Lec 10- Regulation of Oxidative Phosphorylation
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
what is key idea of the pathway of various fuels (list them), that transfer electrons to xxx (list them), how does oxidative phosphorylation work in regards to those molecules?
ENERGY FROM REDUCED FUELS IS USED TO SYNTHESIZE ATP
❖ CHO, lipids, and AA are reduced fuels
❖ Electrons are transferred to cofactors NAD or FAD
❖ In oxidative phosphorylation, energy from NADH and FADH2 is
used for ATP synthesis
Mitochondria: where does DNA come from? how many genes, how many proteins? what are cristae? what tissues contain more mitochondria? what is involved w oxidative phosphorylation here?
Mitochondrial DNA is maternally inherited—. from mom!
Mitochondiral genom cotains only 37 genes – 22 tRNA, 2 rRNA and 13 protein coding genes
14 proteins: 13 proteins (subunits/isoforms) of electrontransporters
aprox 1200 proteins found in mitochondria; 300 have unknown function
-cristae = folds to increase the surface area
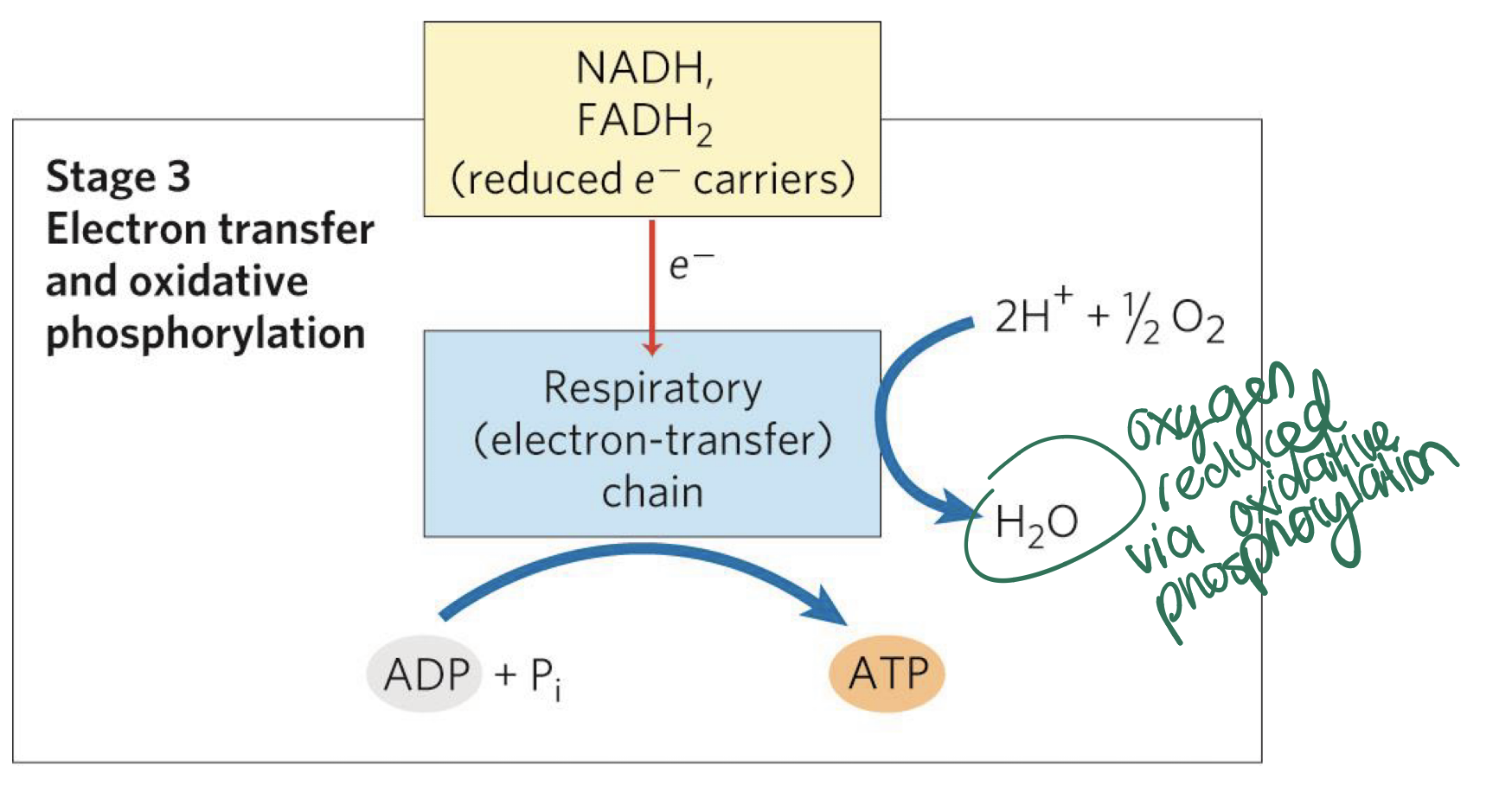
-Oxidative phosphorylation involves reduction of O2 to
H2O using electrons donated from NADH and FADH2-
what is chemiosmotic theory
= energy derived from electron transfer reactions is temporarily stored as a transmembrane
difference in charge and pH, which subsequently drives formation of ATP in oxidative phosphorylation
what is ubiquinone/ coenzyme Q? what is reduced/redical/oxidized form
electron carrier that picks up electrons from Complex I and Complex II and delivers them to Complex III.
FULLY OXIDIZED= Ubiquinone - Q
SEMIQUINONE RADICAL= Ubisemiquinone - ⋅QH
FULLY REDUCED= Ubiquinol QH2
what are cytochromes? where does it carry xxx from?
= proteins that contain a Heme group
three types: a, b,c
shuttles electrons from Complex III to Complex IV.
what are iron-sulfur proteins?
= proteins that contain iron in association with sulfur atoms and/or with the sulfur atoms of Cys
residues in the protein participate in one-electron transfers
they stay put, don’t really move between complexes like cyt c.
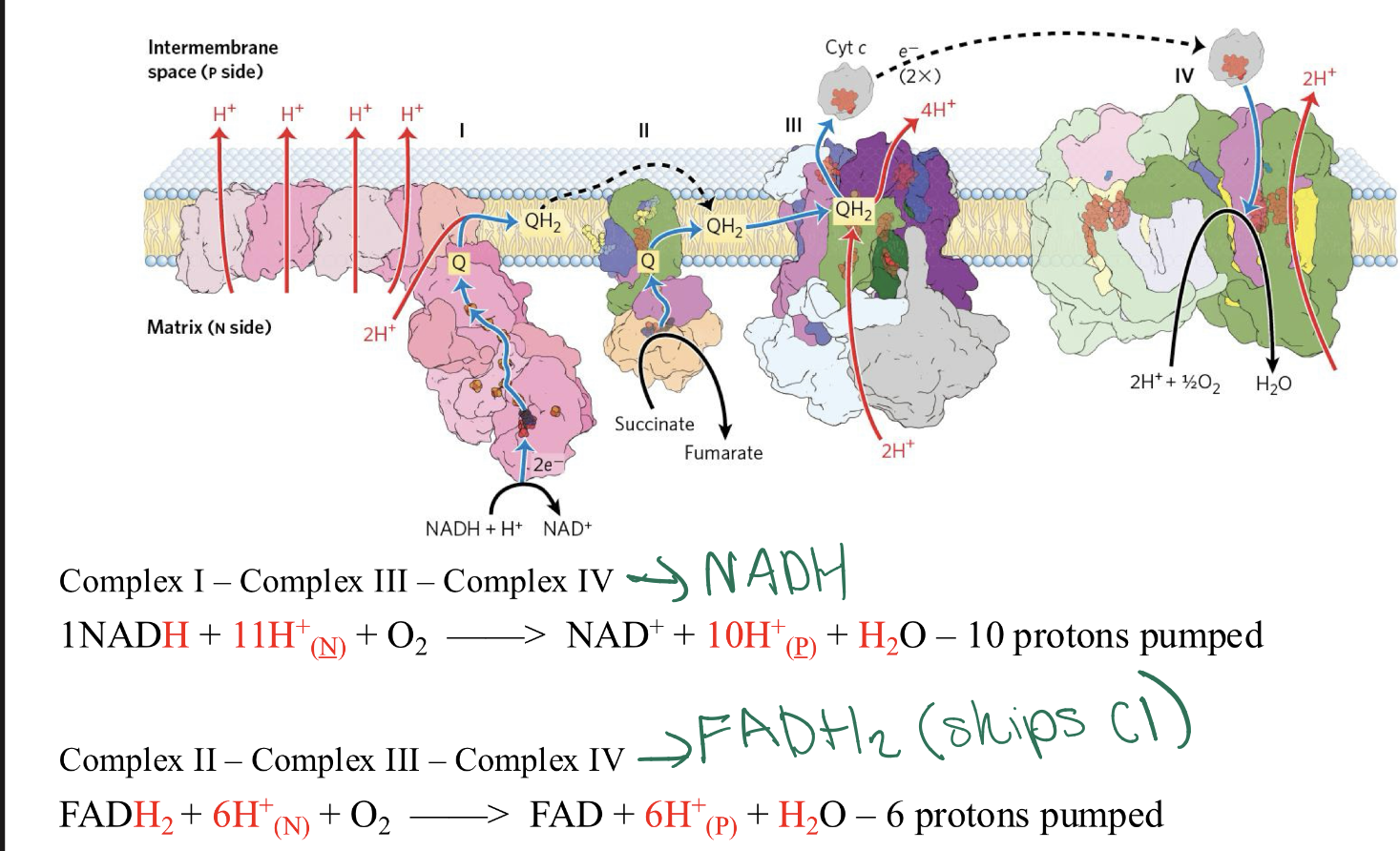
overview of reactions in ETC: complex I? complex II? complex III? complex IV?
CI=will catalyzes the electron transfer FROM NADH TO ubiquinone, pumps 4 protons
CII= will catalyzes the electron transfer FROM FADH2 TO ubiquinone, does NOT pump protons
CIII= carries electrons from ubiquinol (ubiquinone now fully reduced) to cytochrome C
CIV= completes the sequence by transferring electrons from cytochrome C to O2
Complex I: what does it contain? what is it? what are subunits? what happens here what is overall rxn in this complex?
= NADH-QUINONE OXIREDUCTASE
CONTAINS: 45 diff polypeptide chains, including FMN- containing
flavoprotein and at least 8 iron-sulfur centers.
IT IS: a proton pump driven by the energy of electron transfer.
THREE SUBUNITS: Nqo12, Nqo13, and Nqo14 related to H+ pumpin
WHAT HAPPENS:
NADH donate electrons to ubiquinone and are once again available as NAD—> 4 protons H+ are pumped to the intermembrane space, generating an
electrochemical gradient
Electrons travel through a Flavin mononucleotide (FMN) and several Iron-Sulphur (Fe-S) clusters.
The Result: The energy from these electrons allows Complex I to pump 4 protons into the intermembrane space.
Handoff: The electrons are given to Ubiquinone (Q).
OVERALL RXN= NADH + Q + 5H+(from matrix) = NAD+ + QH2 + 4 H+(into inter membrane)
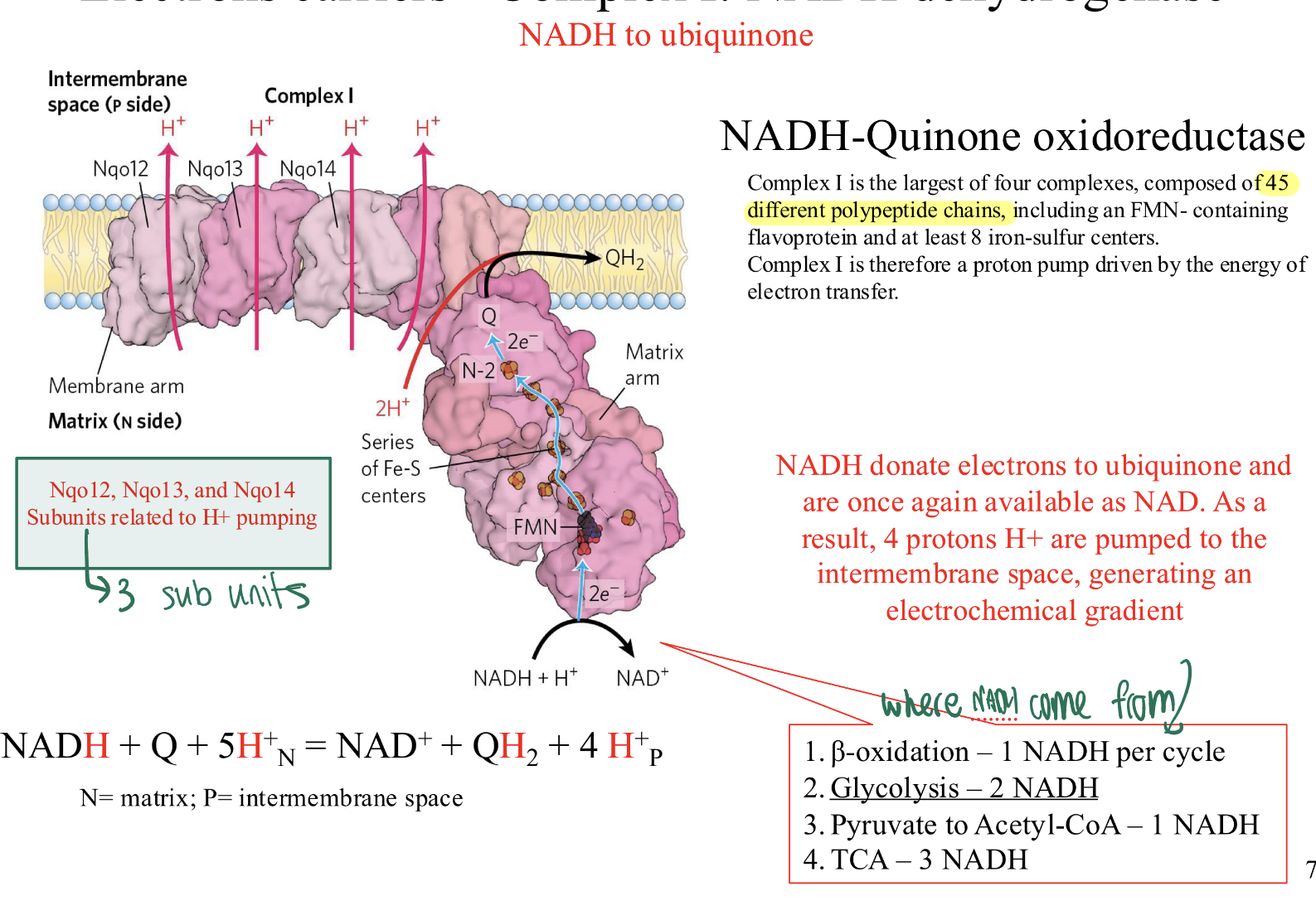
where does complex I get its electrons passed from? what processes can generate this molecule.
NADH passes its electrons through complex I to ubiquinone
NADH may come from:
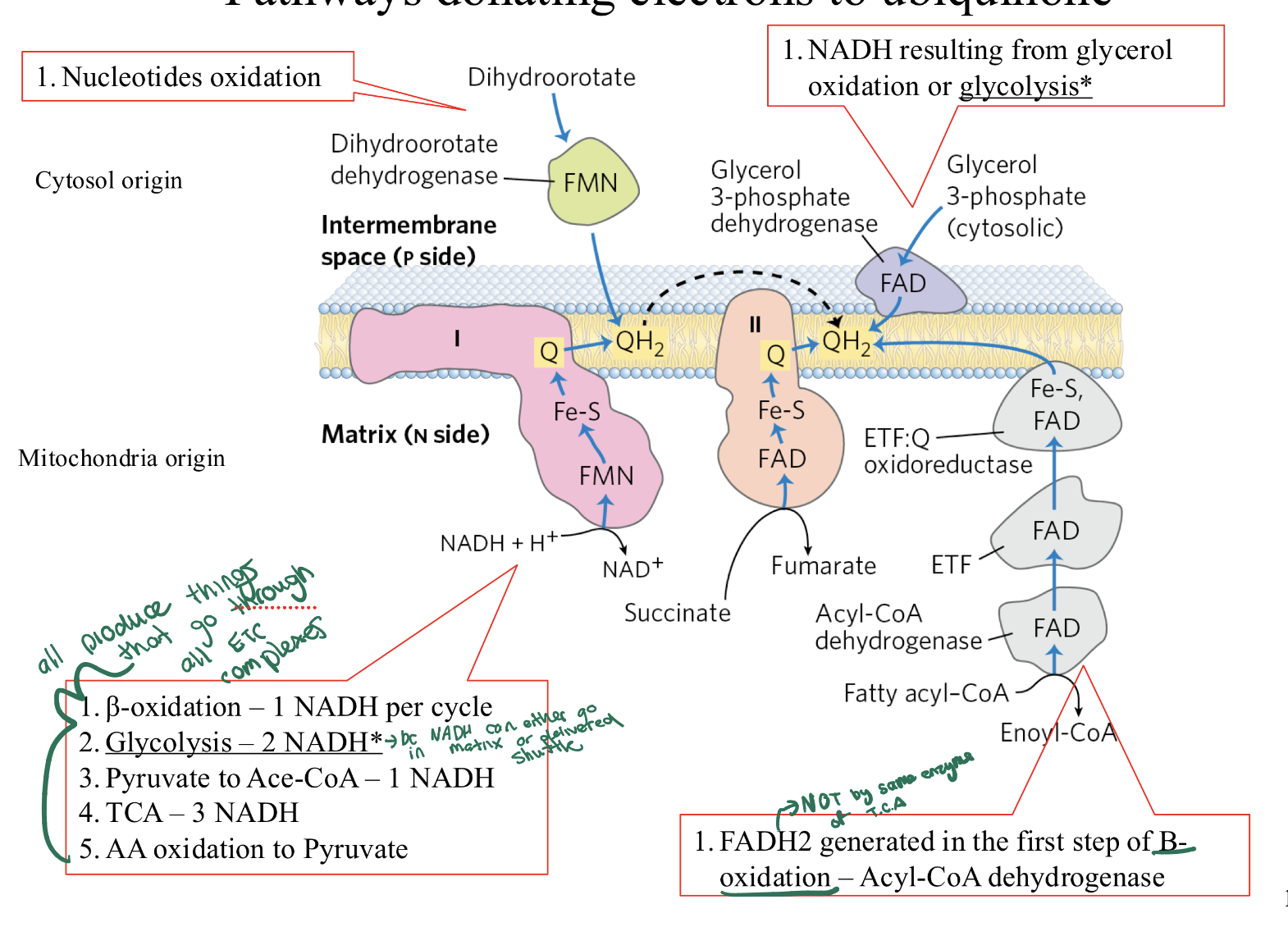
1. β-oxidation – 1 NADH per cycle
2. Glycolysis – 2 NADH
3. Pyruvate to Acetyl-CoA – 1 NADH
4. TCA – 3 NADH
why does NADH produces more ATP than FADH2 in the ETC?
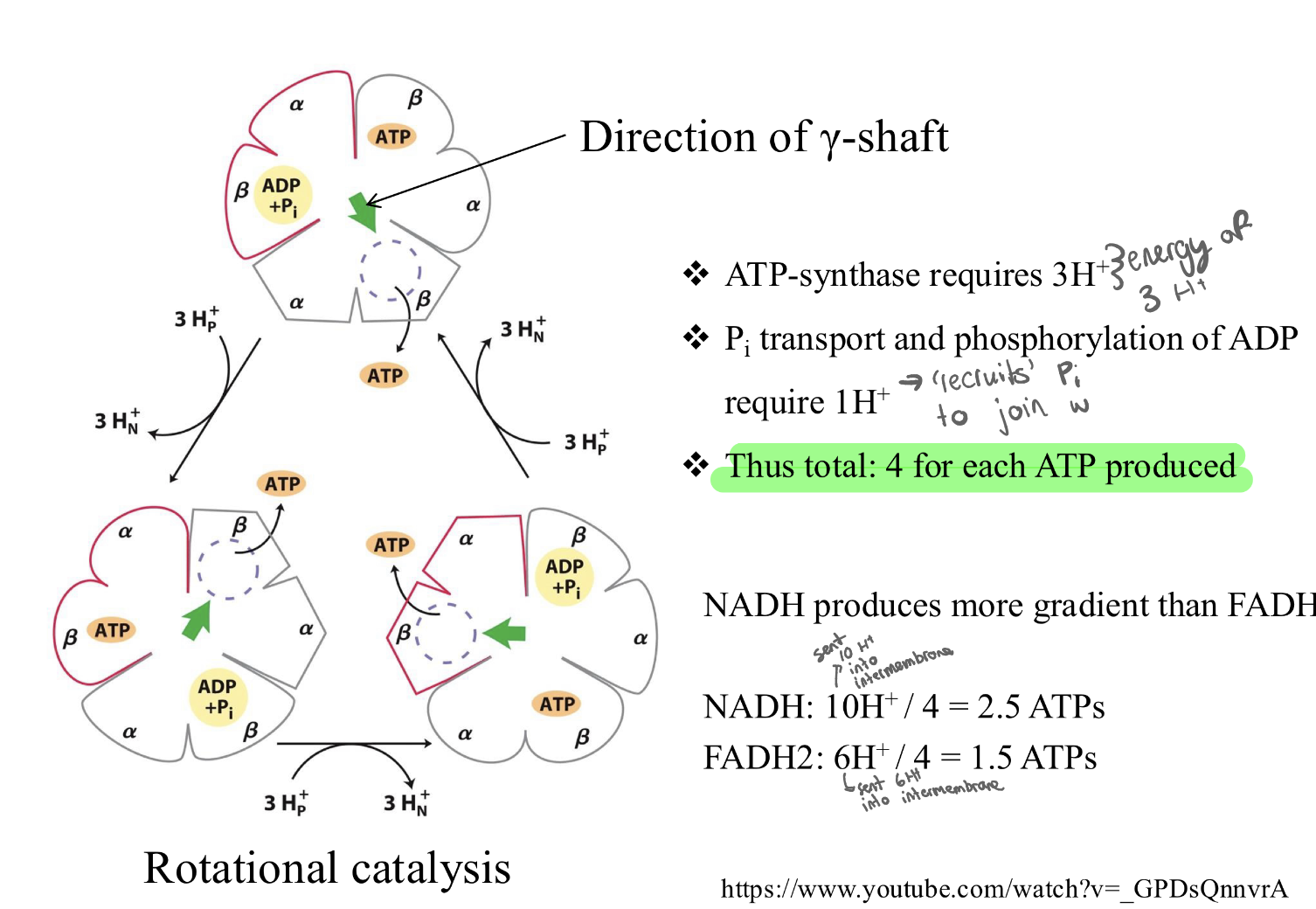
NADH produces more ATP than FADH2:
NADH enters at Complex I
→Pumps 10 protons total (4 from I, 4 from III, 2 from IV).
FADH2 enters at Complex II
→Pumps 6 protons total (0 from II, 4 from III, 2 from IV).
Since it takes about 4 protons to make 1 molecule of ATP, you get roughly 2.5 ATP from one NADH, but only 1.5 ATP from one FADH2.
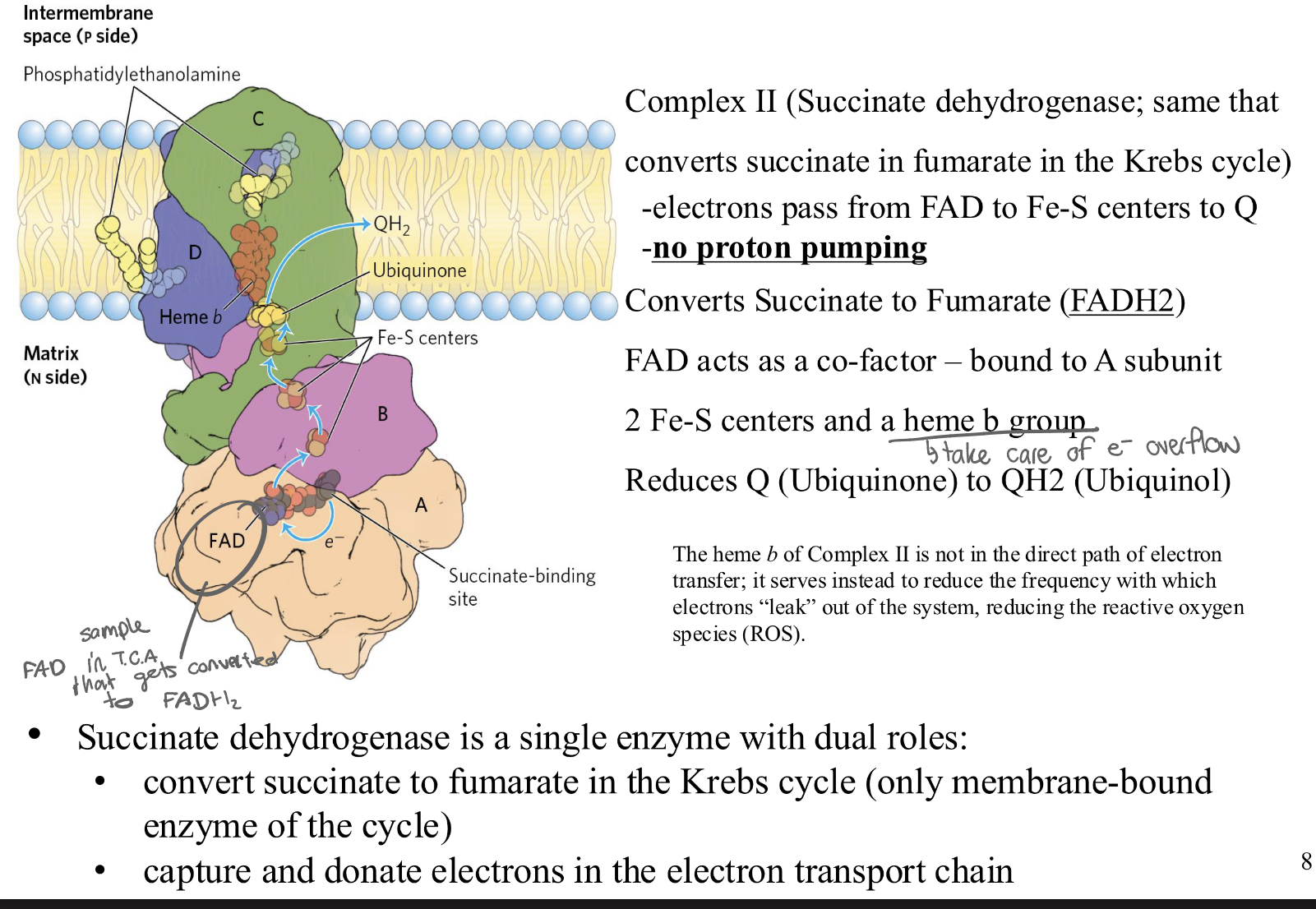
Complex II: what does it contain? what is it? what are subunits? what happens here what is overall rxn in this complex?
= succinate dehydrogenase
-electrons pass from FAD to Fe-S centers to Q
-NO proton pumping
❖ Converts Succinate to Fumarate (FADH2)
❖ FAD acts as a co-factor – bound to A subunit
❖ 2 Fe-S centers and a heme b group (cyt b)
❖ Reduces Q (Ubiquinone) to QH2 (Ubiquinol)
DUAL ROLES:
• convert succinate to fumarate in the TCA cycle (it’s the only membrane-bound
enzyme of the TCAcycle)
• capture and donate electrons in the electron transport chain
Start: Succinate (from the Krebs cycle) arrives at Complex II.
Reduction of FAD: Succinate drops off 2 electrons and 2 protons onto the FAD that is "bolted" to the protein. The FAD becomes FADH2( stays in CII)
The Relay: Those electrons then leave the FADH2 and travel through the Iron-Sulphur (Fe-S) centers (the "wire").
Reduction of Q: Finally, those electrons (and 2 protons from the matrix) are handed off to Ubiquinone (Q).
Finish: Ubiquinone (Q) becomes Ubiquinol (QH2)
where does CIII obtain its ubiquinol from?
Source 1 (Complex I): One "empty" Q picks up electrons from NADH and becomes a full Ubiquinol
Source 2 (Complex II): Another "empty" Q picks up electrons from Succinate/FADand becomes a second full Ubiquinol
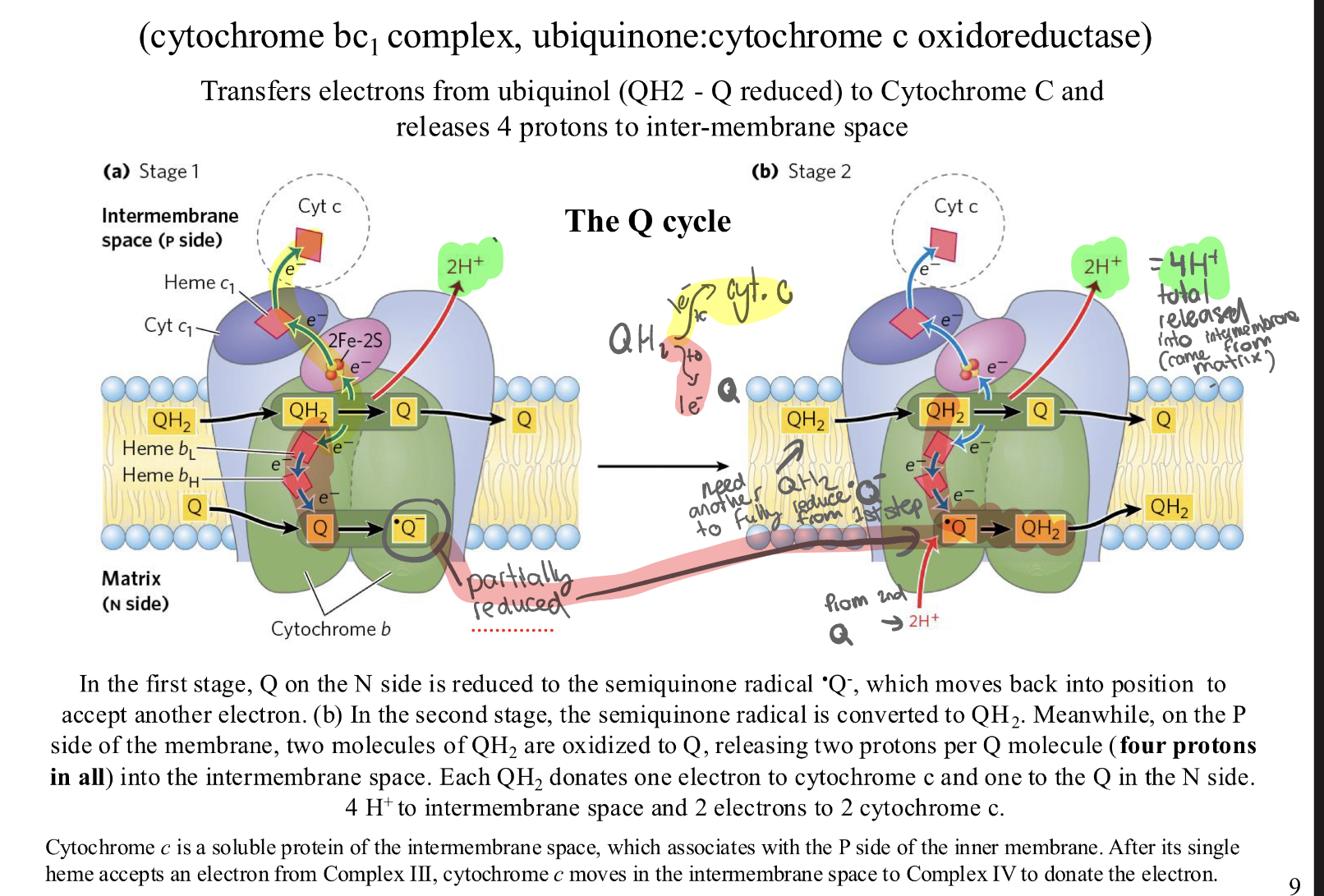
Complex III: what is the Q cycle and why does it occur?
WHY—> Ubiquinol (QH2) carries two electrons, but Cytochrome c is a "one-seater"—it can only take one electron at a time. To fix this, Complex III splits the pair of electrons.
Stage 1: The First Handoff
One QH2 molecule (enters Complex III.
Electron 1 (The "Up" Path): Goes through an Iron-Sulphur center (2Fe-2S) and Cytochrome c1 to Cytochrome c. This electron is now on its way to Complex IV.
Electron 2 (The "Down" Path): Goes through Heme 𝑏𝐿 and Heme bH to an empty Qmolecule waiting at the bottom.
The Result: The waiting Q becomes a Semiquinone Radical ⋅Q−
Protons: 2H+ pumpled into the intermembrane space (P-side).
Stage 2: Completing the Circuit
A second QH2 molecule enters the complex.
Electron 1 (The "Up" Path): Goes to a second Cytochrome c.
Electron 2 (The "Down" Path): Goes down to that "half-full" Semiquinone ⋅Q−
The Recycling: That Semiquinone picks up 2 H+ from the Matrix (N-side) and becomes a second QH2
Protons: Another 2 H+ pumped out into the intermembrane space.
OR : Stage 1 (The Left Side of your Image)
A (Ubiquinol) swims up to the top (the P-side).
Complex III "strips" its 2 electrons.
The Drop-off: The 2 protons that were holding onto those electrons are now "homeless." They fall off into the Intermembrane space. (Look at the first 2H+ in the green box).
The "Parking": One electron goes to Cytochrome c, but the other electron goes to a waiting Q at the bottom (N-side).
Stage 2 (The Right Side of your Image)
A second QH2 swims up to the top (the P-side).
Again, it is "stripped" of its electrons.
The Drop-off: Its 2 protons also fall off into the Intermembrane space. (Look at the second 2H+ in the green box).
The Pickup: One electron goes to Cytochrome c, and the other goes down to that "parked" electron from Stage 1.
The Refill: Now that the "parked" spot has two electrons again, it grabs 2 new protons from the Matrix to become a full QH2 again
The Final "Proton Math" (The Green Box at the top right)
Look at the green math at the top right of your slide:
Released (at the top): 4 protons total were dumped into the Intermembrane space because two QH2 "backpacks" were opened at the top.
Taken (at the bottom): 2 protons were sucked out of the Matrix to "re-fill" the recycled QH2
how is complex Iv inhibited?
the electron transfer to O2 is inhibited by cyanide and carbon monoxide
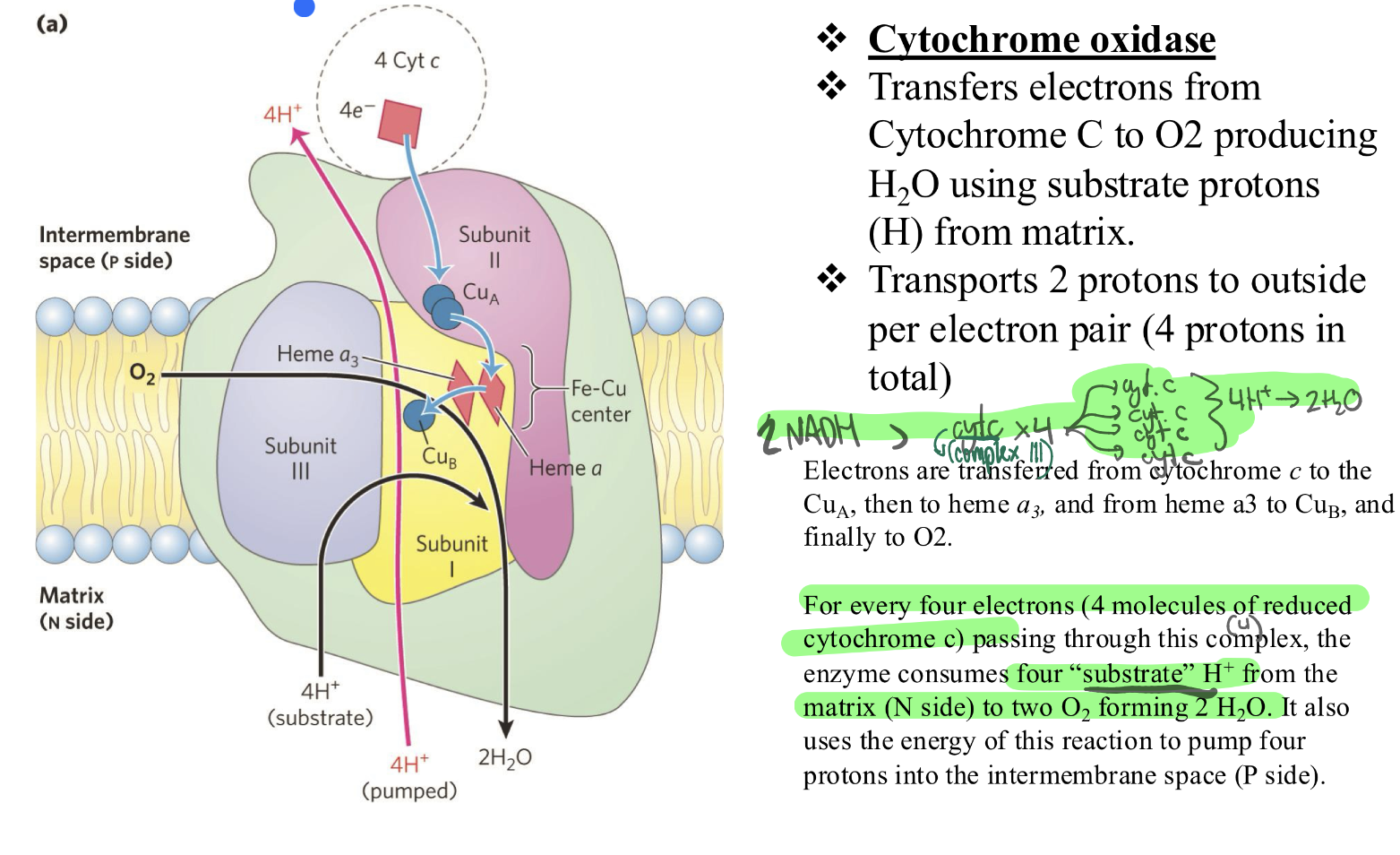
CIV: what does it do? where do electrons come from?
= cytochrome oxidase
❖ Transfers electrons from Cytochrome C (coming straight from CIII) to O2 producing
H2O using substrate protons from matrix.
❖ Transports 2 protons to outside per electron pair (4 protons in
total)
Inside the Complex IV protein: two different physical paths for protons:
One path leads straight to the Oxygen to make water.
The other path leads all the way Through the Protein to the other side of the membrane.
The "electricity" from the falling electrons is strong enough to force protons through bothpaths at the same time.
SO:
For every one O2 molecule (which requires 4 electrons), the complex makes twoseparate withdrawals from the Matrix bank:
Withdrawal A: The "Ingredients" (4 Protons)
These protons are taken into the "active site" of the protein.
They are physically glued to the oxygen and the electrons.
Result: They disappear from the matrix and become part of H2O molecule.
Total cost: 4H+
Withdrawal B: The "Export" (4 Protons)
While the water is being made, the complex uses the "leftover" energy to grab 4 more protons from the matrix.
Instead of making water with them, it shoves them through a separate tunnel and kicks them out into the Intermembrane Space.
Total cost: 4H+
in complex IV, what happens with 2 NADH
For every one NADH, you get 2 electrons. To make two water molecules
you need a total of 4 electrons.
So, with 2 NADH, here is exactly what happens at Complex IV:
The Electrons (The Fuel): You have 4 electrons (2 from each NADH).
The Water (The Chemistry): Those 4 electrons combine with 1 Oxygen molecule and 4 protons from the matrix to make 2 water molecules.
The Pumping (The Work): While those electrons are moving through the "wire" to meet the oxygen, they release a burst of energy. Complex IV uses that energy to grab 4 more protons and pump them into the intermembrane space.
Result: With just 2 NADH, you have produced 2 water molecules AND pumped 4 protons. It's not "one or the other"—it’s a simultaneous reaction.
NADH in ETC VS FADH2 in ETC
THIS reflect the Difference in number of protons transported reflects differences in ATP
synthesized
NADH produces more electrochemical gradient than FADH2 —> bc FADH2 can’t be used in C1 so 4 less H+ pumped out
what are the diff ways ubiquinone can be reduced to ubiquinol ?
Concept:
Q=is the universal collector for electrons from carbohydrates, fats, and nucleotides. All
QH2 created here travels to Complex III.
The 5 Donors:
Complex I: From NADH (TCA, Glycolysis, β-ox).
Complex II: From Succinate (TCA cycle).
Electron-transferring Flavoprotein:Q Oxidoreductase: From Fatty Acid β-oxidation (via ETF).
Glycerol 3-P Dehydrogenase: From Cytosolic NADH (Glycerol shuttle).
To FMN Dehydrogenase From Nucleotide Synthesis.
Key Note: Only Complex I pumps protons before reaching Q. All other "side doors" (2, 3, 4, 5) enter the chain without pumping protons at the start.
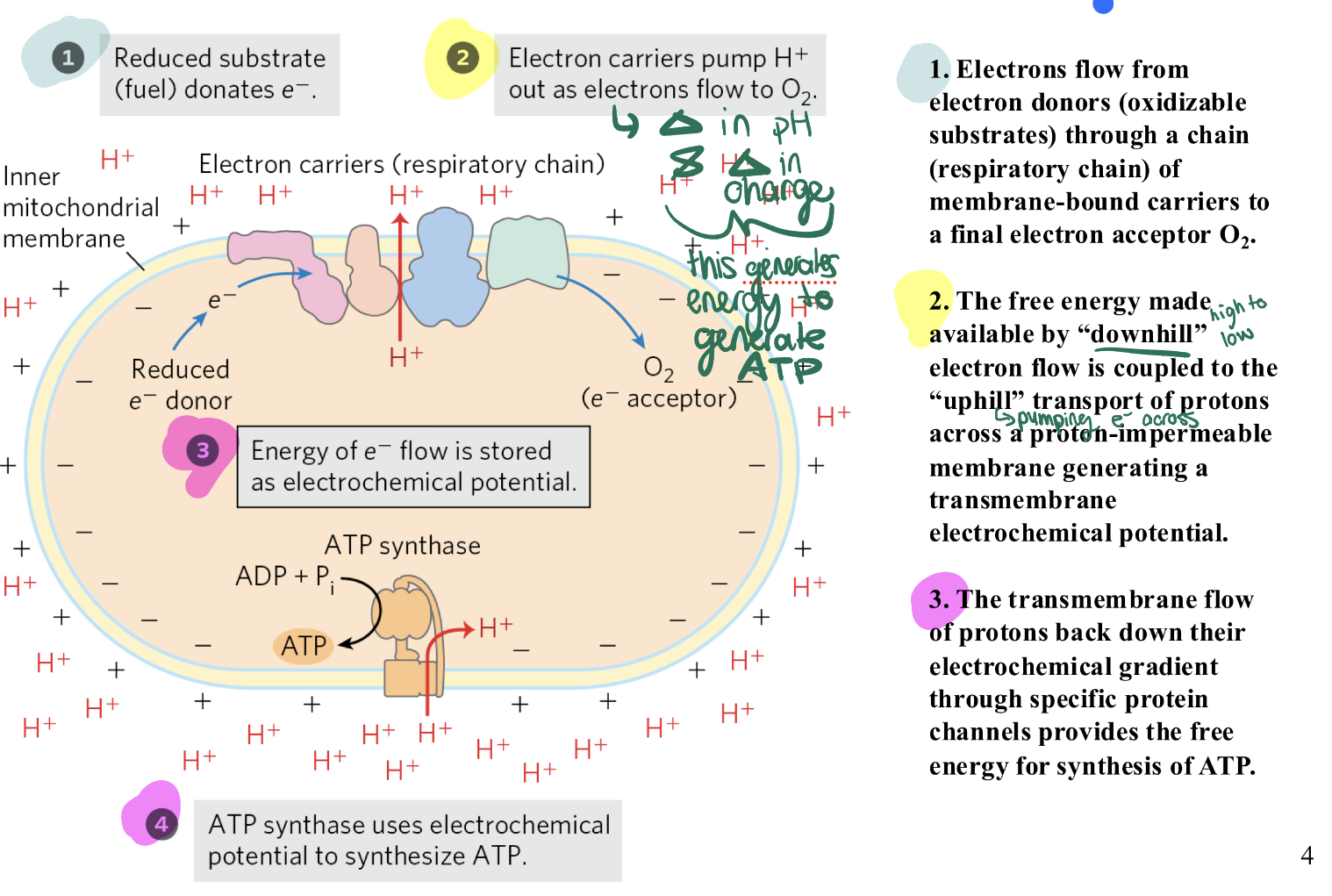
ATP synthase: how is electrochemical gradient created? where do p+ get pumped? what gets reduced and what gets oxidized? how does inter membrane vs Matrox differ in H+ and charge?
electrochemical proton gradient is created by:
❖ active transport of protons across the membrane
Complex I and IV (they use the energy from electrons to physically push protons against a "crowd")
❖ Release of protons into intermembrane space
Oxidation of QH2 (complex III)
❖ Chemical removal of protons from the matrix
Reduction of Q (complex I, II, III) and oxygen (complex IV)
P-Side (Intermembrane Space): High[H+] Acidic, Positive charge.
N-Side (Matrix): Low [H+], Alkaline, Negative charge.
![<p><u> electrochemical proton gradient is created by:</u></p><p>❖ active transport of protons across the membrane</p><ul><li><p>Complex I and IV (t<span><span>hey use the energy from electrons to physically </span></span><strong>push</strong><span><span> protons against a "crowd")</span></span></p></li></ul><p>❖ Release of protons into intermembrane space</p><ul><li><p> Oxidation of QH2 (complex III)</p></li></ul><p>❖ Chemical removal of protons from the matrix</p><ul><li><p> Reduction of Q (complex I, II, III) and oxygen (complex IV)</p></li></ul><p><span><strong><u><span>P-Side (Intermembrane Space):</span></u></strong><u><span> High[H+] </span></u><strong><u><span>Acidic</span></u></strong><u><span>, </span></u><strong><u><span>Positive</span></u></strong><u><span> charge.</span></u></span></p><p><span><strong><u><span>N-Side (Matrix):</span></u></strong><u><span> Low [H+], </span></u><strong><u><span>Alkaline</span></u></strong><u><span>, </span></u><strong><u><span>Negative</span></u></strong><u><span> charge.</span></u></span></p><p></p>](https://assets.knowt.com/user-attachments/8395403a-73c3-4574-9543-8e78cfd7c951.png)
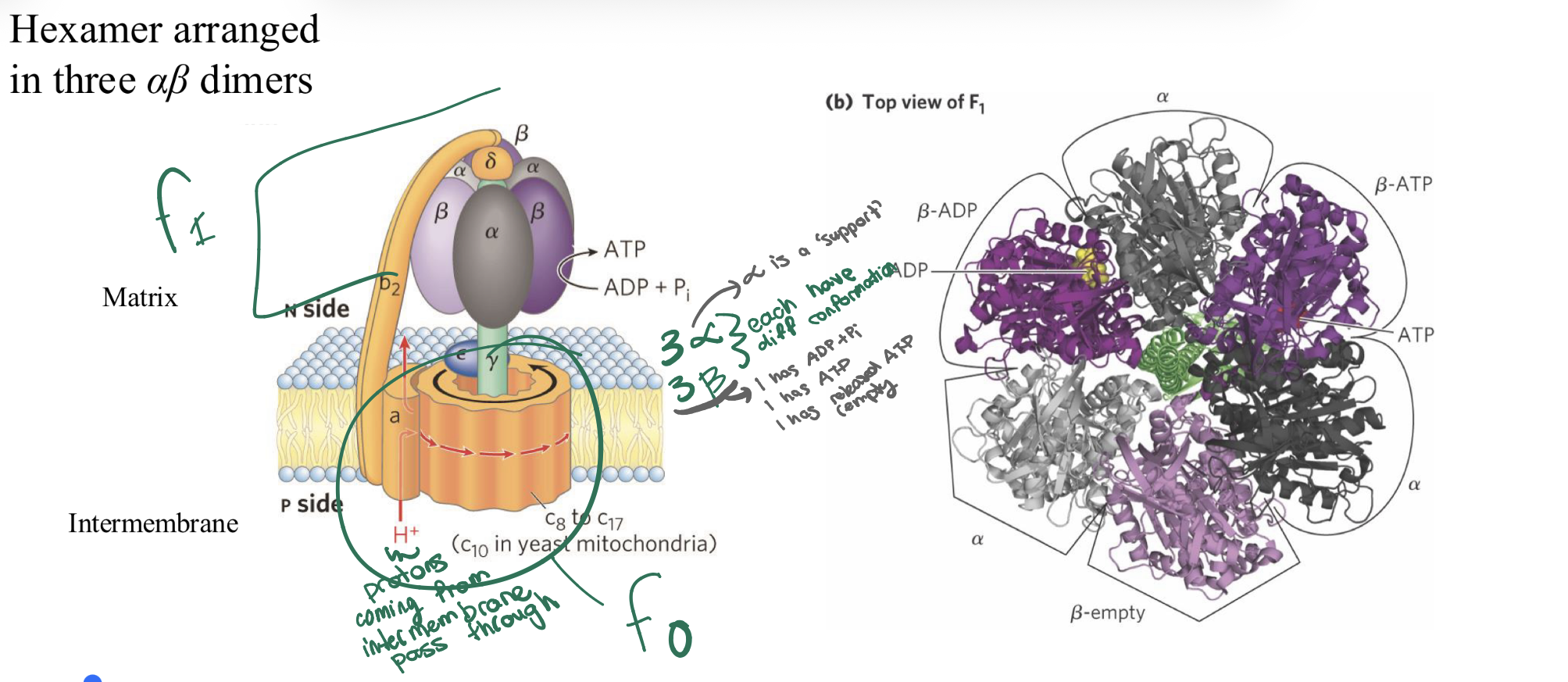
what are two units of ATP synthase? what is difference in alpha and beta subunits?
❖ Fo: membrane bound; transports protons down the gradient;
transfers energy to F1 to catalyze phosphorylation of ADP
❖ F1: in the matrix; catalyzes the hydrolysis of ATP
binding change of F0F1 ATP synthase: how many H+ required for 1 ATP?
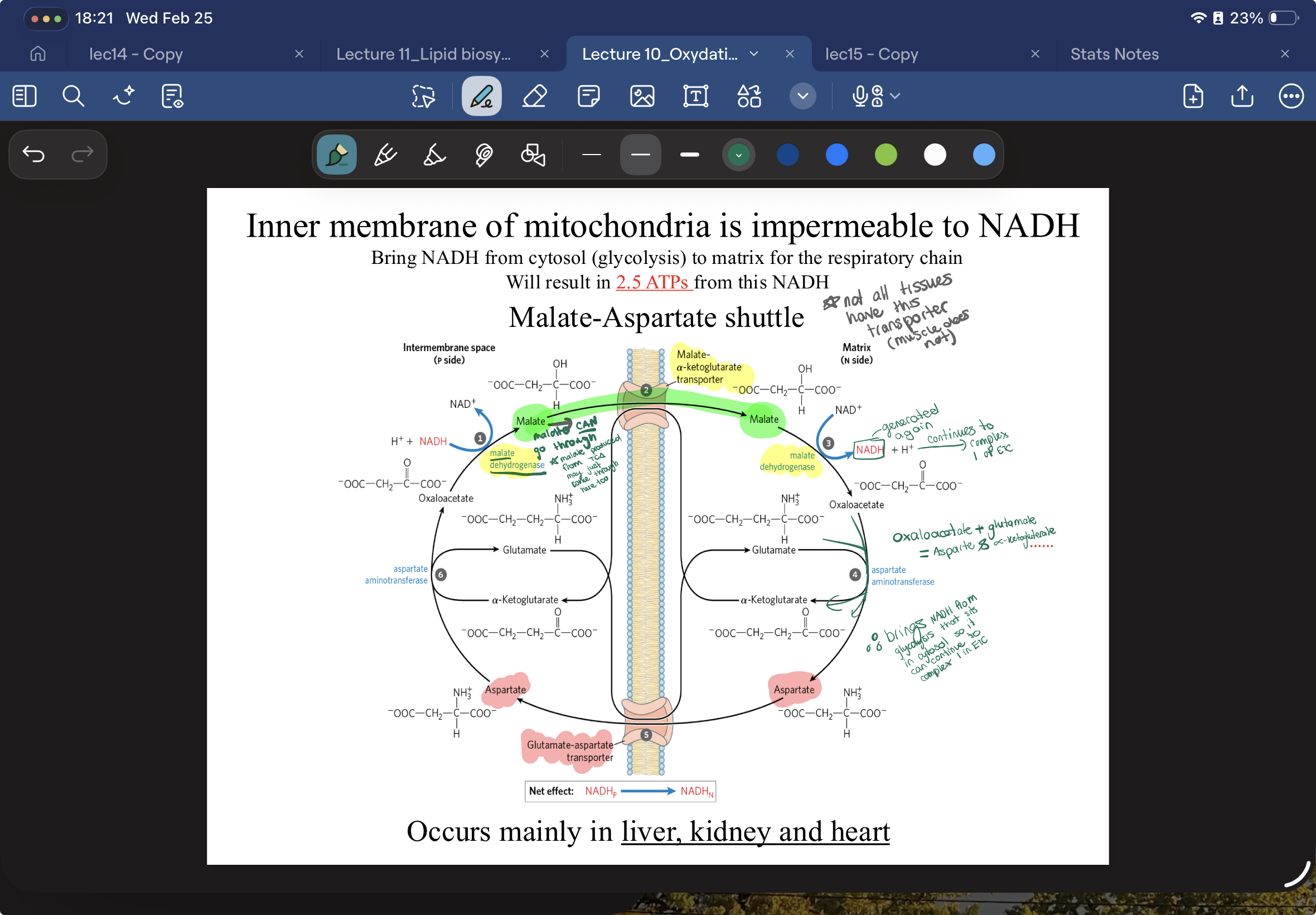
how is NADH from glycolysis able to be used in the respiratory chain of mitochondria? what tissues doe this occur in?
IMM is impermeable to almost everything, including NADH and Protons, so NADH must use MALATE ASPARTATE SHUTTLE
How it works:
In the cytosol, NADH gives its electrons to Oxaloacetate, turning it into Malate.
The Crossing: There is a specific transporter that will let Malate into the mitochondria. (MALATE-ALPHA-KETOGLUTERATE TRANSPORTER)
The Handoff: Once inside, Malate turns back into Oxaloacetate and gives the electrons to a Mitochondrial NAD+ via MALATE DEHYDROGENASE—> creating Mitochondrial NADH.
Oxaloacete is converted to Aspartate which can pass back into inter membrane via GLUTMATE-ASPARTATE SHUTTLE…Aspartate back to oxaloacteate and cycle continues….
OCCURS in liver kidney heart bc they need a lot of ATP
if a tissue does not posses the Malate-aspartate shuttle, how does NADH from glycolysis get used in ETC? where does this occur?
Must use Glycerol 3-Phosphate shuttle
occurs in skeletal muscle and brain
Instead of entering in the matrix the NADH is delivered in the inner membrane
Will result in 1.5 ATPs from this NADH = FADH2
CI is skipped, enters at mitochondria G3P dehydrogenase (diff then CII) and reduced FAD+ to FADH2 which then donates its electrons to form Q𝐻2
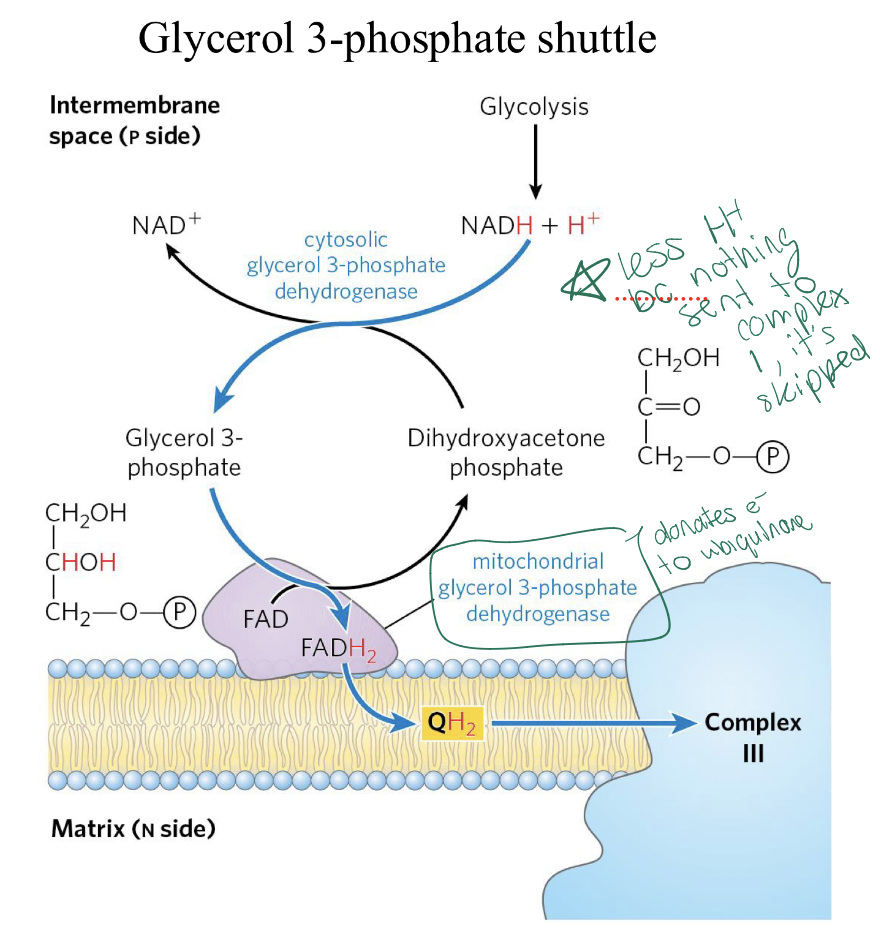
how many ATP per one NADH coming from Malate-aspartate shuttle VS Glycerol 3-Phosphate shuttle? how does this differ in the yield of total ATP per 1 glucose?
Malate-aspartate shuttle = 2.5 ATP per NADH
Glycerol 3-Phosphate shuttle= 1.5

Ox. Phosphorylation regulation: what is biggest controller ?what does inhibition of Ox. Phos cause?
Oxygen is biggest controller bc if no final electron acceptor, system stops
PRIMAIRILY REGULATED BY:
substrate availability
-ADP and Pi } too much OR too little—> this determines continuation or inhibition of Ox. Phos
THEN:
The whole process (cellular respiration) in general:
-ADP
-ATP, NADH
Inhibition of OxPhos leads to accumulation of NADH.
Causes a cascade feedback inhibition up glycolysis BC build up of reduced (high E) molecules and there’s no where to dump energy!
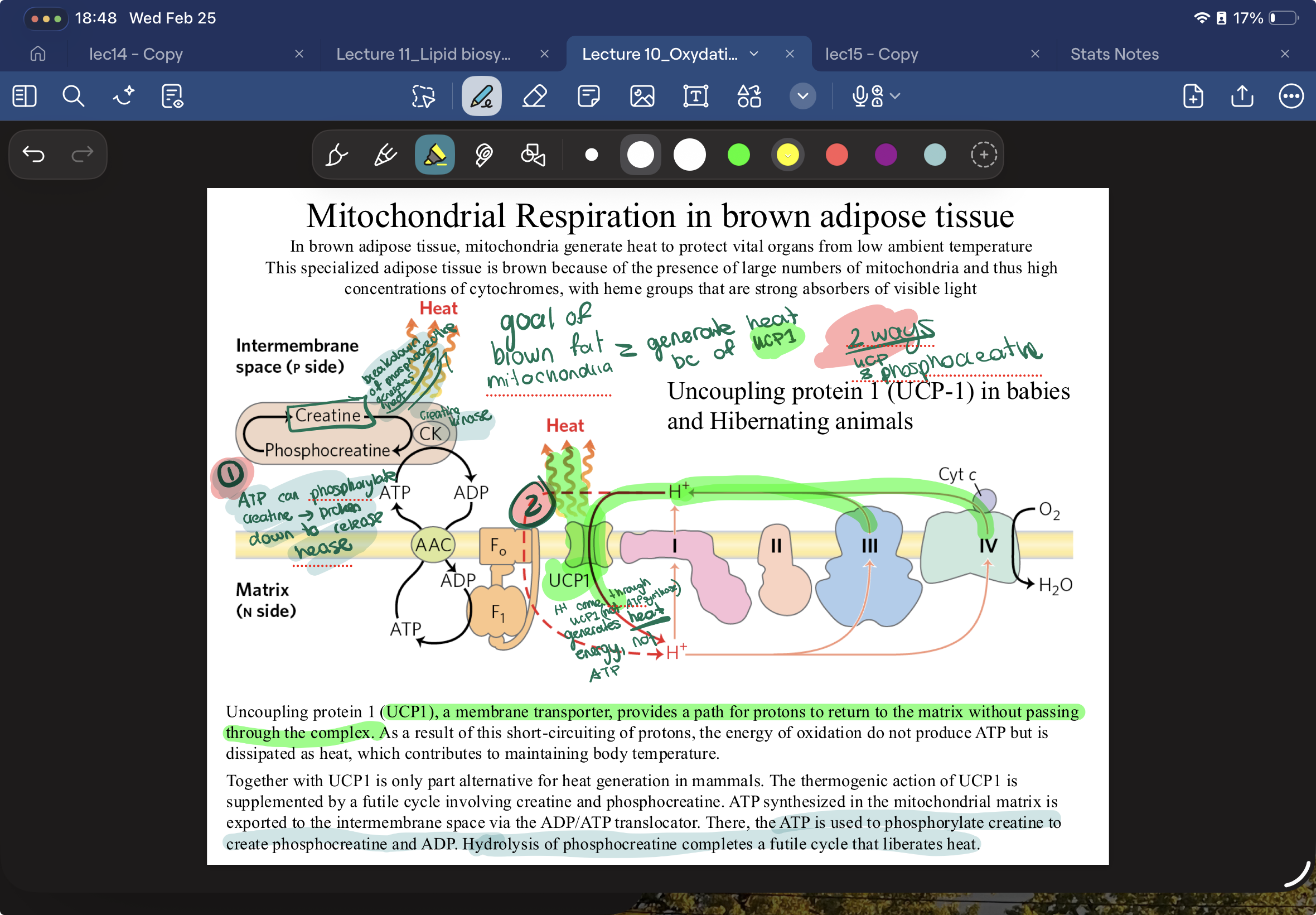
what are the two ways mitochondria in Brown fat tissue generate heat instead of JUST ATP ?
via UCP1, protons flow through here instead of ATP synthase and generate heat instead of ATP
AND
ATP used to phosphorylate creatine to create phosphocreatine and ADP. Hydrolysis of phosphocreatine liberates heat.
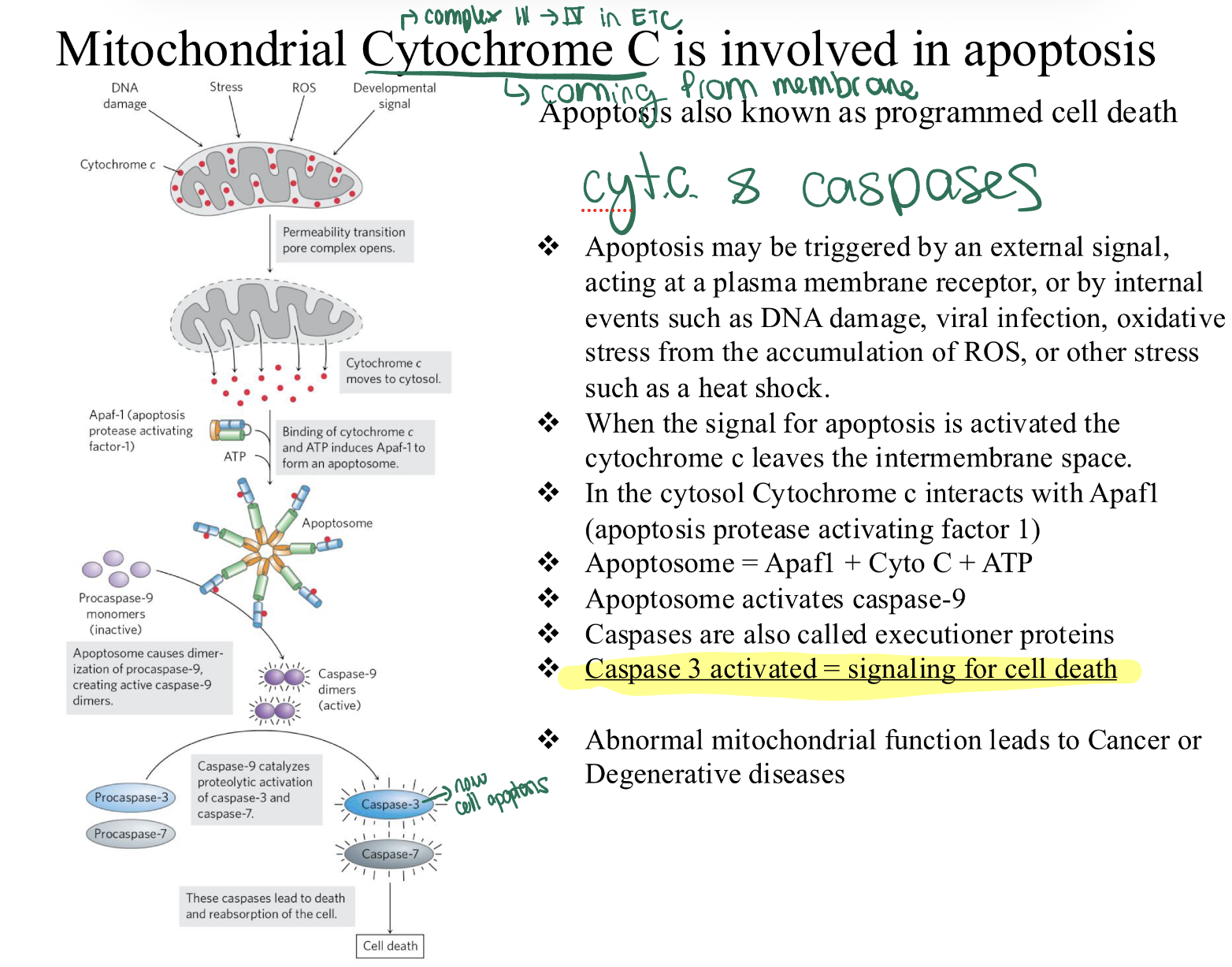
how is Cyt. C involved in apoptosis?
Stress/Signal—> Cyto C leaks into cytosol.
Cyto C + Apaf1 + ATP—> Apoptosome.
Apoptosome—> Activates Caspase-9.
Caspase-9—> Activates Caspase-3 (Executioner).
Result: Cell Death.
BASICALLY IF Cytochrome c is in the Cytosol: It is a death-trigger.