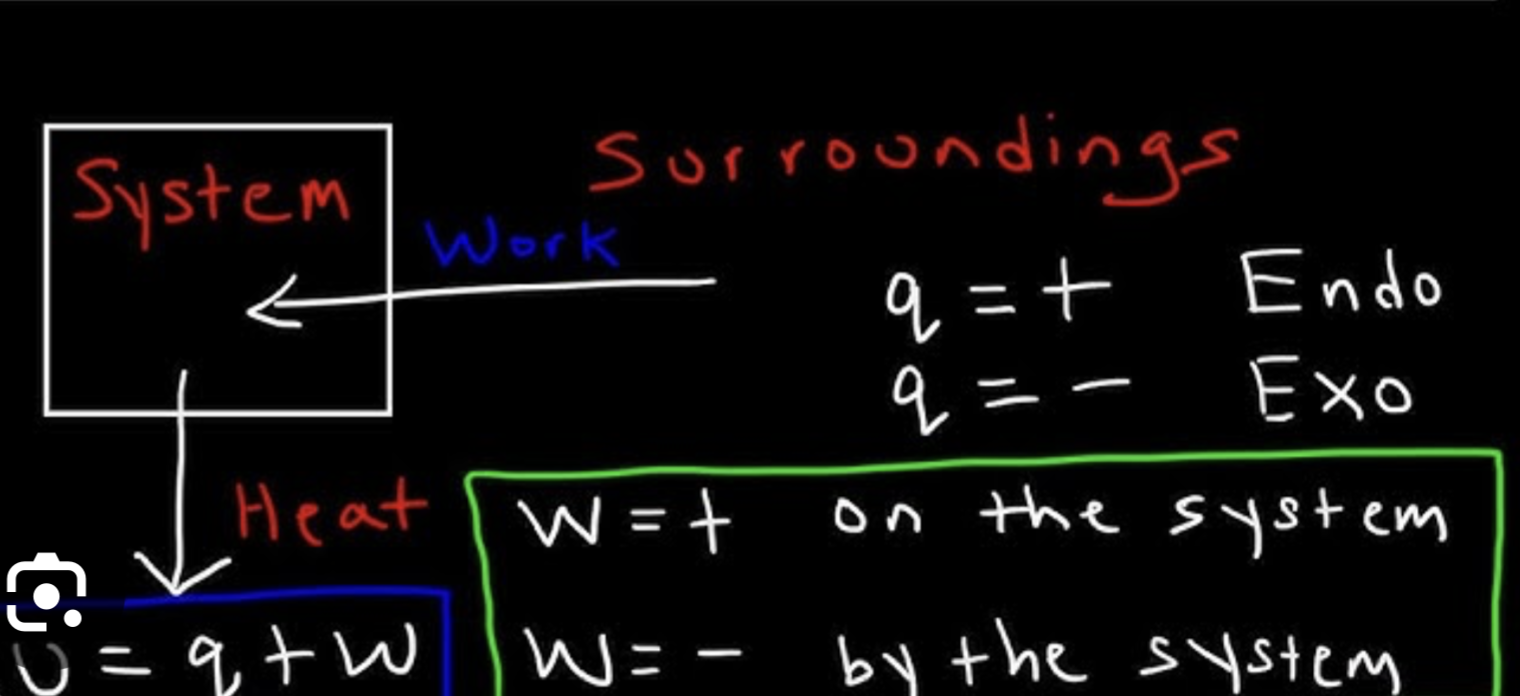
Bomb calorimetry volume
0
Coffee cup calorimetry pressure
0
When work is small…
heat, enthalpy, and internal energy are around equal
Draw O2 lewis structure
O2 = 2(6) = 12 valence electrons
O-O
Count formal charge: Valence electrons—(number of lone dots+number of lines) → do this for each individual atom, make sure it’s 0. and add the charges together
Make sure it meets the octet rule: for each atom, count all the electrons (but this time, each line is equal to 2 electrons, not 1 electron)
O-O doesn’t meet this, so add one more line
O double bond O → correct
How to Determine Enthalpies of Reaction from Bond Energies
Draw the CORRECT lewis structures
Count the number of types of bonds in the reactants and add all of the energies together
Count the number of types of bonds in the products and add all of the energies together
Energy of reactants—energy of products = final answer
IF GIVEN A TABLE THAT SHOWS REACTIONS:
(look at OChem tutor YouTube video)
How to know if a compound has a standard enthalpy of zero?
If the element is in its natural state
ex) Pb → lead, so natural state is solid, so if' it’s in solid form it’s standard enthalpy of zero
Combustion reactions
O2 is reactant, CO2 and H2O are products
all are exothermic
molecules that O2 reacts with are called the fuel
How to calculate standard enthalpy of formation
Sum of products — sum of reactants
(if a compound is in its most stable state, then you write it as 0)
Endo vs exo
Endothermic reactions absorb heat energy from their surroundings, causing a decrease in temperature
exothermic reactions release energy into their surroundings, causing a temperature increase
Endo vs exo: enthalpy
Endo: positive enthalpy
exo: negative enthalpy
how to rank compounds based on vapor pressure
weaker IMFs result in higher vapor pressure, while stronger IMFs mean lower vapor pressure
Label each process as endothermic or exothermic
Vaporization:
Deposition:
Fusion:
Condensation:
Melting:
Sublimation:
Vaporization: Endothermic
Deposition: Exothermic
Fusion: Endothermic
Condensation: Exothermic
Melting: Endothermic
Sublimation: Endothermic
Sublimation
solid → gas
Deposition
gas → solid
Draw a phase diagram
Image